 |
 |
| Anim Biosci > Volume 34(6); 2021 > Article |
|
Abstract
Objective
Skeletal muscle satellite cells (SMSCs) are significant for the growth, regeneration, and maintenance of skeletal muscle after birth. However, currently, few studies have been performed on the isolation, culture and inducing differentiation of goose muscle satellite cells. Previous studies have shown that C1q and tumor necrosis factor-related protein 3 (CTRP3) participated in the process of muscle growth and development, but its role in the goose skeletal muscle development is not yet clear. This study aimed to isolate, culture, and identify the goose SMSCs in vitro. Additionally, to explore the function of CTRP3 in goose SMSCs.
Methods
Goose SMSCs were isolated using 0.25% trypsin from leg muscle (LM) of 15 to 20 day fertilized goose eggs. Cell differentiation was induced by transferring the cells to differentiation medium with 2% horse serum and 1% penicillin streptomycin. Immunofluorescence staining of Desmin and Pax7 was used to identify goose SMSCs. Quantitative realtime polymerase chain reaction and western blot were applied to explore developmental expression profile of CTRP3 in LM and the regulation of CTRP3 on myosin heavy chains (MyHC), myogenin (MyoG) expression and Notch signaling pathway related genes expression.
Results
The goose SMSCs were successfully isolated and cultured. The expression of Pax7 and Desmin were observed in the isolated cells. The expression of CTRP3 decreased significantly during leg muscle development. Overexpression of CTRP3 could enhance the expression of two myogenic differentiation marker genes, MyHC and MyoG. But knockdown of CTRP3 suppressed their expression. Furthermore, CTRP3 could repress the mRNA level of Notch signaling pathway-related genes, notch receptor 1, notch receptor 2 and hairy/enhancer-of-split related with YRPW motif 1, which previously showed a negative regulation in myoblast differentiation.
Skeletal muscle is an important organ for body movement and energy metabolism, accounting for about 40% of the total animal weight [1]. Meanwhile, itŌĆÖs also a main part of the meat products consumed by the consumers [2]. To promote meat production, we must first fully understand the process of skeletal muscle formation and growth, as well as its regulatory mechanism. Skeletal muscle satellite cells are muscle-derived stem cells, which are located between the basal lamina and sarcolemma of the muscle fibers, with a potential for cell differentiation and proliferation. They are of great significance for the growth, regeneration and maintenance of skeletal muscle after birth [3]. Skeletal muscle satellite cells were first discovered in the frog, subsequently isolated in multiple species such as humans [4], rats [5], bovine [6], sheep [7], chickens [8], ducks [9]. However, currently, few studies have been performed on the isolation, culture and inducing differentiation of goose muscle satellite cells. Besides, the regulatory factors and mechanisms involved during myogenic differentiation of goose muscle satellite cells still need further exploration.
Currently, studies have found that there are many genes which regulate skeletal muscle growth and development, such as muscle cell transcriptional regulator paired box 3/7 (Pax3/7), myogenic regulator factors family genes and myocyte enhancer factor 2 [10ŌĆō12]. In addition, non-coding RNAs, such as miRNAs [13], are also involved in various stages during skeletal muscle development. Although the molecular mechanism of skeletal muscle development has been widely studied, there are still many new genes whose functions need to be investigated.
