 |
 |
| Anim Biosci > Volume 36(1); 2023 > Article |
|
Abstract
Objective
Adipocyte differentiation is regulated by a variety of functional genes and non-coding RNAs. However, the role of miRNAs in lipid deposition of goat white adipose tissue is still unclear. Therefore, this study revealed the miRNA expression profile in goat subcutaneous adipocytes by sRNA-seq.
Methods
The miRNA expressed in goat subcutaneous preadipocytes and the mature adipocytes were sequenced by sRNA-seq. The differentially expressed miRNAs (DEm) were screened and gene ontology (GO) and Kyoto encyclopedia for genes and genomes (KEGG) analyses were performed. Gain-of-function and loss-of-function combined with oil red O staining, Bodipy staining, and quantitative reverse-transcription polymerase chain reaction (qPCR) were utilized to determine the effect of miR-133a-3p on adipocyte differentiation.
Results
A total of 218 DEm were screened out. The target genes of these DEm were significantly enriched in GO items such as biological regulation and in KEGG terms such as FAK signaling pathway and MAPK signaling pathway. qPCR verified that the expression trend of miRNA was consistent with miRNA-seq. The gain-of-function or loss-of-function of miR-133a-3p showed that it promoted or inhibited the accumulation of lipid droplets, and CCAAT enhancer binding protein ╬▒ (C/EBP╬▒) and C/EBP╬▓ were extremely significantly up-regulated or down-regulated respectively (p<0.01), the loss-of-function also led to a significant down-regulation of peroxisome proliferator activated receptor gamma (PPAR╬│) (p<0.01).
Conclusion
This study successfully identified miRNAs expression patterns in goat subcutaneous adipocytes, and functional identification indicates that miR-133a-3p is a positive regulator of the differentiation process of goat subcutaneous adipocytes. Our results lay the foundation for the molecular mechanism of lipid deposition in meat-source goats from the perspective of miRNA.
Goats as domesticated animals are one of the main sources of meat [1]. Compared with beef and pork, chevon has a unique flavor and is widely welcomed by consumers in China. Mutton flavor substances are mainly accumulated in adipose tissue [2], adipocyte is the main component of adipose tissue and adipocyte differentiation is the key process of adipose deposition. Studies have shown that the differentiation of animal adipocytes is regulated not only by functional genes but also by non-coding RNAs such as miRNAs [3].
MicroRNAs are a class of endogenous non-coding RNA about 22nt and are widely found in plants and animals [4]. In contrast with plants, miRNAs in animals can cleave or suppress translation by binding to target mRNAs that are not completely complementary, thereby playing an important regulatory role [5]. Pri-miRNA is modified by some ways like lysis, transport, and cleavage to produce mature miRNA [6], the latter guide RNA-induced silencing complex to cut the target mRNA domain by complementing the 3ŌĆ▓UTR base of the target gene, thus reducing the expression of target mRNA or inhibiting its translation [4]. miRNA is a critical regulator of gene regulation in the biogenesis and can regulate adipocyte differentiation by interacting with target genes [7]. It can directly bind to important transcription factors associated with adipocyte differentiation or participate in signaling pathways that regulate cell differentiation [8]. For instance, miR-27b target lipoprotein lipase and inhibit human adipose-derived stem cells differentiation [9]. In 3T3-L1 culture, miR-425 was found to regulate lipid generation and metabolism by controlling multidirectional targets such as MAPK4/P38 and Cab39/AMPK [10]. Researchers have identified 104 differentially expressed genes from intramuscular adipocytes in late-laying-period hens and juvenile hens by RNA-seq. Integrated analysis of miRNA and genes showed that differentially expressed miRNAs/mRNAs were enriched in pathways such as ubiquitin-mediated proteolysis, the PPAR signaling pathway and other lipid metabolism pathways, and further study proved that gga-miR-140-5p promotes the differentiation of intramuscular adipocytes via targeting retinoid X receptor gamma [11].
