 |
 |
| Anim Biosci > Volume 29(10); 2016 > Article |
|
Abstract
Isolation and culture of spermatogonial stem cells (SSCs) are attractive for production of genetic modified offspring. In the present study, buffalo spermatogonial stem-like cells were isolated, cultured and expression pattern of different germ cell marker genes were determined. To recover spermatogonia, testes from age 3 to 7 months of buffalo were decapsulated, and seminiferous tubules were enzymatically dissociated. Two types of cells, immature sertoli cell and type A spermatogonia were observed in buffalo testes in this stage. Germ cell marker genes, OCT3/4 (Pou5f1), THY-1, c-kit, PGP9.5 (UCHL-1) and Dolichos biflorus agglutinin, were determined to be expressed both in mRNA and protein level by reverse transcription polymerase chain reaction and immunostaining in buffalo testes and buffalo spermatogonial stem-like cells, respectively. In the following, when the isolated buffalo buffalo spermatogonial stem-like cells were cultured in the medium supplemented 2.5% fetal bovine serum and 40 ng/mL glial cell-derived neurotrophic factor medium, SSCs proliferation efficiency and colony number were significantly improved than those of other groups (p<0.05). These findings may help in isolation and establishing long term in vitro culture system for buffalo spermatogonial stem-like cells, and accelerating the generation of genetic modified buffaloes.
Water buffalo is an economically important livestock, which partially distributes in south China, providing high quality of milk, meat and work power (Shi et al., 2012). Buffalo is low fertility species, characterized by low conception rate, seasonality estrus and delayed puberty, and is in urgent need of improvement through propagation of superior germ plasma by modern reproductive technologies (Shi et al., 2007). At present, several technologies from cattle have been adapted for improvement of buffalo production, but there are various limitations. Therefore, developing of stem cell technology in buffalo has significance in the genetic improvement of this species (Shi et al., 2007; Mahla et al., 2012). Spermatogonial stem cells (SSCs) have received a great deal of attention in recent years, as they have unique capacity for self-renewal and produce large number of differentiating haploid germ cells which could transmit paternal genetic information to next generations (Kanatsu-Shinohara and Shinohara, 2013). SSCs can also be genetically modified and further differentiated to spermatozoa in vitro, subsequently contribute to produce genetic modified offspring through the method of intracytoplasmic sperm injection (ICSI) or SSCs transplantation into a recipient testis (Kanatsu-Shinohara et al., 2011; Sehgal et al., 2014). Recently, successful germ cell transplantation into bull testis had resulted in production of donor-derived sperm cells (Stockwell et al., 2009). In case of establishment livestock species male germ cells line, researchers can facilely create targeted gene editing animals using TALEN or CRISPR/Cas9 technology (Gaj et al., 2013; Chapman et al., 2015). Nevertheless, no SSC lines have yet been established for livestock species. The most likely reason is lack of sufficient understanding of expression of related markers in the male stem cell, especially water buffalo.
To efficiently isolate and purify SSCs, the availability of SSCs specific expressing markers was of great importance. Oct-4 (also as knows Pou5fl), THY-1, c-kit, PGP9.5 (also as knows UCHL-1) and Dolichos biflorus agglutinin (DBA) are used to identify bovine (Izadyar et al., 2002; Oatley et al., 2004; Reding et al., 2010), goat (Heidari et al., 2012; Abbasi et al., 2013), sheep (Rodriguez-Sosa et al., 2006), pig (Lee et al., 2013; Lee et al., 2014), dog (Harkey et al., 2013), mouse and human (von Kopylow et al., 2010) and chicken SSCs (Momeni-Moghaddam et al., 2014; Sisakhtnezhad et al., 2015). Recently, Oct-4 and c-kit were reported to be experessed in buffalo testis and spermatogonail stem cells (Xie et al., 2010; Yu et al., 2014). In the further, it is essential for determination the morphological, physiological and stem cell potential through various markers and different biological methods. So the aim of this study was to determine expression pattern of different markers and transcripts in prepubertal buffalo testis and SSCs, and optimize the in vitro culture medium of buffalo SSCs.