The CTRP3 gene is a member of the C1q and tumor necrosis factor (TNF)-related protein (CTRPs) family and participates in a series of biological regulation processes. In the liver cells, CTRP3 inhibits gluconeogenesis through the AKT serine/threonine kinase (AKT) signaling pathway, thereby regulating glucose metabolism [14]. In the vascular endothelial cells, exogenous CTRP3 can influence ATP synthesis by activating the mitochondrial ROS (mtROS)/peroxisome proliferator-activated receptor-╬│ coactivator 1╬▒ (PGC-1╬▒) pathway [15]. Moreover, the AKT signaling pathway and the mtROS/PGC-1╬▒ signaling pathway were both discovered to play an important role in muscle cells [16,17]. Previous studies have shown that the expression of CTRP3 was high in mouse embryonic skeletal muscle but was seldomly found in adult skeletal muscle. Meanwhile, CTRP3 was highly expressed during the differentiation of C2C12 cells [18]. However, the potential function of CTRP3 on skeletal muscle development in goose remains unclear. Therefore, the aim of the present study was to isolate and identify goose skeletal muscle satellite cells, then preliminarily investigate the role of CTRP3 on the differentiation of goose skeletal muscle cells.
In this study, we successfully isolated, cultured and identi fied the goose skeletal muscle cell in vitro. Then, we investigated the expression of CTRP3 in leg muscle (LM) of goose from the embryonic to neonatal stages. And we also proved that CTRP3 could regulate the expression of several skeletal muscle cell differentiation related genes in goose.
The fertilized goose eggs used in the experiment were provided by Ningbo Langde Agriculture and Animal Husbandry Co., Ltd. (Ningbo, China). The eggs were put in the same incubator and incubated under the same conditions. The first day after hatching was set as E1. Five eggs were randomly removed at each of seven developmental stages, including embryonic stage of 7 days (E7), embryonic stage of 11 days (E11), embryonic stage of 15 days (E15), embryonic stage of 19 days (E19), embryonic stage of 23 days (E23), and embryonic stage of 27 days (E27) and 3 days post-hatching. Samples of LM were collected, snap frozen into liquid nitrogen and transfer to ŌłÆ80┬░C refrigerator for storage. All animal procedures used in this study were approved by the Ethics Committee for Animal Experiments of Zhejiang A&F University and were performed in accordance with the Guidelines for Animal Experimentation of Zhejiang A&F University (Hangzhou, China).
The 15 to 20 day fertilized goose eggs were disinfected with ethanol to isolate skeletal muscle satellite cells. The LM of the goose embryo was isolated and placed in a petri dish, washed with phosphate buffer solution (PBS) (HyClone, Logan, UT, USA) containing Penicillin-Streptomycin Liquid (Solarbio, Beijing, China) for 3 times and the skin, blood vessels, adipose tissue and connective tissue were removed. Then, the muscle tissues were cut into meat paste, and digested with 0.25% trypsin-ethylenediaminetetraacetic acid (Thermo Fisher, Shanghai, China) at 37┬░C for 20 min. DulbeccoŌĆÖs modified eagle medium (DMEM)/F12 (HyClone, USA) containing 15% fetal bovine serum (FBS, Thermo Fisher, China) was added to terminate the digestion. The suspension was filtered through a 70 ╬╝m mesh sieve and centrifuged at 1,000 r/min for 8 min at room temperature. The supernatant was discarded, cells were resuspended with DMEM/F12 containing 15% FBS and cultured in the 5% CO2 incubator at 37┬░C. One hour later, the fibroblasts had adhered to the bottom of cell culture flask, while the skeletal muscle satellite cells remained in the supernatant. The cell suspension was inhaled into a new cell petri dish and this process was repeated twice to enrich muscle satellite cells and eliminate fibroblasts. To induce cell differentiation, culture medium was switched to differentiation medium (DM) with 2% horse serum and 1% penicillin streptomycin (HyClone, USA).
Total RNA from tissues or cells was obtained using RNAiso reagent and treated with DNase I (Takara, Kyoto, Japan). The concentration and integrity of RNA were measured by spectrophotometer and denatured gel electrophoresis. The PrimeScript first Strand cDNA synthesis kit (Abcam, Shanghai, China) was used to synthesize the cDNAs. The cDNAs were stored at ŌłÆ20┬░C until use.
The cDNA was used as the template for quantitative real-time polymerase chain reaction (qRT-PCR) using the EvaGreen 2X qPCR Master Mix kit (abm, Suzhou, China). Each sample was tested in triplicate, and glyceraldehyde-3-phosphate dehydrogenase was used as a reference gene. Differential gene expression analysis was calculated using the 2ŌłÆ╬ö╬öCT statistical analysis method. The primers were designed using PrimerPremier.5 software (Table 1) and synthesized by Hangzhou Youkang Biotechnology Co., Ltd. (Zhejiang, China).
