 |
 |
| Anim Biosci > Volume 34(2); 2021 > Article |
|
Abstract
Objective
Methods
Results
Conclusion
ACKNOWLEDGMENTS
Figure 1

Figure 2
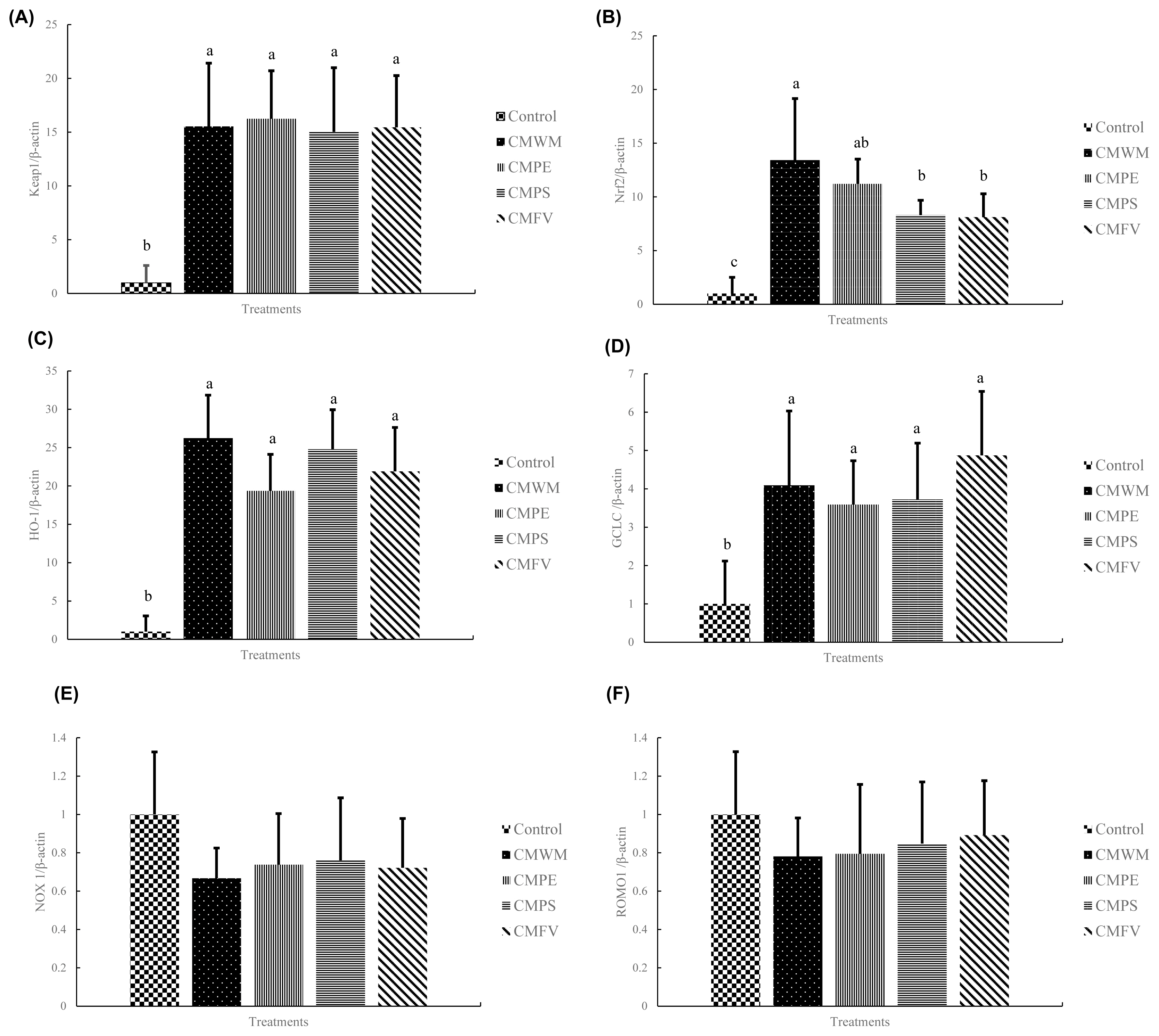
Table 1
| Items | Control1) | CMWM1) | CMPE1) | CMPS1) | CMFV1) |
|---|---|---|---|---|---|
| Ingredients | ----------------------------------------------------------------------- g/kg ---------------------------------------------------------------------- | ||||
| Yellow corn | 488.7 | 472.3 | 474.7 | 474.9 | 473.3 |
| Soybean meal (CP 44.0%) | 345.2 | 330.0 | 314.5 | 330.1 | 332.0 |
| Full fat soybean meal | 83.6 | 103.2 | 120.0 | 100.7 | 100.0 |
| Soybean oil | 35.3 | 37.3 | 33.6 | 37.1 | 37.5 |
| Calcium carbonate | 16.2 | 16.2 | 16.2 | 16.2 | 16.2 |
| Monocalcium phosphate | 18.6 | 18.6 | 18.6 | 18.6 | 18.6 |
| DL-methionine | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 |
| L-lysine-HCl | 3.7 | 3.7 | 3.7 | 3.7 | 3.7 |
| NaCl | 3.9 | 3.9 | 3.9 | 3.9 | 3.9 |
| Choline-Cl (50%) | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| Vitamin premix 2) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Mineral premix 3) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| CM | - | 10.0 | 5.0 | 5.0 | 5.0 |
