2. Perini F, Cendron F, Rovelli G, Castellini C, Cassandro M, Lasagna E. Emerging genetic tools to investigate molecular pathways related to heat stress in chickens: a review. Animals 2021; 11:46
https://doi.org/10.3390/ani11010046

4. Madkour M, Salman FM, El-Wardany I, et al. Mitigating the detrimental effects of heat stress in poultry through thermal conditioning and nutritional manipulation. J Therm Biol 2022; 103:103169
https://doi.org/10.1016/j.jtherbio.2021.103169


6. Nawab A, Ibtisham F, Li G, et al. Heat stress in poultry production: Mitigation strategies to overcome the future challenges facing the global poultry industry. J Therm Biol 2018; 78:131–9.
https://doi.org/10.1016/j.jtherbio.2018.08.010


8. Fathi MM, Galal A, El-Safty S, Mahrous M. Naked neck and frizzle genes for improving chickens raised under high ambient temperature: I. Growth performance and egg production. World’s Poult Sci J 2013; 69:813–32.
https://doi.org/10.1017/S0043933913000834

9. Decuypere E, Huybrechts LM, Kuhn ER, Tixier-Boichard M, Merat P. Physiological alterations associated with the chicken sex-linked dwarfing gene. Crit Rev Poult Biol 1991; 3:191–221.
10. Yalcin S, Testik A, Ozkan S, Settar P, Celen F, Cahaner A. Performance of naked neck and normal broilers in hot, warm, and temperate climates. Poult Sci 1997; 76:930–7.
https://doi.org/10.1093/ps/76.7.930


12. Duangjinda M, Tunim S, Duangdaen C, Boonkum W. Hsp70 genotypes and heat tolerance of commercial and native chickens reared in hot and humid conditions. Braz J Poult Sci 2017; 19:7–18.
https://doi.org/10.1590/1806-9061-2016-0245

15. Yadav AK, Tomar SS, Jha AK, Singh J. Importance of molecular markers in livestock improvement: A review. Int J Agric Innov Res 2017; 5:614–21.
17. Falconer DS, Mackay TFC. Introduction to quantitative genetics. Ed 4Harlow, Essex, UK: Longmans Green; 1996.
18. Aksoy T, İlaslan Çürek D, Narinç D, Önenç A. Effects of season, genotype, and rearing system on broiler chickens raised in different semi-intensive systems: performance, mortality, and slaughter results. Trop Anim Health Prod 2021; 53:189
https://doi.org/10.1007/s11250-021-02629-y


22. Adoligbe C, Fernandes A, Osei-Amponsah R, et al. Native chicken farming: A tool for wealth creation and food security in Benin. Int J Livest Prod 2020; 11:146–62.
https://doi.org/10.5897/IJLP2020.0716

23. Halima H, Neser FWC, Van Marle-Koster E, De Kock A. Village-based indigenous chicken production system in north-west Ethiopia. Trop Anim Health Prod 2007; 39:189–97.
https://doi.org/10.1007/s11250-007-9004-6


26. Bell DD, Weaver WD. Commercial chicken meat and egg production. 5th edNew York, USA: Kluwer Academic Publishers; 2002. p. 102–3.
27. Ewing SA, Lay DC, von Borell E. Farm animal well-being-stress physiology, animal behavior, and environmental design. Upper Saddle River, NJ, USA: Prentice Hall; 1999. p. 27–77.
28. Novero RP, Beck MM, Gleaves EW, Johnson AL, Deshazer JA. Plasma progesterone, luteinizing hormone concentrations, and granulosa cell responsiveness in heat-stressed hens. Poult Sci 1991; 70:2335–9.
https://doi.org/10.3382/ps.0702335


29. Rozenboim I, Tako E, Gal-Garber O, Proudman JA, Uni Z. The effect of heat stress on ovarian function of laying hens. Poult Sci 2007; 86:1760–5.
https://doi.org/10.1093/ps/86.8.1760