Immunofluorescence was used to identify the isolated skeletal muscle satellite cells. Briefly, cells grown in 12-well plate were firstly washed with PBS for three times. and fixed with 4% paraformaldehyde for 15 min. After being washed three times with PBS, the cells were permeabilized with 0.25% Triton X-100 per well for 10 min and blocked at 4┬░C overnight. Afterwards, cells were incubated with 1:100 diluted primary anti-Desmin (Abcam, China) or anti-Pax7 (Abcam, China) for 1 hour at room temperature. After washing, 1:2,000 diluted fluorescent secondary antibody (Thermo Fisher, China) was used to incubate the cells for 1 hour at room temperature. The cells were then washed, added with 4ŌĆ▓,6-diamidino-2- phenylindole (Invitrogen, Carlsbad, CA, USA) and incubated for 15 min at room temperature to stain the cell nuclei. In the end, samples were captured using a fluorescence microscope (OLYMPUS, Tokyo, Japan).
The RNA oligonucleotides, including the CTRP3 siRNAs and the negative control (NC) were all purchased from RiboBio Co., Ltd. (Guangzhou, China). Lipofectamine 3000 (Invitrogen, USA) was used for transfection following the manufacturer s instructions. All oligonucleotides sequences are listed in Table 2.
PcDNA-3.1 CTRP3 was the expression vector. The primers were designed to amplify the entire coding sequence region of CTRP3 and were digested with BamHI and EcoRI restriction sites. The primersŌĆÖ sequences were as follows: 5ŌĆ▓-CGCG GATCCATGGCAGAGAAGGATTTCATC-3ŌĆ▓and 5ŌĆ▓-CCG GAATTCTTACTTGGTTTCAAAGAGA-3ŌĆ▓. The PCR product was then cloned into the pcDNA-3.1 vector.
Total proteins of the cells were prepared and treated as previously described [19]. Cells were homogenized in radio immunoprecipitation assay buffer with 1% phenylmethanesulfonyl fluoride and incubated on ice for 30 min to extract total proteins. Proteins were separated on 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gels and then transferred onto the polyvinylidene fluoride membranes (Millipore, Bllerica, MA, USA). After blocking with 5% bovine serum albumin, the membranes were incubated with primary antibodies for MyHC (sc-32732; Santa Cruz Biotechnology, Santa Cruz, CA, USA), CTRP3 (ab36870; Abcam, China), ╬▓-actin (ab40854; Abcam, USA). Thereafter, the membranes were incubated with secondary antibody for horseradish peroxidase-labeled anti-rabbit/mouse immunoglobulin G (A0208/A0216; Beyotime, Shanghai, China). Finally, the blots were detected through the chemiluminesence detection system (Amersham, Piscataway, NJ, USA) using chemiluminescence (ECL) reagent (Thermo Scientific, Waltham, MA, USA). Image J software was used for gray value analysis.
All results are shown as the mean┬▒standard error of the mean. Each treatment was repeated for three times. Unpaired StudentŌĆÖs t-test was performed to test statistical significance using SPSS 20.0 software. Two-tailed t tests were used in the analysis. p<0.05 was considered as significant.
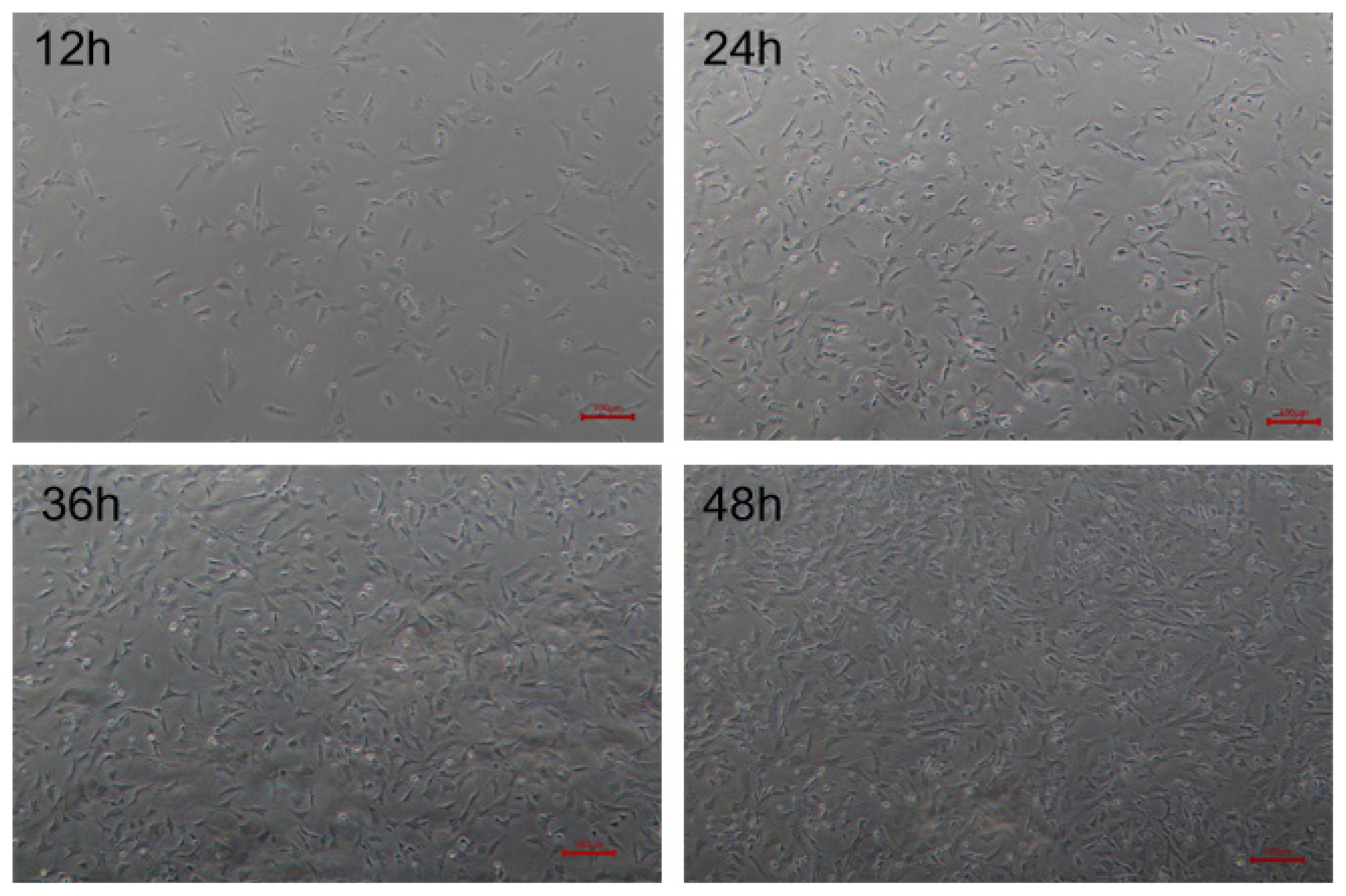
The isolated goose skeletal muscle satellite cells were cultured in growth medium and began to adhere to plates 12 h later. After 24 h, the cells began to grow dispersedly, and some of them had completely adhered. After 36 h, all the cells were completely adhered and gradually extended into elongated shape. The cell confluence approximately reached 90% after 48 h (Figure 1).
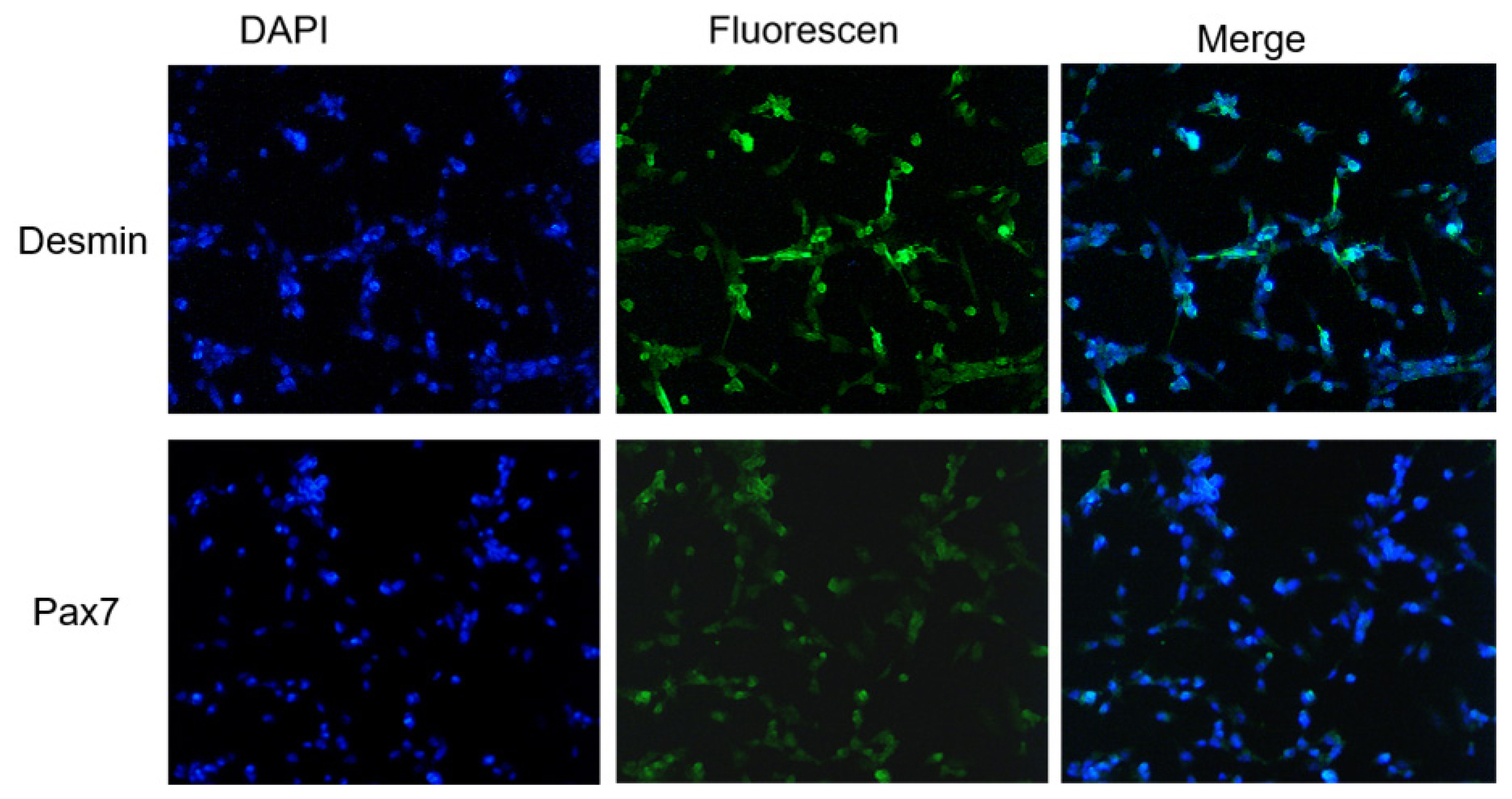
Desmin and Pax7 are special markers of skeletal muscle satellite cells [20], and are often used to distinguish skeletal muscle satellite cell from other cells. After immunofluorescence staining, expression of Pax7 and Desmin were observed in the isolated cells. Pax7 was distributed in the nucleus and Desmin was in the cytoplasm (Figure 2). This result further confirmed the isolated cells were goose skeletal muscle satellite cells.
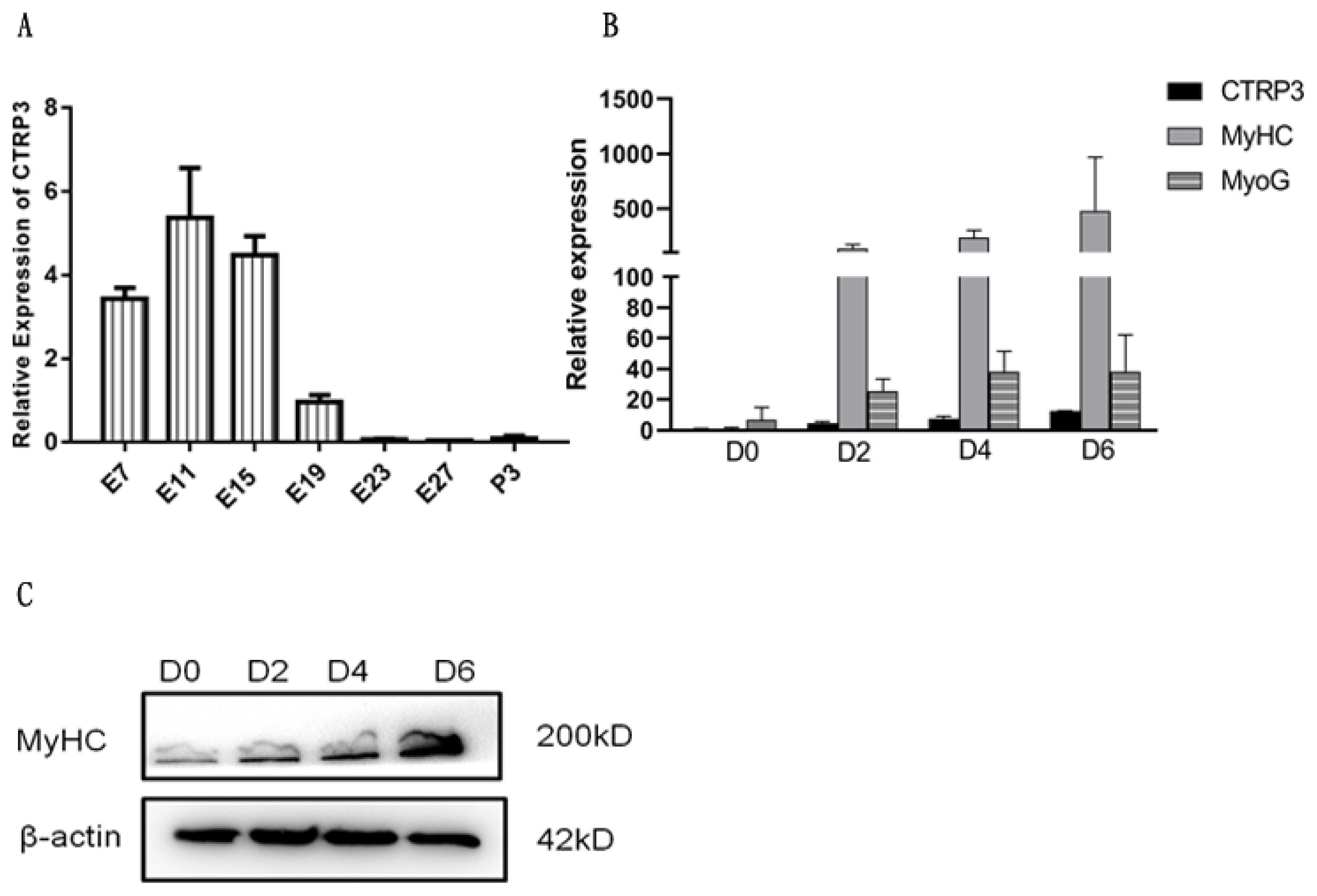
To investigate the expression profile of CTRP3 during LM development, qPCR assay was performed. The mRNA expression level at E19 was regarded as a control and assigned a value of 1. The results show that the expression of CTRP3 reached its highest level in E11 (Figure 3A). The expression level rose from E7 to E11, then decreased sharply from E15 to P3 (Figure 3A), which indicated that CTRP3 might have a potential role in goose skeletal myogenesis.