| PE | - | - | 5.0 | - | - |
| PS | - | - | - | 5.0 | - |
| FV | - | - | - | - | 5.0 |
| Total | 1,000 | 1,000 | 1,000 | 1,000 | 1,000 |
| Calculated nutrient value | |||||
| ME (kcal/kg) | 3,050.0 | 3,050.0 | 3,050.0 | 3,050.1 | 3,050.1 |
| CP (% DM) | 23.0 | 23.0 | 23.0 | 23.0 | 23.0 |
| Calcium (% DM) | 1.05 | 1.05 | 1.05 | 1.05 | 1.05 |
| Total phosphorus (% DM) | 0.76 | 0.76 | 0.76 | 0.76 | 0.76 |
| Available phosphorus, (% DM) | 0.52 | 0.52 | 0.52 | 0.52 | 0.52 |
| Lysine (% DM) | 1.54 | 1.54 | 1.54 | 1.54 | 1.54 |
| Methionine+cystein (% DM) | 0.90 | 0.90 | 0.90 | 0.90 | 0.90 |
| Analyzed nutrient value | |||||
| Crude protein (% DM) | 23.0 | 22.9 | 22.9 | 23.1 | 23.2 |
| Crude fat (% DM) | 6.6 | 6.8 | 7.1 | 7.2 | 6.9 |
CP, crude protein; CM, Cordyceps militaris waster medium; PE, Pleurotus eryngii stalk residues; PS, Pleurotus sajor-caju stalk residues; FV, Fammulina velutipes stalk residues; CP, crude protein; ME, metabolizable energy; DM, dry matter.
1) Control, basal diet; CMWM, basal diet supplemented with 1% CM; CMPE, basal diet supplemented with 0.5% CM+0.5% PE; CMPS, basal diet supplemented with 0.5% CM+0.5% PS; CMFV, basal diet supplemented with 0.5% CM+0.5% FV.