30. Elnagar SA, Scheideler SE, Beck MM. Reproductive hormones, hepatic deiodinase messenger ribonucleic acid, and vasoactive intestinal polypeptide-immunoreactive cells in hypothalamus in the heat stress-induced or chemically induced hypothyroid laying hen. Poult Sci 2010; 89:2001–9.
https://doi.org/10.3382/ps.2010-00728


31. Mashaly MM, Hendricks GL, Kalama MA, Gehad AE, Abbas AO, Patterson PH. Effect of heat stress on production parameters and immune responses of commercial laying hens. Poult Sci 2004; 83:889–94.
https://doi.org/10.1093/ps/83.6.889


32. Deng W, Dong XF, Tong JM, Zhang Q. The probiotic Bacillus licheniformis ameliorates heat stress-induced impairment of egg production, gut morphology, and intestinal mucosal immunity in laying hens. Poult Sci 2012; 91:575–82.
https://doi.org/10.3382/ps.2010-01293


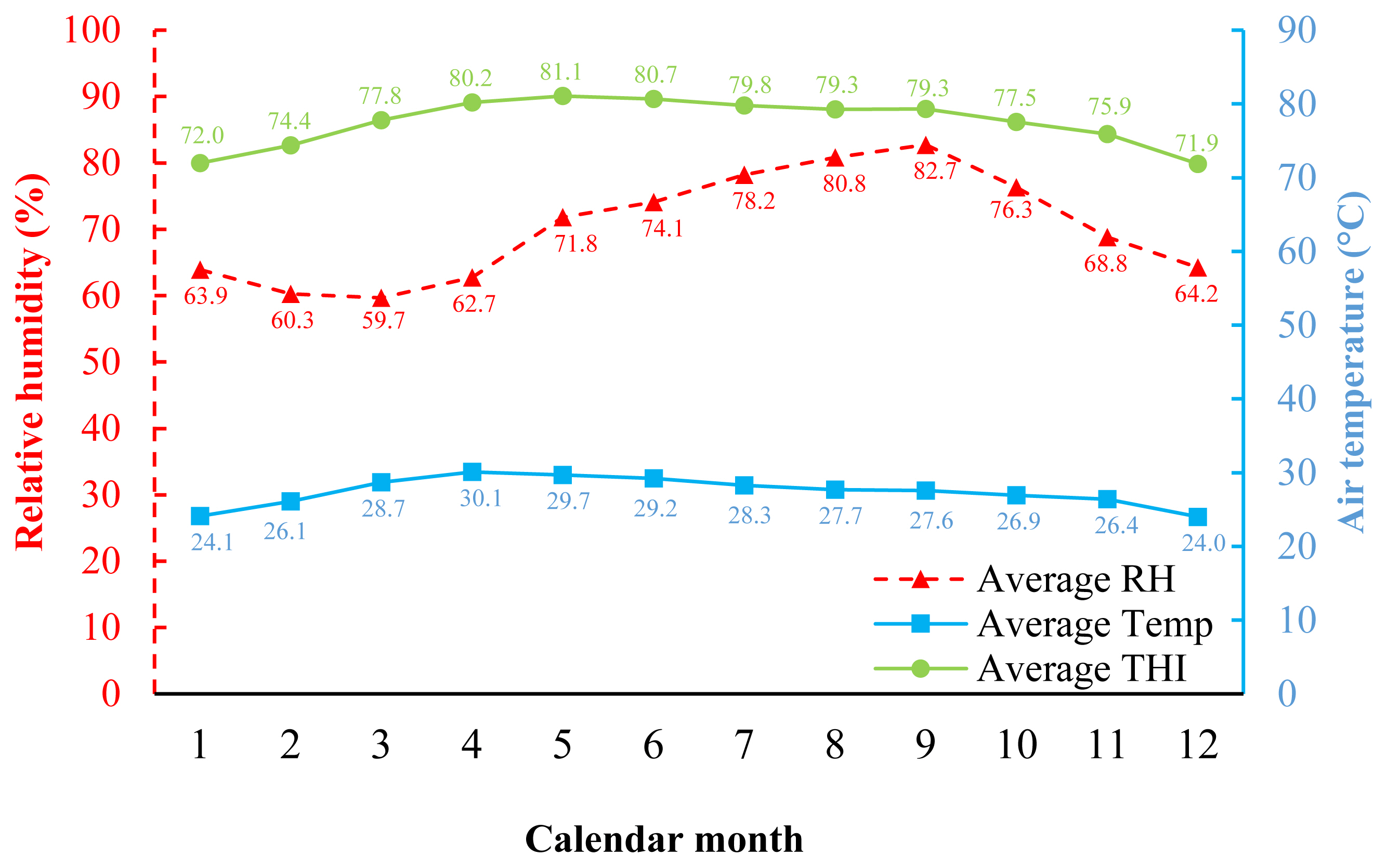