Next, we set up a cell model to detect the function of CTRP3 during myoblast differentiation. Both CTRP3 and two marker genes of myogenic differentiation, MyHC and MyoG, were up-regulated during this period (Figure 3BŌĆō3C). Thus, we could confirm that the in vitro cell model was successfully established.
To assess the role of CTRP3 gene in goose skeletal muscle satellite cell differentiation, we detected muscle-specific gene expression after transfection with siRNA-CTRP3. CTRP3 was significantly down-regulated in muscle cells at 3 day of DM (Figure 4AŌĆō4B). The mRNA level of MyHC and MyoG were decreased remarkably (Figure 4CŌĆō4D), and the protein level of MyHC was decreased as well (Figure 4B). Thus, these results indicated that CTRP3 knockdown might attenuate goose myogenesis.
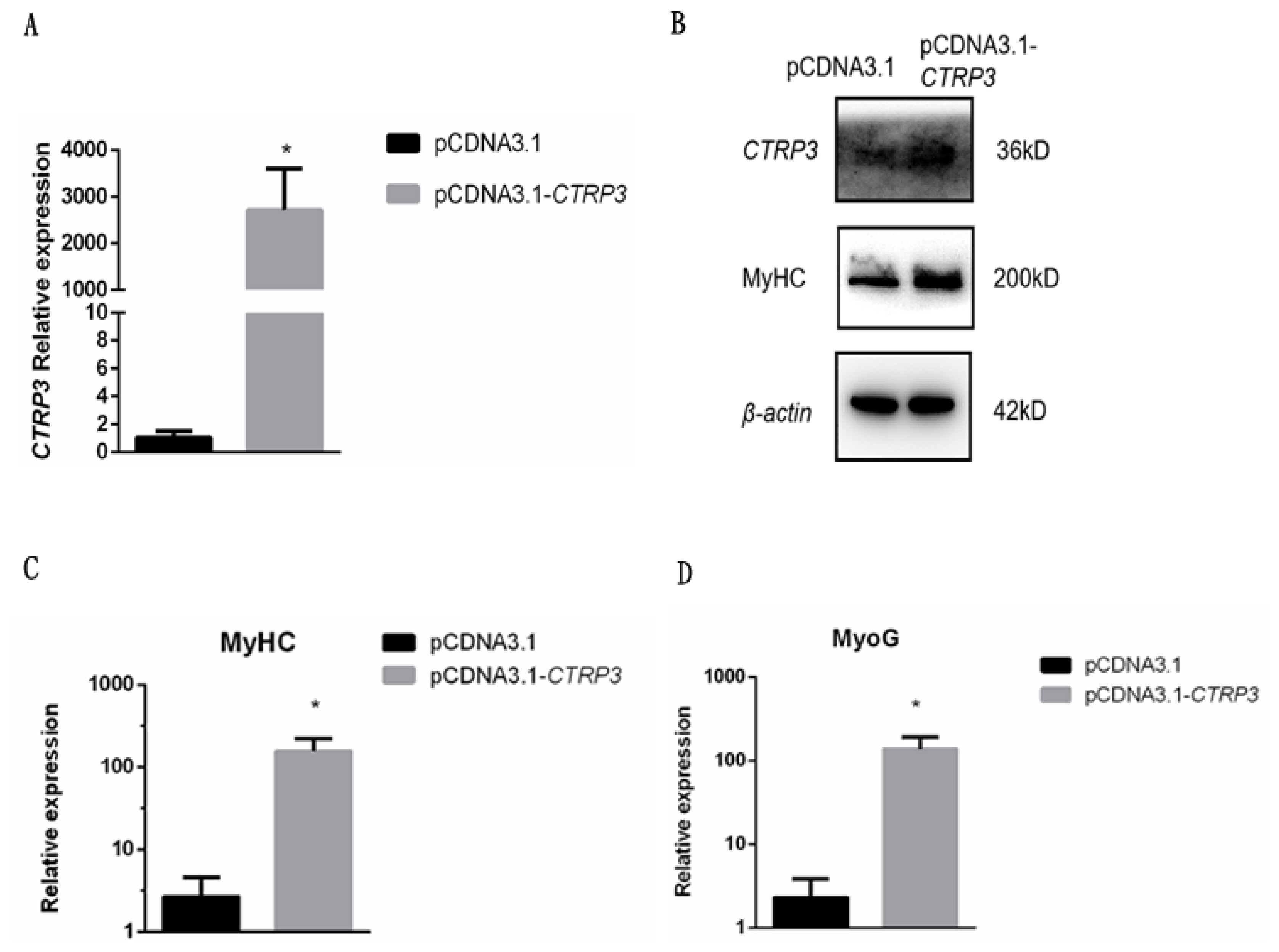
To further investigate the role of CTRP3 in goose myoblast differentiation, the cells were transfected with the expression vector pcDNA3.1-CTRP3. As a result, the expression of CTRP3 was much higher than the control at 3 days post-transfection (Figure 5AŌĆō5B). The mRNA level of MyHC and MyoG were increased significantly (Figure 5C-5D). Meanwhile, the protein level of MyHC was also increased (Figure 5B). These results suggested that overexpression of CTRP3 might induce goose skeletal muscle satellite cell differentiation.
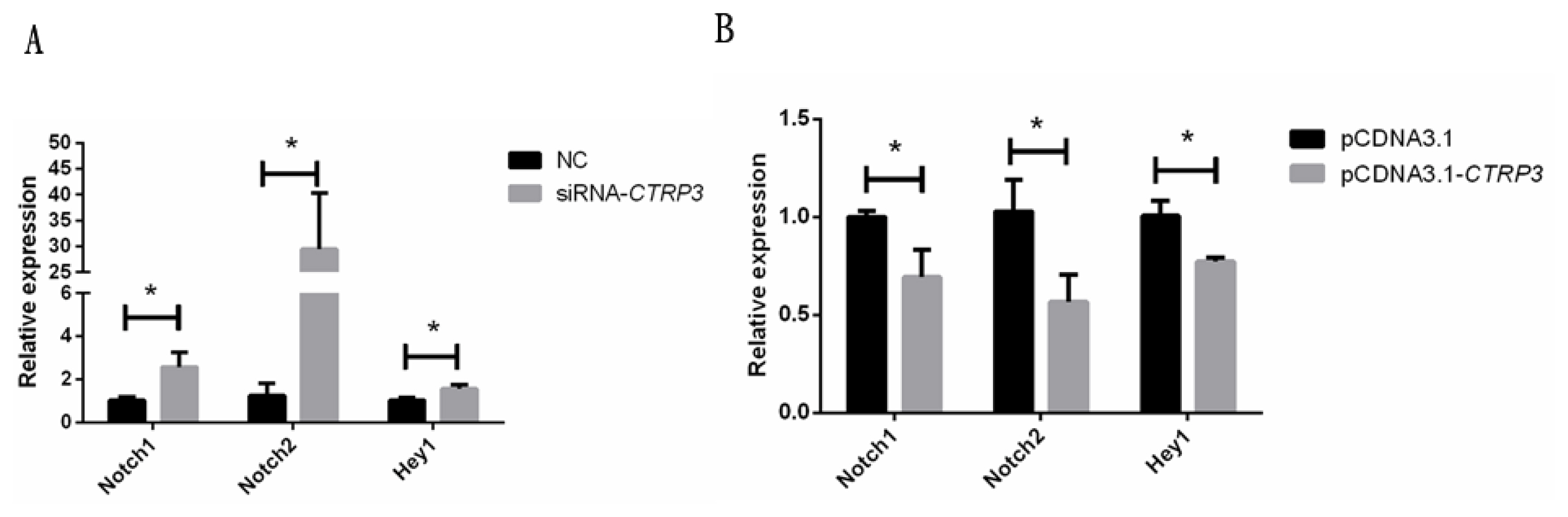
Notch signaling pathway plays an important role in muscle cell differentiation [21]. Previous studies have found that CTRP3 gene could suppress the expression of Notch signaling pathway during renal cell fibrosis [22]. As shown in Figure 6A, the mRNA level of Notch signaling pathway-related genes, notch receptor 1 (Notch1), notch receptor 2 (Notch2), and hairy/enhancer-of-split related with YRPW motif 1 (Hey1), were significantly induced after transfecting with siRNA-CTRP3 in goose skeletal muscle satellite cell. Nevertheless, over-expression of CTRP3 in DM reduced their expression (Figure 6B). Therefore, we illustrated that CTRP3 might also regulate the Notch signaling pathway in goose skeletal muscle cells.
The content of skeletal muscle satellite cells in the body of animals is related to their age. Studies have shown that the number of skeletal muscle satellite cells continue to decrease as age increase. In adult animals, muscle satellite cells contain only 1% to 5% [23]. Therefore, it is especially important to select the appropriate embryo age to isolate and culture the goose muscle satellite cells. Previous study has found that it was better to isolate skeletal muscle satellite cells from 10 to 18 day embryonic chicks [24]. Therefore, the goose embryos between 15 to 20 days were selected for the isolation of skeletal muscle satellite cells in our study. The results showed that the cells were in a good growth state and had a good differentiation ability, which could meet the later experimental needs.
Pax7 is a specific marker protein of muscle satellite cell and a marker gene in the process of postnatal muscle development. Desmin is a component of the muscle cytoskeleton that prompts the function of skeletal muscle, and also one of the early muscle derived marker proteins [25]. Previous studies showed that only a small proportion of poultry myoblasts expressed desmin in culture. However, desmin was extensively expressed in poultry myotubes. Meanwhile, Pax7 was readily detected in the nuclei of myoblasts but not in myotubes of poultry primary skeletal muscle cultures [26]. In our study, we found that both Desmin and Pax7 were highly expressed in the isolated cells. Therefore, it could be inferred that there might be both myoblasts and differentiated myotubes in our cultured cells, and it fully confirmed that the isolated cells were goose skeletal muscle satellite cells.