Transcriptome sequencing (RNA-seq) is widely used in detecting the overall transcription level of the species. Besides, it is also used to find new transcripts by analyzing the structure and expression level of the enriched single-stranded mRNA, which confirms the results of the expression level of low-abundance genes [12]. Jianzhou DaŌĆÖer goat is a new breed that is crossed between Jianyang native goat and Nub goat in southwest China, it has the characteristics of fast growth and strong reproductive performance. Therefore, we took subcutaneous adipocytes from Jianzhou daŌĆÖer goat as the object and cultured in vitro, then miRNA-seq was used to explore the expression patterns of miRNA in preadipocytes and adipocytes. Except that, we further characterized the effect of observed miR-133a-3p on the differentiation of goat subcutaneous adipocytes through function gain and function loss experiments. In summary, this study provided a theoretical basis for elucidating the functional mechanism of miRNA-mediated target genes and a reliable mathematical basis for the construction of a goat adipocytes differentiation regulatory network.
All experimental procedures were reviewed and approved by the Institutional Animal Care and Use Committee, Southwest Minzu University (Chengdu, Sichuan, China) (No.18032), and all the experiments complied with the requirements of the directory of the Ethical Treatment of Experimental Animals of China.
The preparation of sample is the same as our previous work [13] and pre-adipocytes (0 d) were regarded as the control PC, and mature adipocytes formed after 3 days of culture were regarded as PE (n = 5). Total RNA from preadipocytes and adipocytes was isolated following protocol of TRIzol (Takara, Dalian, China). Nanodrop (IMPLEN, Palo Alto, CA, USA) was used to detect RNA purity and integrity.
The 15ŌĆō35 nt RNA fragments were separated and collected by gel, 3ŌĆ▓ and 5ŌĆ▓ adapters were connected at both ends of the RNA fragments, then the fragments were reverse transcribed into cDNA, amplified by polymerase chain reaction (PCR) to establish a sequencing library. The constructed library was sequenced by Illumina HiSeq. The process of library construction for RNA-seq was as Figure 1.
The raw reads obtained by RNA-Seq were filtered to remove low-quality reads and reads with adapters to obtain clean reads. Qphred = ŌłÆ10 log10 (e) was used to calculate the error rate. Novoaligen was used to compare clean reads with the miRBase database to identify miRNAs, then mirDeep was used to predict novel miRNAs. WhatŌĆÖs more, novoaligen and samtools were used for quantitative analysis, and reads per million was used for standardization.
DEGSeq was used to analyze the differentially expressed miRNA, Padjust<0.05 was used to indicate the significance of differentially expressed miRNAs (DEm); DEms were subjected to gene ontology (GO) and Kyoto encyclopedia for genes and genomes (KEGG) enrichment analysis; miRanda was used to predict miRNA target genes.
Five differentially expressed (DE) miRNAs were selected randomly for quantitative PCR (qPCR) to verify the accuracy of the sequencing results. Taken 1 ╬╝g of total RNA for reverse transcription according to the instructions of the Mir-X miRNA First-Strand Synthesis kit (Takara, China), qPCR system and procedures were as the kit instructions. miRNAs 5ŌĆ▓ primer information was listed in Table 1, U6 was utilized to normalize miRNA expression, the miRNA 3ŌĆ▓ primer and U6 primer were provided by the kit.
The F3 goat subcutaneous preadipocytes (12-well plate) were transfected with chemically synthesis chi-miR-133a-3p mimics, inhibitors, and negative control (Genepharma, Shanghai, China) by TurboFect Transfection Reagent (Thermo Fisher Scientific, Waltham, MA, USA) when the cell confluence reached 80%. All operations were in accordance with the instructions. After 14 hours of transfection, added 2 mL 50 ╬╝M oleic acid (Sigma, St. Louis, MO, USA) medium to each well to induce differentiation, and the adipocytes were collected after 3 days of culture.
Cultured cells (24-well plate, transfection was same as ŌĆ£Cell TransfectionŌĆØ, reagent consumption halved) were washed with phosphate-buffered saline (PBS) and fixed with 4% formaldehyde for 30 min at room temperature. Then the cells were stained using the Bodipy (Thermo Fisher Scientific, USA) working solutions for 20 min under dark conditions, then the cells were washed 3 times with PBS. The shape and number of lipid droplets were observed under the microscope and take images. And the ImageJ was used to quantify Bodipy staining results.