Testes from 3 to 7 month old prepubertal buffalo (Bubalus bubalis) calves were collected from Nanning slaughterhouse. A small piece of testis tissue was submerged immediately after collection in liquid nitrogen frozen for isolation of RNA. For histochemical analysis, testes tissue was immediately fixed in 4% formalin fixative following collection. For SSC isolation, the testes were transported to the laboratory in phosphate buffer saline (PBS) containing penicillin (100 U/mL), and streptomycin (100 μg/mL) (Sigma, St Louis, MO, USA, P4333).
Cell isolation was performed as described previously (Xie et al., 2010). Briefly, testes from 3 to 7 age of months buffalo were immediately washed several times by PBS containing penicillin (100 U/mL), and streptomycin (100 μg/mL). Then the tunica albuginea of testes was manually decapsulated, and parenchyma was used for cell isolation. A small piece of testis parenchyma was cut and separated into seminiferous tubes by the pinhead of 1 mL syringe. Then seminiferous tubes were incubated for 15 min at 37°C in PBS containing 1% collagenase (Type V; Sigma, USA), and digested into fragments by beating upon during incubation. Most of the Leydig cells were discarded after washing several times with Dulbecco modified eagle medium (DMEM; Invitrogen Corporation, Carlsbad, CA, USA). Seminiferous cord fragments were centrifuged three times (each time 1,000 rpm for 3 min) with DMEM containing 10% fetal bovine serum (Hyclone, Logan, UT, USA). Following incubation in DMEM containing 2.5% trypsinase (Sigma, USA) for 6 to 8 min, when the cells were separated into a single pellet from the remaining tube fragments, digestion was ceased with DMEM containing 10% fetal bovine serum (FBS; Hyclone, USA). To remove debris, cell suspension was filtered using a 200 μm nylon cell strainer (Falcon, Becton Dickinson Labware, NY, 10010/USA). Next, the collected cell suspension was removed by centrifugation at 1,200 rpm for 5 min, and cells were cultured in DMEM containing 10% FBS for 4 to 6 hours. Non-adherent cells were poured over a Percoll density gradient (11%, 19%, 21%, 35%) and centrifuged at 1,800 rpm for 40 min. Then the cells were cultured in DMEM with different concentrations of FSB (0%, 1.5%, 2.5%, 3.5%) and different concentrations of glial cell-derived neurotrophic factor (GDNF) (0 ng/mL, 20 ng/mL, 40 ng/mL, 80 ng/mL) overnight at 37°C in a high humidity 5% (v/v) CO2 in air atmosphere. Cell proliferation was assessed by Brdu incorporation assay.
Fixed testicular tissues from 3 to 7 ages of month buffalo testes were dehydrated, and then embedded in paraffin and sectioned 5 μm thick using standard procedures. Sections were processed through xylene and ethanol into water, stained with hematoxylin and eosin, or used for immunohistochemistry. These sections were washed by PBS-Tween-20 for 3 min, and exposed 3% H2O2-methanol for 30 min. All sections were placed into sodium citrate-hydrochloric acid buffer solution (pH 6.0), and were heated three times (6 min each time). After heated, sections were washed by PBS-tween-20 for 3 to 5 min, blocked with 5% FBS in PBS for 1 hour at room temperature, and incubated with primary antibody for overnight at 4°C, including anti-Oct4 (Abcam, ab18976, 1:100), anti-THY-1 (Abcam, ab3105, 1:100), anti-PGP9.5 (Abcam, ab10404, 1:50) and anti-c-kit (Abcam, ab5506, 1:100) respectively. After washing three times with PBS-tween-20 for 5 min each, these sections were incubated with 3% H2O2 for 10 min, washed with PBS-tween-20 for 3 min. These sections were exposed for 45 min at room temperature to secondary antibody (HRP-conjugated goat anti-rabbit IgG, Abcam, ab672, 1:500), washed with PBS-tween-20 as above. The sections were also stained with DBA substrate kit (Vector Laboratories, Burlington, ON, Canada) according to the manufacture’s instructions.