According to our previous study, we chose seven stages included embryonic and postnatal period to detect the expression of CTRP3 [27]. E19 was considered as the fastest growth stage of embryonic muscle of Peking duck [28]. In the present study, we discovered the expression of CTRP3 decreased sharply from E15 to E19, which indicated that it might have a function in goose skeletal myogenesis. The high expression of MyHC and MyoG genes were generally regarded as a sign of successful differentiation of skeletal muscle cells [19]. Our results showed that the both genes were up-regulated in the differentiated goose muscle satellite cells, suggesting that our in vitro cell differentiation model was successfully constructed. We found that CTRP3 up-regulated during C2C12 cell differentiation, suggesting that it might play a role in myogenic differentiation.
In our model, the e xpression of MyHC and MyoG were down-regulated with the knockdown of CTRP3. On the other hand, over-expression of CTRP3 promotes the expression of MyHC and MyoG. Therefore, CTRP3 might play a positive role in myogenic differentiation. To further investigate the potential mechanism of CTRP3 in goose muscle satellite cell differentiation, we demonstrated that CTRP3 could inhibit the expression of Notch signaling pathway related genes. Activated Notch1 expression in C2C12 myoblasts inhibit muscle cell fusion [29]. Overexpression of Notch2 showed a negative regulation in myoblast differentiation [30]. Besides, Hey1 was found to inhibit myogenesis by repressing expression of key myogenic targets [31]. Thus, we speculated that CTRP3 might regulate the goose skeletal muscle satellite cell differentiation through the Notch signaling pathway. However, further experimental research will be needed to fully explain the relationship between them.
In conclusion, we successfully isolated and cultured goose skeletal muscle satellite cells and established a cell differentiation model in vitro. Moreover, our study preliminarily showed that CTRP3 could promote the expression of MyHC and MyoG, which are two marker genes of goose myogenic differentiation. We also speculated that CTRP3 might regulate the differentiation of goose skeletal muscle satellite cells through the Notch signaling pathway.
ACKNOWLEDGMENTS
This research was supported by the General projects of Zhejiang Natural Science Foundation (LY18C170001), Student Research and Training Project of Zhejiang A&F University (KX20180234) and the National Natural Science Foundation of China (31702153).
Figure┬Ā1
Morphological observation of isolated goose skeletal muscle satellite cells at different time. Cells were observed at 12, 24, 36, and 48 h after culture under microscope with white light (├Ś200).
Figure┬Ā2
Identification of goose skeletal muscle satellite cells. Desmin and Pax7 were detected with Immunofluorescence; 4ŌĆ▓,6-diamidino-2-phenylindole (DAPI) was used to stain the nuclei of goose skeletal muscle cells (├Ś200).
Figure┬Ā3
Expression profile of the goose CTRP3 in LM and the marker genes during myogenic differentiation. (A) The CTRP3 mRNA expression level was measured by qPCR during leg muscle development of goose. (B) The mRNA level of MyHC, MyoG, and CTRP3 in 0, 2, 4, and 6 days of cell differentiation. The fold change was relative to day 0 of DM expression. GAPDH was used as the reference gene for Q-PCR. (C) The protein level of MyHC in 0, 2, 4, and 6 days of cell differentiation. ╬▓-actin as controls for western blot. Results are expressed as mean┬▒standard error of the mean (n = 3). CTRP3, C1q and tumor necrosis factor-related protein 3; LM, leg muscle; qPCR, quantitative polymerase chain reaction; MyHC, myosin heavy chains; MyoG, myogenin; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; DM, differentiation medium; qPCR, quantitative polymerase chain reaction.
Figure┬Ā4
Knockdown of CTRP3 inhibited the expression of MyHC and MyoG during differentiation. (A) The mRNA level of CTRP3 was detected by qPCR in myoblast transfected with siRNA-CTRP3 or NC at day 3 of DM. (B) The protein levels of CTRP3 and MyHC were detected by western blot in myoblast transfected with siRNA-CTRP3 or NC at day 3 of DM. (C) The mRNA level of MyHC was detected by qPCR in myoblast transfected with siRNA-CTRP3 or NC at day 3 of DM. (D) The mRNA level of MyoG was detected by qPCR in myoblast transfected with siRNA-CTRP3 or NC at day 3 of DM. Results are expressed as mean┬▒standard error of the mean (n = 3). * p<0.05. CTRP3, C1q and tumor necrosis factor-related protein 3; MyHC, myosin heavy chains; MyoG, myogenin; qPCR, quantitative polymerase chain reaction; NC, negative control; DM, differentiation medium.