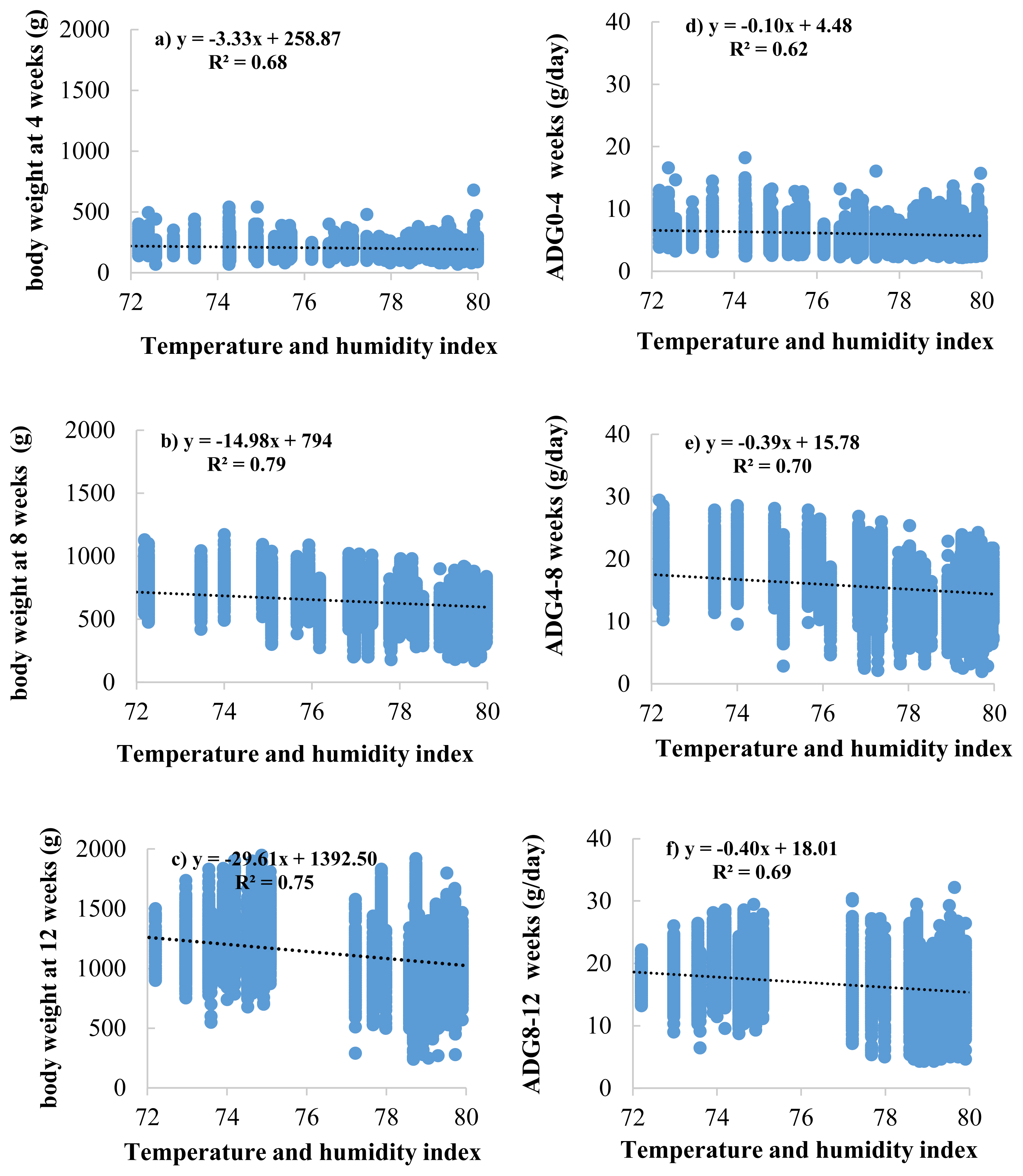
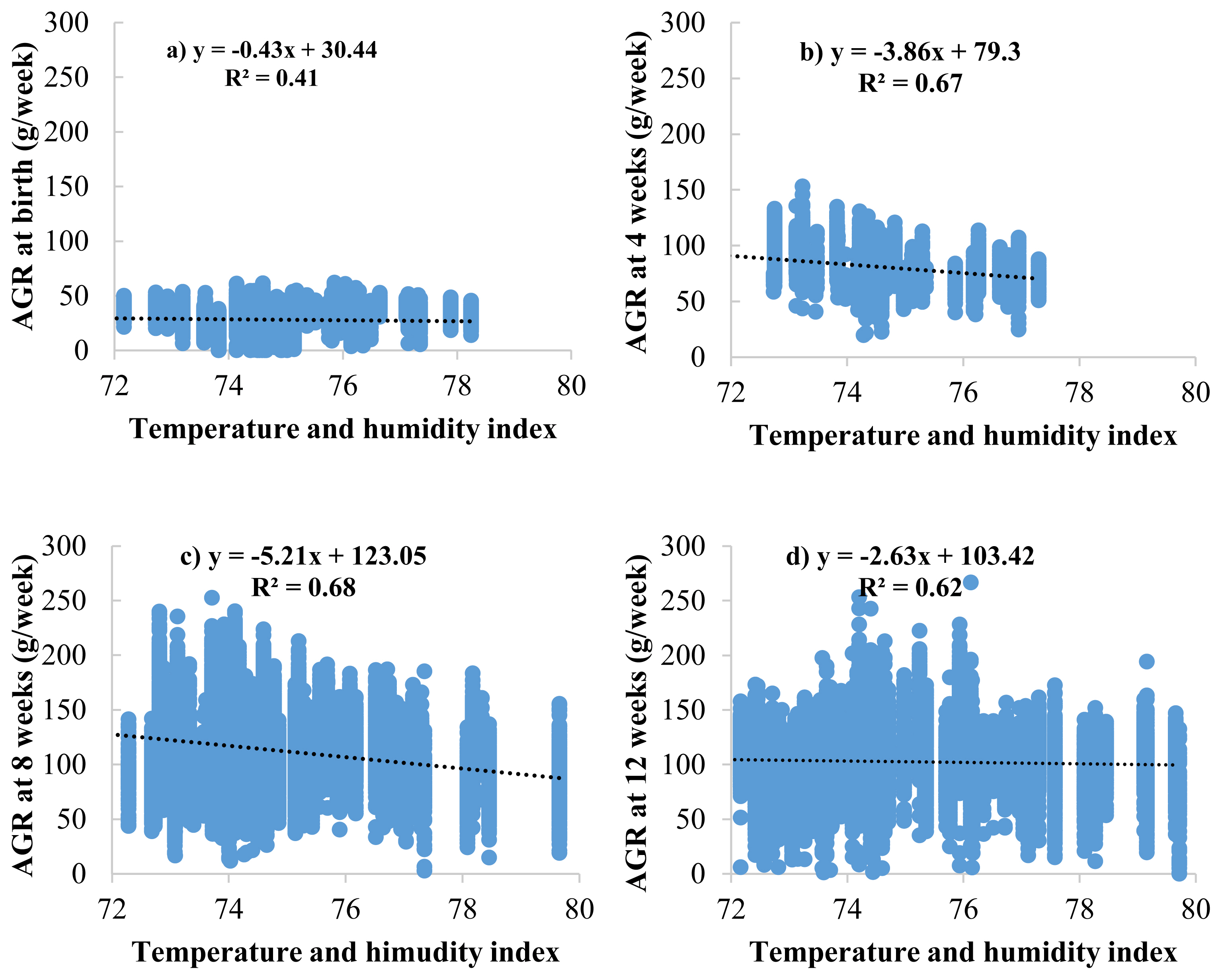
33. Purswell JL, Dozier WA, Olanrewaju HA, Davis JD, Xin H, Gates RS. Effect of temperature-humidity index on live performance in broiler chickens grown from 49 to 63 days of age. In : IX International Livestock Environment Symposium, Valencia Conference Centre; Valencia, Spain. 2012;
https://doi.org/10.13031/2013.41619