Primary culture of buffalo spermatogonial stem-like cells was treated under different concentrations of FBS and GDNF in cultures plates for 12 days. After washing, cell colonies were fixed with 4% paraformaldehyde in PBS at 4°C for 1 hour. Following, cell colonies were washed three times with PBS for 5 min each, and permeabilized with 1% Triton-100 for 30 min. The colonies were again washed three times with PBS for 5 min each, and blocked 1% bovine serum albumin (BSA) in PBS for 1 h. Then, the colonies were incubated with anti-Oct4 (Abcam, ab18976, 1:200), anti-THY-1 (Abcam, ab3105, 1:200), anti-PGP9.5 (Abcam, ab10404, 1:100), and anti-c-kit (Abcam, ab5506, 1:200) respectively overnight at 4°C. Then the incubated colonies were washed three times with PBS for 5 min each, following incubation with secondary antibody: FITC-conjugated goat anti-rabbit IgG for 90 min at 37°C. For the nuclear staining, colonies were counterstained with Hochest33342 (Life Technology, 62249, NY, NY, USA) for 5 min, washed three times with PBS for 5 min each, and analyzed under fluorescence microscope (N-STORM, Nikon).
Expression of alkaline phosphatase (AP) activity in cell colonies was detected by AP staining. The purified and cultured Buffalo spermatogonia were washed three times with PBS before they were fixed with 4% (w/v) paraformaldehyde for 30 min. Following these colonies were washed three times with PBS for 5 min each, the nitroblue tetrazolium chloride/5-bromo-4-chloro-3-indolylphosphate toluidine (NBT/BCIP; Amresco, Solon, OH, USA) were used as substrates for 15 to 30 min at 37°C in dark.
The 5-bromode-oxyuridine (Brdu; Sigma, USA) and DBA were used to determine buffalo spermatogonia proliferation. For Brdu incorporation assay, the number of cells in each 24-well plate was counted under inverted microscope (CKX41; Olympus, Tokyo, Japan), and were seeded at a density of 5.0×104 cells/mL, with three wells a group. On 0, 1, 2, 3, 4, 5, and 6 days after culture, Brdu (0.3 μg/μL) was added to each well of the parallel samples, and cultured to 7 days, fixed with 4% paraformaldehyde for 15 min at room temperature. Then the cells were permeabilized with 1% Triton-100 for 30 min at room temperature, washed three times with PBS for 3 min each. In the following the cells were denaturalized with 4 M hydrochloric acid for 30 min at room temperature, and washed again three times using PBS for 3 min each. The cells were blocked with 1% BSA in PBS for 1 h at room temperature, and incubated anti-Brdu (2 μg/mL) overnight at 4°C. Next day, the cells were heated for 30 min at 37°C, and washed two times with PBS for 5 min each. Then the cells were incubated with secondary antibody:FITC-conjugated goat anti-rabbit IgG (1:200) for 90 min at 37°C in dark, and washed again three times with PBS for 5 min each. For the nuclear staining, the cells were counterstained with Hochest33342 (Life Technology, 62249, USA) for 5 min, washed three times with PBS for 5 min each, and analyzed under fluorescence microscope (N-STORM, Nikon, Tokyo, Japan).
Total RNA was purified from testes and culture spermatogonia, respectively. The stored testes tissue pieces were picked out from liquid nitrogen, and the in vitro culture spermatogonia were washed twice with precooling PBS, then processed for RNA isolation using TRizol reagent according to the manufacture’s instructions. The extracted RNA were diluted with DEPC-water and incubated with 10 units of RNase-free DNase for 30 min at 37°C, following inactivation of DNase by adding 1 μL eathylene diamine tetraacetic acid. The first strand cDNA was synthesized according with M-MLV reverse transcriptase manufacture’s instructions (GenScript Corporation, Piscataway, NJ, USA). The primers sequences were list as in Table 1. The reaction mixture for polymerase chain reaction (PCR) contained 2×PCR reaction mixture buffer 10 μL, cDNA 1 μL and specific primers (20 μm each primer), Oct4 and c-kit (annealing at 52°C, 35 cycles), THY-1 and PGP9.5 (annealing at 56°C, 35 cycles). The PCR products were separated and visualized by 2% agarose gel eletrophoresis containing ethidium bromide.
The results are presented as mean±standard error of the mean and statistical analysis was performed by analysis of variance or Student’s t-test after arcsine transformation of the proportional data of spermatid-like cell formation and viability. Duncan’s multiple comparisons test was used to compare mean values among treatments.