Figure┬Ā5
Overexpression of CTRP3 promoted the expression of MyHC and MyoG during differentiation. (A) The mRNA level of CTRP3 was detected by qPCR in myoblast transfected with pCDNA3.1-CTRP3 or pCDNA3.1 at day 3 of DM. (B) The protein levels of CTRP3 and MyHC were detected by western blotting in myoblast transfected with pCDNA3.1-CTRP3 or pCDNA3.1 at day 3 of DM. (C) The mRNA level of MyHC was detected by qPCR in myoblast transfected with pCDNA3.1-CTRP3 or pCDNA3.1 at day 3 of DM. (D) The mRNA level of MyoG was detected by qPCR in myoblast transfected with pCDNA3.1-CTRP3 or pCDNA3.1 at day 3 of DM. Results are expressed as mean┬▒standard error of the mean (n = 3). * p<0.05. CTRP3, C1q and tumor necrosis factor-related protein 3; MyHC, myosin heavy chains; MyoG, myogenin; qPCR, quantitative polymerase chain reaction; NC, negative control; DM, differentiation medium.
Figure┬Ā6
Effect of CTRP3 gene on the expression of Notch signaling pathway related genes. (A) The mRNA level of Notch1, Notch2, and Hey1 was detected by qPCR in myoblasts transfected with siRNA-CTRP3 or NC at day 3 of DM. (B) The mRNA levels of Notch1, Notch2, and Hey1 were detected by qPCR in myoblast transfected with pCDNA3.1-CTRP3 or pCDNA3.1 at day 3 of DM. Results are expressed as mean┬▒standard error of the mean (n = 3). * p<0.05. CTRP3, C1q and tumor necrosis factor-related protein 3; Notch1, notch receptor 1; Notch2, notch receptor 2; Hey1, hairy/enhancer-of-split related with YRPW motif 1; qPCR, quantitative polymerase chain reaction; NC, negative control; DM, differentiation medium.
Table┬Ā1
Primers for real-time polymerase chain reaction
REFERENCES
1. G├╝ller I, Russell AP. MicroRNAs in skeletal muscle: their role and regulation in development, disease and function. J Phsiol 2010; 588:4075ŌĆō87.
https://doi.org/10.1113/jphysiol.2010.194175