The operations of oil red O staining were the same as Bodipy. After being photographed, oil red O dye (Solarbio, Beijing, China) was extracted from stained adipocytes with 100% isopropanol (Jinshan, Chengdu, China), and the Oil red signal was quantified by measuring the optical density at 490 nm.
RNA isolated was the same as ŌĆ£Preparation of RNA-seq SamplesŌĆØ, the efficiency of chi-miR-133a-3p mimics and inhibitor was detected by miRNAs qPCR. mRNA was reverse transcribed by RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, USA), and the cDNA was used to detect the genes expression level. Ubiquitously expressed transcript gene (UXT) was selected as internal reference gene to normalize the mRNA levels in adipocyte. The qPCR system included TB Green Premix Ex Taq II (2├Ś) (Takara, China) 10 ╬╝L, sense/antisense primer 1 ╬╝L, cDNA 1 ╬╝L, ddH2O up to 20 ╬╝L, 95┬░C 3 min, 95┬░C 10 s, melting-out temperature (TM) 10 s, 72┬░C 15 s, 40 cycles, primer information was shown as Table 2.
After removing low-quality reads, about 9.5 to 12.1 million and 9.5 to 11.4 million of clean reads were obtained from the PC and PE, respectively. The error rate of each sample was less than 0.01%, and both Q20 and Q30 were greater than 94% (Table 3). The mapping ratio of the samples in the sRNA library was between 84% to 90% (Table 4). The above data comprehensively indicated the data with high quality, which provided a reliable basis for further analysis.
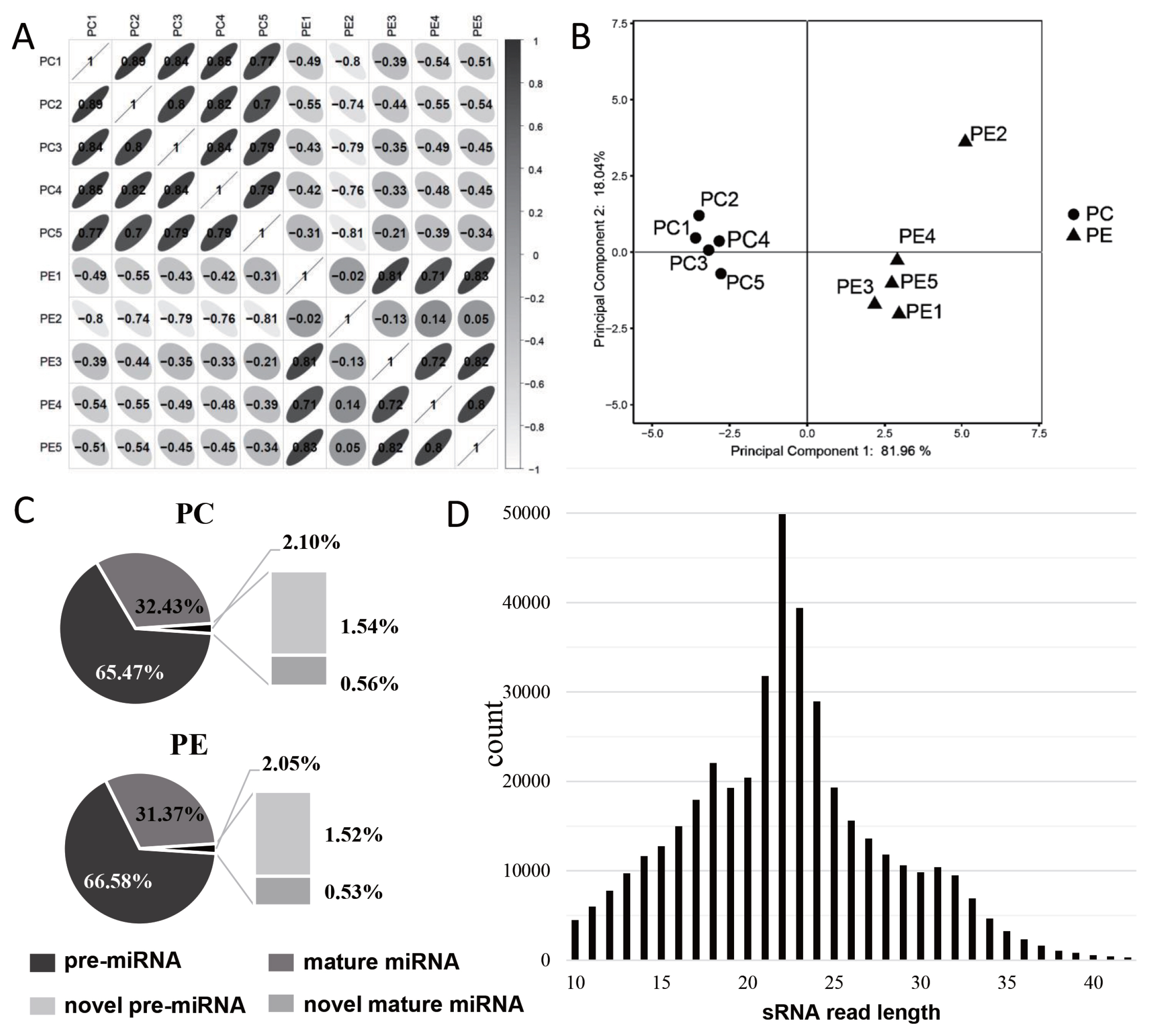
Correlation analysis of the RNA-seq samples showed that the samples were clearly divided into two groups, and the correlation coefficient of the samples in each group ranged from 0.952 to 0.995 (Figure 2A), but the correlation between PE2 and other samples was relatively low. The results of principal component analysis (PCA) showed that the samples were well correlated, however, PE2 was separated from PE group (Figure 2B). Therefore, the data of PE2 were excluded from subsequent sequencing data analysis to avoid affecting the accuracy of the data.