The reverse transcription polymerase chain reaction (RT-PCR) analysis revealed that Oct-4, PGP9.5, THY-1 and c-kit were all expressed in testes of prepubertal buffalo, with PCR product fragments of 312 bp for Oct-4, 195 bp for c-kit, 502 bp for THY1, 358 bp for PGP9.5 and 199 bp for β-actin (Figure 1A), respectively. No bands were visible in the case of negative controls, where the cDNA was replaced with purified water. The Hematoxylin eosin (HE) stained germ cells in the testicular sections from the 3 to 7 months old buffalo were easy to identify by their large size, topological position and morphology (Figure 2A). The affinity of the specific antibodies Oct-4, PGP9.5, THY-1, c-kit, and DBA was restricted to germ cells without staining the somatic cells (Figure 2B–F). Five specific antibodies staining was localized on the surface of germ cells, and the cells showed weak affinity for Oct4 (Figure 2D) and strong affinity for PGP9.5 (Figure 2C).
Spermatogonial stem-like cells were isolated and cultured from prepubertal buffalo through differential plating and subsequent Percoll gradient separation (Figure 3A). The buffalo spermatogonial stem-like cells were observed as round cells with a high ratio of nucleus: cytoplasm (Figure 3B). After 24 h culture, SSCs were observed to grow with adherence by morphology (Figure 3C). With the extension of in vitro culture, SSC clusters were observed in the following 3 to 9 days (Figure 3D–F). On the 9 day, the spermatogonial stem-like cell colonies (AP-positive) appeared (4A) and had different forms as single (Figure 4B), cluster (Figure 4C) and aligned (Figure 4D).
With total RNA of buffalo spermatogonial stem-like cell colonies as template, we found that germ cell marker Oct-4, PGP9.5, THY-1, and c-kit mRNA were all expressed by RT-PCR (Figure 1B). In the further, the results of immunocytochemistry analysis of colonies revealed that germ cell markers PGP9.5, THY-1, Oct-4, and c-kit were also expressed in in vitro culture buffalo spermatogonial stem-like cell colonies (Figure 6A–D). It was observed that Oct-4 expression was weaker than other markers both in SSC colonies and in the testis (Figure 2D and Figure 6D).
To optimize the in vitro culture medium of buffalo spermatogonial stem-like cells, different concentrations of FBS and GDNF were added to DMEM. After 9 days in vitro culture, the proportion of DBA-positive cells in 2.5% concentration of FBS group was significantly higher than that of other groups (p<0.05, Figure 5, Table 2). To further optimize the in vitro culture in order to achieve the best possible conditions for buffalo SSCs, these cells were cultured with different concentrations of GDNF in DMEM containing 2.5% FBS. There were significantly more DBA-positive cells in 40 ng/mL GDNF added group than in the 0 ng/mL and 20 ng/mL groups, but there were no significant difference between 40 ng/mL and 80 ng/mL groups (p<0.05, Table 3). Cell proliferation was also determined by Brdu incorporation assay, we found the same result as described above.
In vitro culture SSCs provide an opportunity as an alternative approach to promote the production of genetic modification animals. The key points to advance this research is acquisition of enough quantities of SSCs and determination of specific gene markers, thus the use of different isolation methods and various markers for enrichment and identification of these cells are of paramount importance (Ebata et al., 2008). In the present study, we successfully isolated and identified buffalo spermatogonial stem-like cell using specific germ cell markers, and tested the effects of GDNF and FBS on buffalo SSCs proliferation.
Spermatogonial stem cells were initially distinguishable by their topological position and morphology in different species and different development stages (Heidari et al., 2012). In buffalo, two different subpopulations of type A spermatogonia were distinguished in perpubertal testis through histological examination. The different type A spermatogonia in buffalo testis may be related to the differentiation status of cells.