Table 2
| Items | Control1) | CMWM1) | CMPE1) | CMPS1) | CMFV1) |
|---|---|---|---|---|---|
| Ingredient | --------------------------------------------------- g/kg --------------------------------------------------- | ||||
| Yellow corn | 536.0 | 522.8 | 523.0 | 523.0 | 523.0 |
| Soybean meal (CP 44.0%) | 280.8 | 263.8 | 291.4 | 291.5 | 291.5 |
| Full fat soybean meal | 100.0 | 117.3 | 86.0 | 86.0 | 86.0 |
| Soybean oil | 42.1 | 45.0 | 48.5 | 48.4 | 48.4 |
| Calcium carbonate | 13.4 | 13.4 | 13.4 | 13.4 | 13.4 |
| Monocalcium phosphate | 16.6 | 16.6 | 16.6 | 16.6 | 16.6 |
| DL-methionine | 1.3 | 1.3 | 1.3 | 1.3 | 1.3 |
| L-lysine-HCl | 3.2 | 3.2 | 3.2 | 3.2 | 3.2 |
| NaCl | 3.8 | 3.8 | 3.8 | 3.8 | 3.8 |
| Choline-Cl (50%) | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| Vitamin premix2) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Mineral premix3) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| CM | - | 10.0 | 5.0 | 5.0 | 5.0 |
| PE | - | - | 5.0 | - | - |
| PS | - | - | - | 5.0 | - |
| FV | - | - | - | - | 5.0 |
| Total | 1,000 | 1,000 | 1,000 | 1,000 | 1,000 |
| Calculated nutrient value | |||||
| ME (kcal/kg) | 3,173.63 | 3,173.65 | 3,173.63 | 3,173.63 | 3,173.64 |
| Crude protein (% DM) | 21.01 | 21.01 | 21.01 | 21.01 | 20.01 |
| Calcium (% DM) | 0.89 | 0.89 | 0.89 | 0.89 | 0.89 |
| Total phosphorus (% DM) | 0.70 | 0.70 | 0.70 | 0.70 | 0.70 |
| Available phosphorus (% DM) | 0.47 | 0.47 | 0.47 | 0.47 | 0.47 |
| Lysine (% DM) | 1.38 | 1.38 | 1.37 | 1.37 | 1.37 |
| Methionine+cysteine (% DM) | 0.79 | 0.78 | 0.78 | 0.78 | 0.78 |
| Analyzed nutrient value | |||||
| Crude protein (% DM) | 21.1 | 20.9 | 21 | 21.2 | 21.1 |
| Crude fat (% DM) | 8.9 | 9.2 | 9.3 | 9.4 | 8.7 |
CM, Cordyceps militaris waster medium; PE, Pleurotus eryngii stalk residues; PS, Pleurotus sajor-caju stalk residues; FV, Fammulina velutipes stalk residues; CP, crude protein; ME, metabolizable energy; DM, dry matter.
1) Control, basal diet; CMWM, basal diet supplemented with 1% CM; CMPE, basal diet supplemented with 0.5% CM+0.5% PE; CMPS, basal diet supplemented with 0.5% CM+0.5% PS; CMFV, basal diet supplemented with 0.5% CM+0.5% FV.
Table 3
Table 4
Table 5
| Items | Treatment1) | SEM | p-value | ||||
|---|---|---|---|---|---|---|---|
|
|
|||||||
| Control | CMWM | CMPE | CMPS | CMFV | |||
| 1–21 d | |||||||
| Weight gain (kg) | 0.799a | 0.818a | 0.742b | 0.734b | 0.790a | 0.005 | <0.001 |
| Feed consumption (kg) | 0.981ab | 0.938ab | 0.920b | 0.926b | 1.018a | 0.041 | 0.103 |
| FCR | 1.227 | 1.149 | 1.239 | 1.261 | 1.291 | 0.018 | 0.190 |
| 22–35 d | |||||||
| Weight gain (kg) | 1.115 | 1.125 | 1.124 | 1.117 | 1.112 | 0.009 | 0.975 |
| Feed consumption (kg) | 1.758a | 1.759a | 1.588b | 1.648ab | 1.769a | 0.017 | 0.042 |
| FCR | 1.578a | 1.567a | 1.414b | 1.531ab | 1.572a | 0.020 | 0.095 |
| 1–35 d | |||||||
| Weight gain (kg) | 1.872ab | 1.900a | 1.824b | 1.822b | 1.861ab | 0.009 | 0.022 |
| Feed consumption (kg) | 2.739ab | 2.697ab | 2.508c | 2.574bc | 2.787a | 0.024 | 0.040 |
| FCR | 1.463ab | 1.419ab | 1.376b | 1.413ab | 1.498a | 0.014 | 0.161 |
1) Control, basic feed; CMWM, 1% Cordyceps militaris waster medium; CMPE, 0.5% Cordyceps militaris waster medium+0.5% Pleurotus eryngii stalk residues; CMPS, 0.5% Cordyceps militaris waster medium+0.5% Pleurotus sajor-caju stalk residues; CMFV, 0.5% Cordyceps militaris waster medium+0.5% Fammulina velutipes stalk residues.