2. Luo W, Nie Q, Zhang X. MicroRNAs involved in skeletal muscle differentiation. J Genet Genom 2013; 40:107ŌĆō16.
https://doi.org/10.1016/j.jgg.2013.02.002

3. Rhoads RP, Fernyhough ME, Liu X, et al. Extrinsic regulation of domestic animal-derived myogenic satellite cells II. Domest Anim Endocrinol 2009; 36:111ŌĆō26.
https://doi.org/10.1016/j.domaniend.2008.12.005


4. Blau HM, Webster C. Isolation and characterization of human muscle cells. Proc Natl Acad Sci USA 1981; 78:5623ŌĆō7.
https://doi.org/10.1073/pnas.78.9.5623



5. Rosenblatt JD, Lunt AI, Parry DJ, Partridge TA. Culturing satellite cells from living single muscle fiber explants. In Vitro Cell Dev Biol Anim 1995; 31:773ŌĆō9.
https://doi.org/10.1007/BF02634119


6. Dodson MV, Martin EL, Brannon MA, Mathison BA, McFarland DC. Optimization of bovine satellite cell-derived myotube formation in vitro
. Tissue Cell 1987; 19:159ŌĆō66.
https://doi.org/10.1016/0040-8166(87)90001-2


7. Wu H, Ren Y, Li S, et al.
In vitro culture and induced differentiation of sheep skeletal muscle satellite cells. Cell Biol Int 2012; 36:579ŌĆō87.
https://doi.org/10.1042/CBI20110487


8. Bennett VD, Cowles E, Husic HD, Suelter CH. Muscle cell cultures from chicken breast muscle have increased specific activities of creatine kinase when incubated at 41┬░C compared with 37┬░C. Exp Cell Res 1986; 164:63ŌĆō70.
https://doi.org/10.1016/0014-4827(86)90454-4


9. Liu H, Li L, Chen X, et al. Characterization of in vitro cultured myoblasts isolated from duck (Anas platyrhynchos) embryo. Cytotechnology 2011; 63:399ŌĆō406.
https://doi.org/10.1007/s10616-011-9356-7



10. Megeney LA, Kablar B, Garrett K, Anderson JE, Rudnicki MA. MyoD is required for myogenic stem cell function in adult skeletal muscle. Genes Dev 1996; 10:1173ŌĆō83.
https://doi.org/10.1101/gad.10.10.1173