The reads in the sRNA library were analyzed, and it was found that about 98% of the reads in PC and PE were mapped to the annotated miRNA, and pre-miRNA accounted for a higher proportion in either annotated miRNA or novel miRNA (Figure 2C). The lengths of the screened miRNAs were analyzed, and we found that the miRNAs were mainly concentrated in 21 to 24 nt, about 35.73%; and the miRNAs with lengths less than 18 nt, 18 to 24 nt, and more than 24 nt were 20.32%, 50.44%, and 29.24%, respectively (Figure 2D).
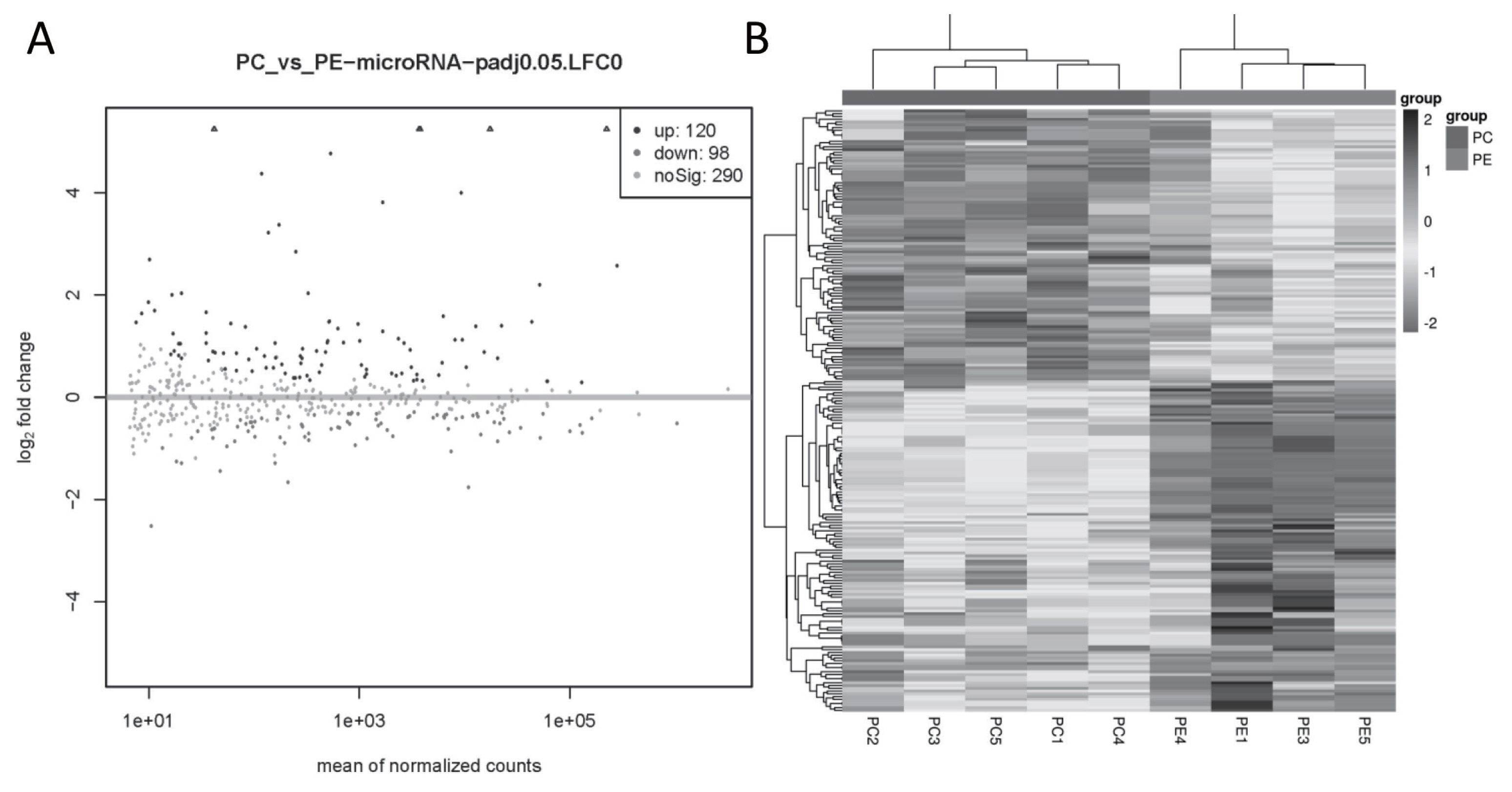
A total of 218 miRNAs were DE in the PC and PE group, of which 120 were up-regulated (89 annotated and 31 novel miRNAs) and 98 were down-regulated (67 annotated and 31 novel miRNAs) (Figure 3A). The cluster analysis of DE miRNAs found that the samples were clearly clustered into two categories (Figure 3B).