Germ cell markers are widely used in identifying and sorting of SSCs, but the expression pattern and level of germ cell markers are different between species. It is essential to determine availability of markers for identification of SSCs in vitro for special species (Ebata et al., 2008). The germ cell marker gene Oct-4 was reported expressing in pluripotent cells of morula, inner cell mass, epiblasts (Lengner et al., 2008), primordial germ cells (Encinas et al., 2012), hematopoietic and mesenchymal progenitor cells (Lengner et al., 2008). C-kit has been known to show pleiotropic effects on sterility, hypoplastic anemia, and depletion of masts cells and melanocytes (Ohta et al., 2000). THY-1, a member of Ig super family, was also shown to be highly expressed on rat gonocytes (Ryu et al., 2004), enriched SSCs from mice (Kubota et al., 2003), goat (Abbasi et al., 2013) and prepubertal bulls (Reding et al., 2010) by fluorescent activated cell sorting or magnetic activated cell sorting. DBA and PGP9.5 have been found to be specific markers for spermatogonia in several species such as: mouse (Ebata et al., 2008), pig (Lee et al., 2013), goat (Heidari et al., 2012), bovine (Izadyar et al., 2002) and human (Ebata et al., 2008). In buffalo, DBA, PGP9.5 and Oct-4 have been used to identify spermatogonia in prepubertal buffalo (3 to 7 month) testis (Mahla et al., 2012). Here, the expression of germ cell marker genes PGP9.5, THY-1, c-kit, Oct-4, and DBA were determined in buffalo spermatogonia in both the prepubertal buffalo testis and in vitro cultured SSC colonies by RT-PCR and immunocytochemistry analysis. The Oct-4 expression was weaker than other markers in buffalo, which was in agreement with a previous report (Mahla et al., 2012). The determination of germ cell marker genes in buffalo spermatogonial stem-like cell can do help in identification and sorting of buffalo SSCs in the future.
It was reported that the proliferation efficiency of goat and bovine in vitro cultured SSCs was affected by culture conditions, especially the concentration of growth factors. GDNF alone or combination with other growth factors induced the presence of a higher number of bovine SSC colonies in culture (Aponte et al., 2008). To optimize the in vitro culture condition of buffalo spermatogonial stem-like cell, the effects of different concentrations FBS and GDNF were studied. We found that the number of DBA-positive colonies was increased significantly when supplemented with 2.5% FBS and 40 ng/mL GDNF in culture medium. Additional serum resulted in suppression of SSCs proliferation, suggesting that there were unknown suppression factors in serum. The somatic Sertoli cell secreted factor GDNF was crucial for SSCs maintenance, mice lacking GDNF die within the first day of birth with renal and neuronal abnormalities, but GDNF+/− mice could survive to adulthood and are fertile (Meng et al., 2000; Hofmann, 2008). Histological analysis of GDNF+/− mice testes has shown that the depletion of germ cells often resulted in Sertoli cells only seminiferous tubules, and in other seminiferous tubules the rate of spermatogonial proliferation was significantly reduced (Hofmann, 2008). In this study, we showed that GDNF affected proliferation of in vitro cultured buffalo SSCs in a dose dependent manner, which suggested that GDNF may play a crucial role in maintenance of buffalo spermatogonial stem-like cells.
ACKNOWLEDGMENTS
This work was funded by the China Transgenic Project (2011ZX08007-003) and the National Natural Science Fund (NSFC 31260552).
Figure 1
Detection of germ cell marker transcripts expressed in the prepubertal buffalo testis (A) and spermatogonial stem-like cells colonies (B). M, DNA ladder; Oct-4 (line 1, 312 bp); c-kit (line 3, 195 bp); THY-1 (line 5, 502 bp); PGP9.5 (line 7, 358 bp). Negative control (line 2, 4, 6, 8, 10); β-actin (line 9, 199 bp), positive control. In the (B), the position of PGP9.5 (line 5, 358 bp) and THY-1 (line 7, 502 bp) was exchanged.
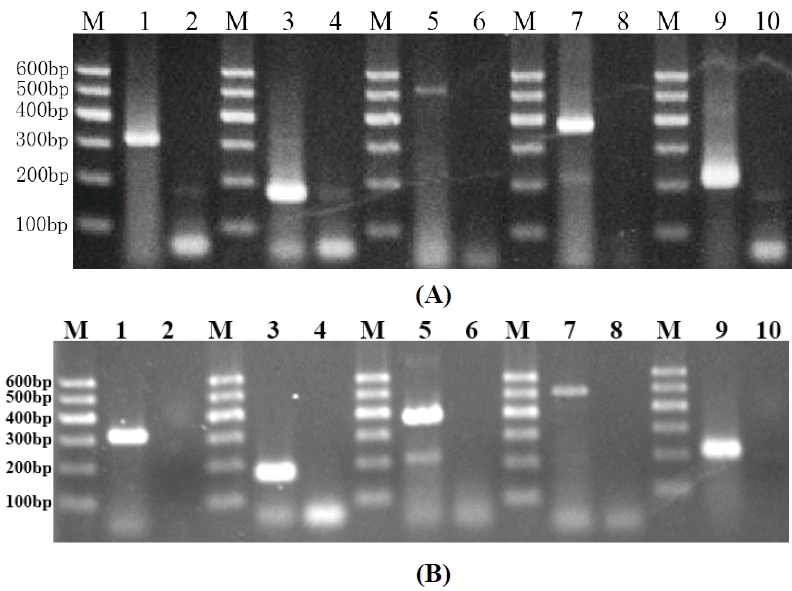
Figure 2
Analysis of germ cell markers expressed in the prepubertal buffalo testis sections by histochemistry. Histological sections of prepubertal buffalo testis were stained with Hematoxylin and Eosin (A). Antibodies against germ cell markers; (B) DBA, (C) PGP9.5, (D) Oct-4, (E) THY-1, and (F) c-kit in histological sections of prepubertal buffalo testis. Scale bar, 100 μm.
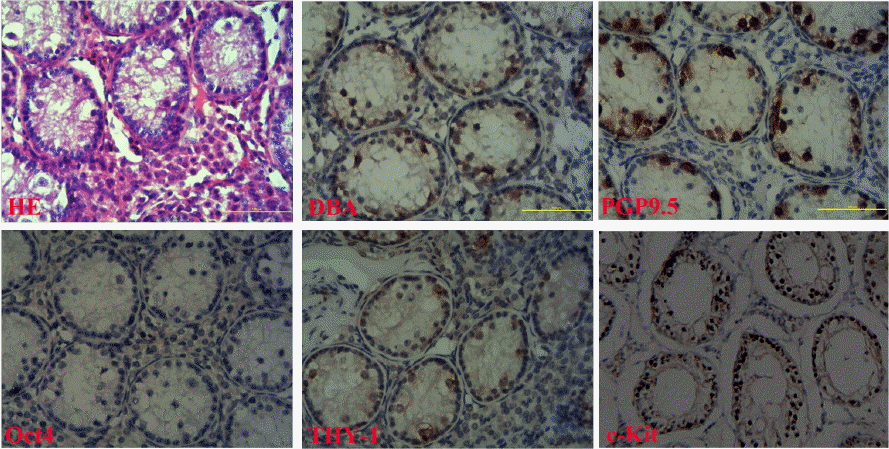
Figure 3
Observation of in vitro culture buffalo spermatogonial stem-like cells. Percoll density gradient isolation of buffalo spermatogonial stem-like cells (A); in vitro culture of Percoll density gradient isolated cells (B); 24 hours after in vitro culture (C); 3 days after in vitro culture (D); 6 days after in vitro culture (E); 9 days after in vitro culture (F); Scale bar, 100 μm.