34. National Oceanic and Atmospheric Administration. Livestock Hot Weather Stress. Washington, DC, USA: US Government Printing Office; 1976.
35. Mignon-Grasteau S. Genetic parameters of growth curve parameters in male and female chickens. Br Poult Sci 1999; 40:44–51. Br Poult Sci 1999; 40:44–51.
https://doi.org/10.1080/00071669987827


36. Bohmanova J, Misztal I, Tsuruta S, Norman HD, Lawlor TJ. Short communication: Genotype by environment interaction due to heat stress. J Dairy Sci 2008; 91:840–6.
https://doi.org/10.3168/jds.2006-142


41. Tasonieroa G, Cullerea M, Baldan G, Zotte AD. Productive performances and carcase quality of male and female Italian Padovana and Polverara slow-growing chicken breeds. Ital J Anim Sci 2018; 17:530–9.
https://doi.org/10.1080/1828051X.2017.1364611

43. Promket D, Ruangwittayanusorn K. The comparatives of growth and carcass performance of the Thai native chicken between economic selection (Chee KKU12) and natural selection (Chee N). Vet Integr Sci 2021; 19:247–57.
https://doi.org/10.12982/VIS.2021.022

44. Manjula P, Park HB, Seo D, et al. Estimation of heritability and genetic correlation of body weight gain and growth curve parameters in Korean native chicken. Asian-Australas J Anim Sci 2018; 31:26–31.
https://doi.org/10.5713/ajas.17.0179


45. Narinc D, Karaman E, Aksoy T, First MZ. Genetic parameter estimates of growth curve and reproduction traits in Japanese quail. Poult Sci 2014; 93:24–30.
https://doi.org/10.3382/ps.2013-03508


46. Adeyinka IA, Oni OO, Nwagu BI, Adeyinka FD. Genetic parameter estimates for body weights of naked neck broiler chickens. Int J Poult Sci 2006; 5:589–92.
47. Saatchi M, Omed H, Dewi IA. genetic parameters from univariate and bivariate analyses of egg and weight traits in Japanese quail. Poult Sci 2006; 85:185–90.
https://doi.org/10.1093/ps/85.2.185


49. Renaudeau D, Collin A, Yahav S, Basilio V, Gourdine JL, Collier RJ. Adaptation to hot climate and strategies to alleviate heat stress in livestock production. Animal 2012; 6:707–28.
https://doi.org/10.1017/S1751731111002448


50. Mutibvu T, Chimonyo M, Halimani TE. Physiological responses of slow-growing chickens under diurnally cycling temperature in a hot environment. Braz Poult Sci J 2017; 19:567–76.
https://doi.org/10.1590/1806-9061-2017-0485

52. Settar P, Yalcin S, Turkmut L, Ozkan S, Cahanar A. Season by genotype interaction related to broiler growth rate and heat tolerance. Poult Sci 1999; 78:1353–8.
https://doi.org/10.1093/ps/78.10.1353


53. Al-Batshan HA. Performance and heat tolerance of broilers as affected by genotype and high ambient temperature. Asian-Australas J Anim Sci 2002; 15:1502–6.
https://doi.org/10.5713/ajas.2002.1502

54. Aengwanich W. Effects of high environmental temperature on blood indices of Thai indigenous chickens, Thai indigenous chickens crossbred and broilers. Int J Poult Sci 2007; 6:427–30.









 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