11. Le Grand F, Rudnicki MA. Skeletal muscle satellite cells and adult myogenesis. Curr Opin Cell Biol 2007; 19:628ŌĆō33.
https://doi.org/10.1016/j.ceb.2007.09.012



12. Cserjesi P, Olson EN. Myogenin induces the myocyte-specific enhancer binding factor MEF-2 independently of other muscle-specific gene products. Mol Cell Biol 1991; 11:4854ŌĆō62.
https://doi.org/10.1128/MCB.11.10.4854



13. Chen JF, Mandel EM, Thomson JM, et al. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat Genet 2006; 38:228ŌĆō33.
https://doi.org/10.1038/ng1725


14. Peterson JM, Seldin MM, Wei Z, Aja S, Wong GW. CTRP3 attenuates diet-induced hepatic steatosis by regulating triglyceride metabolism. Am J Physiol Gastrointest Liver Physiol 2013; 305:G214ŌĆō24.
https://doi.org/10.1152/ajpgi.00102.2013



15. Feng H, Wang JY, Zheng M, et al. CTRP3 promotes energy production by inducing mitochondrial ROS and up-expression of PGC-1╬▒ in vascular smooth muscle cells. Exp Cell Res 2016; 341:177ŌĆō86.
https://doi.org/10.1016/j.yexcr.2016.02.001


16. Chakravarthy MV, Abraha TW, Schwartz RJ, Fiorotto ML, Booth FW. Insulin-like growth factor-I extends in vitroreplicative life span of skeletal muscle satellite cells by enhancing g1/s cell cycle progression via the activation of phosphatidylinositol 3ŌĆ▓-kinase/akt signaling pathway. J Biol Chem 2000; 275:35942ŌĆō52.
https://doi.org/10.1074/jbc.M005832200


17. Yuzefovych L, Wilson G, Rachek L. Different effects of oleate vs. palmitate on mitochondrial function, apoptosis, and insulin signaling in L6 skeletal muscle cells: role of oxidative stress. Am J Physiol Endocrinol Metab 2010; 299:E1096ŌĆō105.
https://doi.org/10.1152/ajpendo.00238.2010



18. Otani M, Furukawa S, Wakisaka S, Maeda T. A novel adipokine C1q/TNF-related protein 3 is expressed in developing skeletal muscle and controls myoblast proliferation and differentiation. Mol Cell Biochem 2015; 409:271ŌĆō82.
https://doi.org/10.1007/s11010-015-2531-y


19. Wang H, Zhang Q, Wang BB, et al. miR-22 regulates C2C12 myoblast proliferation and differentiation by targeting TGFBR1. Eur J Cell Biol 2018; 97:257ŌĆō68.
https://doi.org/10.1016/j.ejcb.2018.03.006


20. Li F, Hou L, Ma Y, Pang Q, Guan W. Isolation, culture, identification and muscle differentiation of skeletal muscle satellite cells in Beijing fatty chicken. Sci Agric Sin 2010; 43:4725ŌĆō31.
https://doi.org/10.3864/j.issn.0578-1752.2010.22.021

21. Mourikis P, Sambasivan R, Castel D, Rocheteau P, Bizzarro V, Tajbakhsh S. A critical requirement for notch signaling in maintenance of the quiescent skeletal muscle stem cell state. Stem Cells 2012; 30:243ŌĆō52.
https://doi.org/10.1002/stem.775


22. Chen X, Wu Y, Diao Z, et al. C1q/tumor necrosis factor-related protein-3 improves renal fibrosis via inhibiting notch signaling pathways. J Cell Physiol 2019; 234:22352ŌĆō64.
https://doi.org/10.1002/jcp.28801


23. Renault V, Rolland E, Thornell LE, Mouly V, Butler-Browne G. Distribution of satellite cells in the human vastus lateralis muscle during aging. Exp Gerontol 2002; 37:1513ŌĆō4.
https://doi.org/10.1016/s0531-5565(02)00095-5


24. Hartley RS, Bandman E, Yablonka-Reuveni Z. Skeletal muscle satellite cells appear during late chicken embryogenesis. Dev Biol 1992; 153:206ŌĆō16.
https://doi.org/10.1016/0012-1606(92)90106-Q



25. Paulin D, Li Z. Desmin: a major intermediate filament protein essential for the structural integrity and function of muscle. Exp Cell Res 2004; 301:1ŌĆō7.
https://doi.org/10.1016/j.yexcr.2004.08.004


26. Baquero-Perez B, Kuchipudi SV, Nelli RK, Chang KC. A simplified but robust method for the isolation of avian and mammalian muscle satellite cells. BMC Cell Biol 2012; 13:16
https://doi.org/10.1186/1471-2121-13-16



27. He K, Ren T, Zhu S, Liang S, Zhao A. Transiently expressed pattern during myogenesis and candidate miRNAs of Tmem8C in goose. J Genet 2017; 96:39ŌĆō46.
https://doi.org/10.1007/s12041-016-0737-8


28. Gu L, Xu T, Huang W, Xie M, Sun S, Hou S. Identification and profiling of microRNAs in the embryonic breast muscle of pekin duck. PLoS One 2014; 9:e86150
https://doi.org/10.1371/journal.pone.0086150



29. Shawber C, Nofziger D, Hsieh JJ, et al. Notch signaling inhibits muscle cell differentiation through a CBF1-independent pathway. Development 1996; 122:3765ŌĆō73.


30. Ono Y, Sensui H, Okutsu S, Nagatomi R. Notch2 negatively regulates myofibroblastic differentiation of myoblasts. J Cell Physiol 2007; 210:358ŌĆō69.
https://doi.org/10.1002/jcp.20838


31. Buas MF, Kabak S, Kadesch T. The Notch effector Hey1 associates with myogenic target genes to repress myogenesis. J Biol Chem 2010; 285:1249ŌĆō58.
https://doi.org/10.1074/jbc.M109.046441


- TOOLS
-
METRICS

- Related articles
-
Isolation, Culture and Identification of Porcine Skeletal Muscle Satellite Cells2015 August;28(8)






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print