Figure 4
Buffalo spermatogonial stem-like cells colonies formed after 9 days of culture and were alkaline phosphatase (AP)-positive. After 9 days in vitro culture, the spermatogonial stem-like cells colonies were identify by alkaline phosphatase staining (A) and had different forms: single (B); cluster (C) and aligned (D). Scale bar, 100 μm.
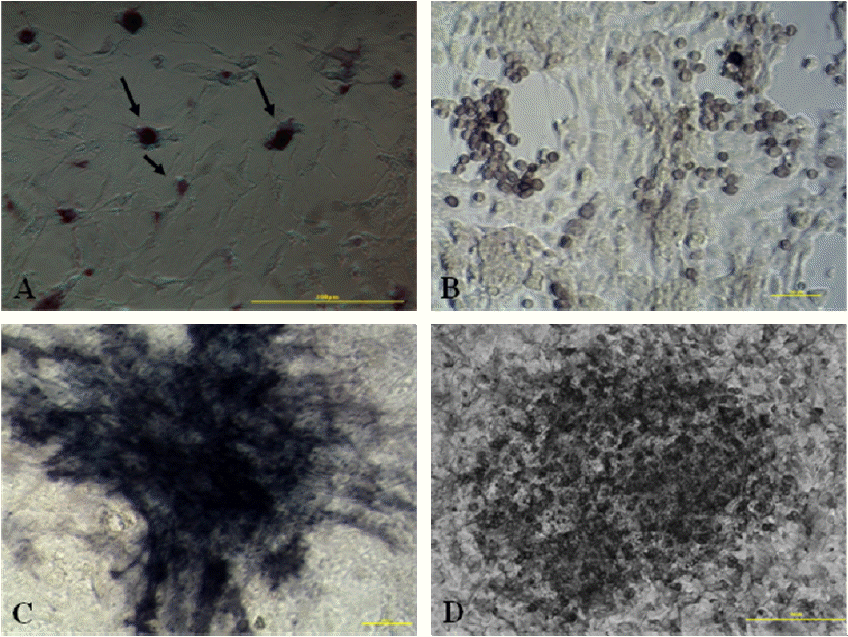
Figure 5
Cell proliferation was detected by BrdU at different concentrations of glial cell-derived neurotrophic factor (GDNF) (ng/mL).
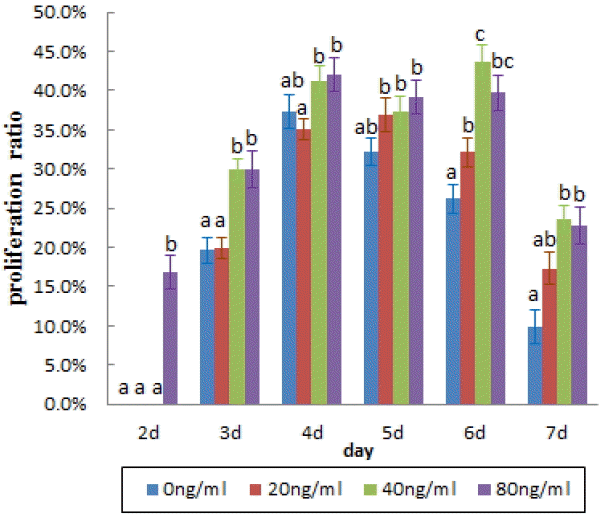
Figure 6
Immunofluorescence characterization of cultured buffalo spermatogonial stem-like cells colonies. Overlapping images after double-fluorescence staining of 9 days cultured cells with Hochest33342 and antibodies. Hochest33342 positive cells stained blue and cells positive for antibodies appear green. PGP9.5 (A); THY-1 (B); c-kit (C); Oct-4 (D). Scale bar, 100 μm.
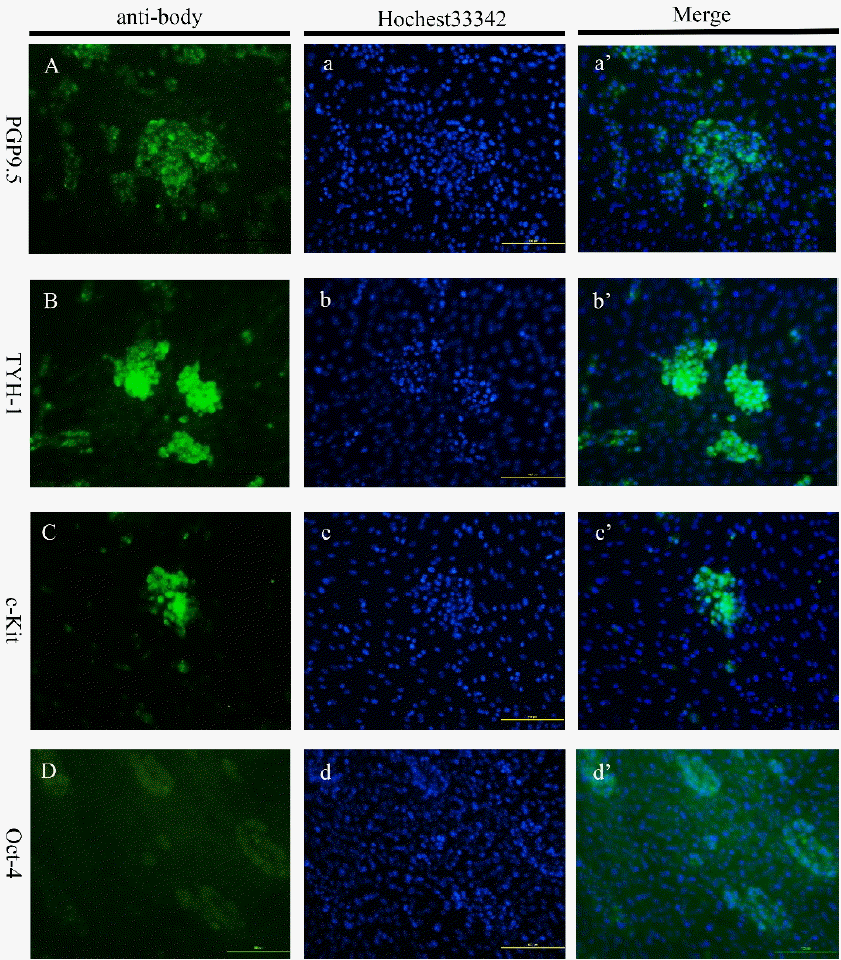
Table 1
Primers for amplification of germ cell marker genes
Table 2
The number of DBA-positive cells at different concentrations of fetal bovine serum groups
| FBS concentration (%) | Times | Average number of positive DBA |
|---|---|---|
| 0 | 3 | 1.33±0.58c |
| 1.5 | 3 | 7.67±1.53b |
| 2.5 | 3 | 12.00±2.22a |
| 3.5 | 3 | 8.67±1.53b |
REFERENCES
Abbasi H, Tahmoorespur M, Hosseini SM, Nasiri Z, Bahadorani M, Hajian M, Nasiri MR, Nasr-Esfahani MH. 2013. Thy1 as a reliable marker for enrichment of undifferentiated spermatogonia in the goat. Theriogenology 80:923–932.