Table 6
| Microbial parameter (log CFU/g) | Treatment1) | SEM | p-value | ||||
|---|---|---|---|---|---|---|---|
|
|
|||||||
| Control | CMWM | CMPE | CMPS | CMFV | |||
| Lactobacillus spp. | |||||||
| Ileum | 7.03 | 7.19 | 7.02 | 7.15 | 7.23 | 0.042 | 0.442 |
| Caecum | 8.33 | 8.48 | 8.37 | 8.35 | 8.49 | 0.026 | 0.198 |
| Coliform | |||||||
| Ileum | 7.47 | 6.99 | 7.20 | 7.00 | 7.18 | 0.029 | 0.339 |
| Caecum | 9.64 | 9.46 | 9.60 | 9.49 | 9.47 | 0.024 | 0.126 |
| Clostridium perfringens | |||||||
| Ileum | 6.93 | 6.75 | 7.15 | 7.07 | 7.05 | 0.060 | 0.332 |
| Caecum | 9.19 | 9.05 | 9.30 | 8.88 | 8.87 | 0.027 | 0.361 |
1) Control, basic feed; CMWM, 1% Cordyceps militaris waster medium; CMPE, 0.5% Cordyceps militaris waster medium+0.5% Pleurotus eryngii stalk residues; CMPS, 0.5% Cordyceps militaris waster medium+0.5% Pleurotus sajor-caju stalk residues; CMFV, 0.5% Cordyceps militaris waster medium+0.5% Fammulina velutipes stalk residues.
Table 7
| Items | Treatment1) | SEM | p-value | ||||
|---|---|---|---|---|---|---|---|
|
|
|||||||
| Control | CMWM | CMPE | CMPS | CMFV | |||
| Jejunum | |||||||
| Villus height (μm) | 1,237.1 | 1,240.3 | 1,323.4 | 1,343.9 | 1,292.1 | 19.4 | 0.300 |
| Crypt depth (μm) | 214.5a | 177.1c | 188.9bc | 203.7ab | 193.8bc | 2.67 | <0.01 |
| Tunica muscularis (μm) | 262.8 | 247.4 | 246.0 | 259.8 | 264.1 | 3.74 | 0.372 |
| Villus:crypt | 6.18b | 7.40a | 7.21a | 6.81ab | 6.99ab | 0.12 | 0.03 |
| Ileum | |||||||
| Villus height (μm) | 1,040.9 | 994.5 | 993.6 | 1,018.7 | 997.1 | 9.55 | 0.447 |
| Crypt depth (μm) | 167.2 | 153.2 | 170.8 | 155.9 | 168.3 | 2.97 | 0.228 |
| Tunica muscularis (μm) | 279.7 | 262.3 | 267.5 | 260.7 | 264.6 | 4.78 | 0.737 |
| Villus:crypt | 6.52 | 6.94 | 6.19 | 6.77 | 6.08 | 0.13 | 0.167 |
1) Control, basic feed; CMWM, 1% Cordyceps militaris waster medium; CMPE, 0.5% Cordyceps militaris waster medium+0.5% Pleurotus eryngii stalk residues; CMPS, 0.5% Cordyceps militaris waster medium+0.5% Pleurotus sajor-caju stalk residues; CMFV, 0.5% Cordyceps militaris waster medium+0.5% Fammulina velutipes stalk residues.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