Aponte PM, Soda T, Teerds KJ, Mizrak SC, van de Kant HJG, de Rooij DG. 2008. Propagation of bovine spermatogonial stem cells in vitro. Reproduction 136:543–557.


Chapman KM, Medrano GA, Jaichander P, Chaudhary J, Waits AE, Nobrega MA, Hotaling JM, Ober C, Hamra FK. 2015. Targeted germline modifications in rats using CRISPR/Cas9 and spermatogonial stem cells. Cell Rep 10:1828–1835.



Ebata KT, Yeh JR, Zhang X, Nagano MC. 2008. The application of biomarkers of spermatogonial stem cells for restoring male fertility. Dis Markers 24:267–276.




Encinas G, Zogbi C, Stumpp T. 2012. Detection of four germ cell markers in rats during testis morphogenesis: Differences and similarities with mice. Cells Tissues Organs 195:443–455.


Gaj T, Gersbach CA, Barbas CF. 2013. Zfn, talen, and crispr/cas-based methods for genome engineering. Trends Biotechnol 31:397–405.



Harkey MA, Asano A, Zoulas ME, Torok-Storb B, Nagashima J, Travis A. 2013. Isolation, genetic manipulation, and transplantation of canine spermatogonial stem cells: Progress toward transgenesis through the male germ-line. Reproduction 146:75–90.



Heidari B, Rahmati-Ahmadabadi M, Akhondi MM, Zarnani AH, Jeddi-Tehrani M, Shirazi A, Naderi MM, Behzadi B. 2012. Isolation, identification, and culture of goat spermatogonial stem cells using c-kit and pgp9.5 markers. J Assist Reprod Genet 29:1029–1038.



Hofmann MC. 2008. GDNF signaling pathways within the mammalian spermatogonial stem cell niche. Mol Cell Endocrinol 288:95–103.



Izadyar F, Spierenberg GT, Creemers LB, den Ouden K, de Rooij DG. 2002. Isolation and purification of type a spermatogonia from the bovine testis. Reproduction 124:85–94.


Kanatsu-Shinohara M, Kato-Itoh M, Ikawa M, Takehashi M, Sanbo M, Morioka Y, Tanaka T, Morimoto H, Hirabayashi M, Shinohara T. 2011. Homologous recombination in rat germline stem cells. Biol Reprod 85:208–217.


Kanatsu-Shinohara M, Shinohara T. 2013. Spermatogonial stem cell self-renewal and development. Annu Rev Cell Dev Biol 29:163–187.


Kubota H, Avarbock MR, Brinster RL. 2003. Spermatogonial stem cells share some, but not all, phenotypic and functional characteristics with other stem cells. Proc Natl Acad Sci USA 100:6487–6492.



Lee WY, Park HJ, Lee R, Lee KH, Kim YH, Ryu BY, Kim NH, Kim JH, Kim JH, Moon SH, Park JK, Chung HJ, Kim DH, Song H. 2013. Establishment and in vitro culture of porcine spermatogonial germ cells in low temperature culture conditions. Stem Cell Res 11:1234–1249.


Lee YA, Kim YH, Ha SJ, Kim KJ, Kim BJ, Kim BG, Choi SH, Kim IC, Schmidt JA, Ryu BY. 2014. Cryopreservation of porcine spermatogonial stem cells by slow-freezing testis tissue in trehalose. J Anim Sci 92:984–995.


Lengner CJ, Welstead GG, Jaenisch R. 2008. The pluripotency regulator oct4: A role in somatic stem cells? Cell Cycle 7:725–728.


Mahla RS, Reddy N, Goel S. 2012. Spermatogonial stem cells (SSCs) in buffalo (Bubalus bubalis) testis. PLoS One 7:e36020



Meng X, Lindahl M, Hyvönen ME, Parvinen M, de Rooij DG, Hess MW, Raatikainen-Ahokas A, Sainio K, Rauvala H, Lakso M, Pichel JG, Westphal H, Saarma M, Sariola H. 2000. Regulation of cell fate decision of undifferentiated spermatogonia by GDNF. Science 287:1489–1493.


Momeni-Moghaddam M, Matin MM, Boozarpour S, Sisakhtnezhad S, Mehrjerdi HK, Farshchian M, Dastpak M, Bahrami AR. 2014. A simple method for isolation, culture, and in vitro maintenance of chicken spermatogonial stem cells. In Vitro Cell Dev Biol Anim 50:155–161.


Oatley JM, de Avila DM, Reeves JJ, McLean DJ. 2004. Testis tissue explant culture supports survival and proliferation of bovine spermatogonial stem cells. Biol Reprod 70:625–631.


Ohta H, Yomogida K, Dohmae K, Nishimune Y. 2000. Regulation of proliferation and differentiation in spermatogonial stem cells: The role of c-kit and its ligand scf. Development 127:2125–2131.



Reding SC, Stepnoski AL, Cloninger EW, Oatley JM. 2010. Thy1 is a conserved marker of undifferentiated spermatogonia in the pre-pubertal bull testis. Reproduction 139:893–903.


Rodriguez-Sosa JR, Dobson H, Hahnel A. 2006. Isolation and transplantation of spermatogonia in sheep. Theriogenology 66:2091–2103.


Ryu B-Y, Orwig KE, Kubota H, Avarbock MR, Brinster RL. 2004. Phenotypic and functional characteristics of spermatogonial stem cells in rats. Dev Biol 274:158–170.


Sehgal L, Usmani A, Dalal SN, Majumdar SS. 2014. Generation of transgenic mice by exploiting spermatogonial stem cells in vivo. Methods Mol Biol 1194:327–337.


Shi D, Lu F, Wei Y, Cui K, Yang S, Wei J, Liu Q. 2007. Buffalos (bubalus bubalis) cloned by nuclear transfer of somatic cells. Biol Reprod 77:285–291.


Shi DS, Wang J, Yang Y, Lu FH, Li XP, Liu QY. 2012. DGAT1, GH, GHR, PRL and PRLR polymorphism in water buffalo (Bubalus bubalis). Reprod Domest Anim 47:328–334.


Sisakhtnezhad S, Bahrami AR, Matin MM, Dehghani H, Momeni-Moghaddam M, Boozarpour S, Farshchian M, Dastpak M. 2015. The molecular signature and spermatogenesis potential of newborn chicken spermatogonial stem cells in vitro. In Vitro Cell Dev Biol Anim 51:415–425.


Stockwell S, Herrid M, Davey R, Brownlee A, Hutton K, Hill JR. 2009. Microsatellite detection of donor-derived sperm DNA following germ cell transplantation in cattle. Reprod Fertil Dev 21:462–468.


von Kopylow K, Kirchhoff C, Jezek D, Schulze W, Feig C, Primig M, Steinkraus V, Spiess AN. 2010. Screening for biomarkers of spermatogonia within the human testis: A whole genome approach. Hum Reprod 25:1104–1112.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print