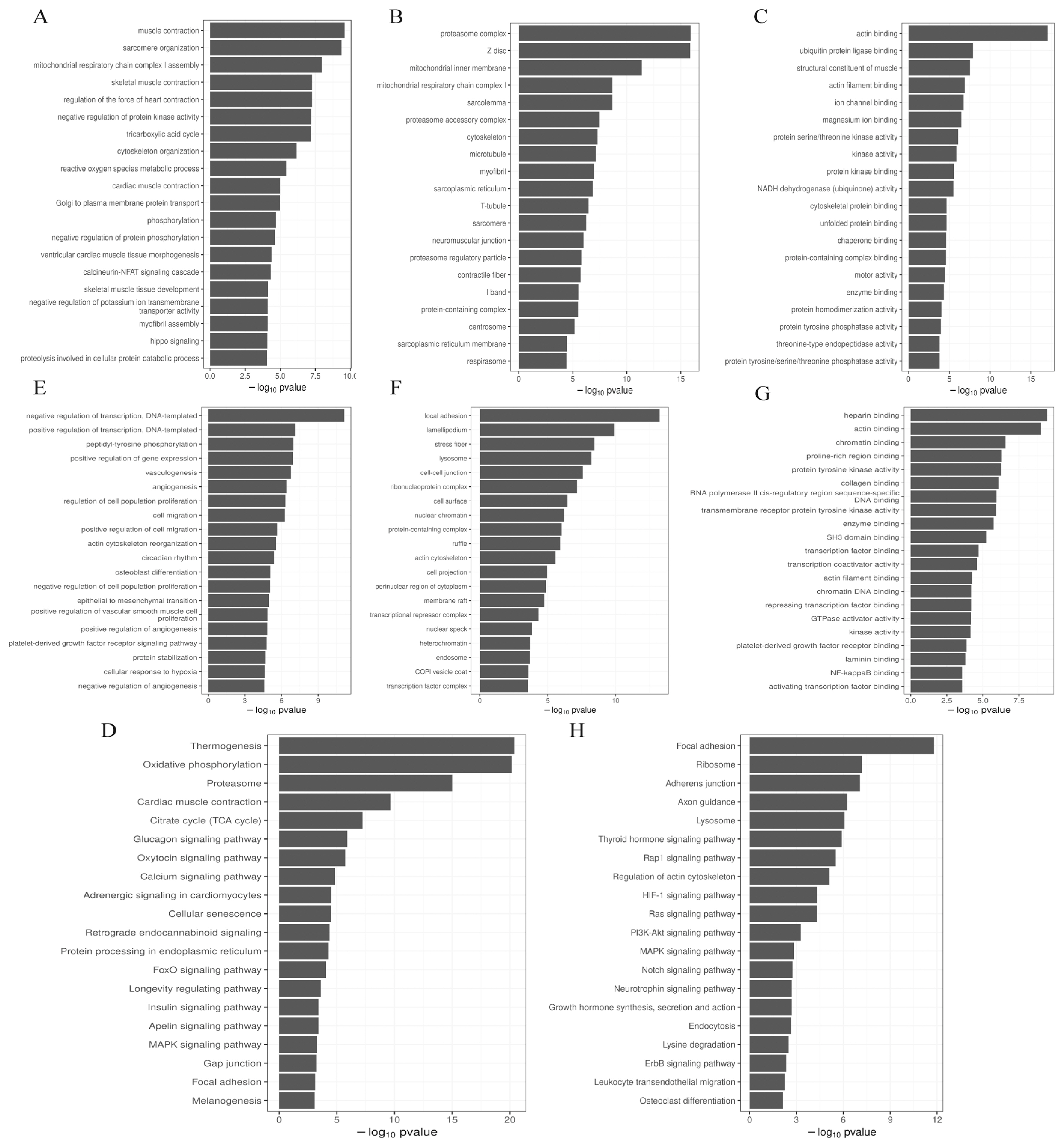
The prediction of the DE miRNA target genes was by GOseq, and perform GO enrichment analysis on the target genes, we found that the most enriched biological process (BP) (Figure 4A), cellular component (CC) (Figure 4B) and molecular function (MF) (Figure 4C) term of the target genes of up-regulated miRNAs were muscle contraction, proteasome complex and actin binding, respectively. The most significantly enriched BP (Figure 4E), CC (Figure 4F), and MF (Figure 4G) term for the target genes of down-regulated miRNAs were negative regulation of transcription (DNA-templated), focal adhesion, and heparin binding, respectively. KEGG enrichment analysis found that the common enrichment pathways for up-regulated and down-regulated miRNAs were Focal adhesion and MAPK signaling pathway. In addition, the up-regulated miRNAs were most enriched in the Thermogenesis pathway, the Oxytocin signaling pathway, FoxO signaling pathway, and insulin signaling pathway were also significantly enriched (Figure 4D). The down-regulated miRNAs were most significantly enriched in the Focal adhesion pathway, and the PI3K-Akt signaling pathway and Notch signaling pathway were also enriched (Figure 4H).
Five DE miRNAs were chosen randomly to verify the expression level by qPCR, the result showed that the expression trend of miRNAs by qPCR was similar to miRNA-seq (Figure 5).
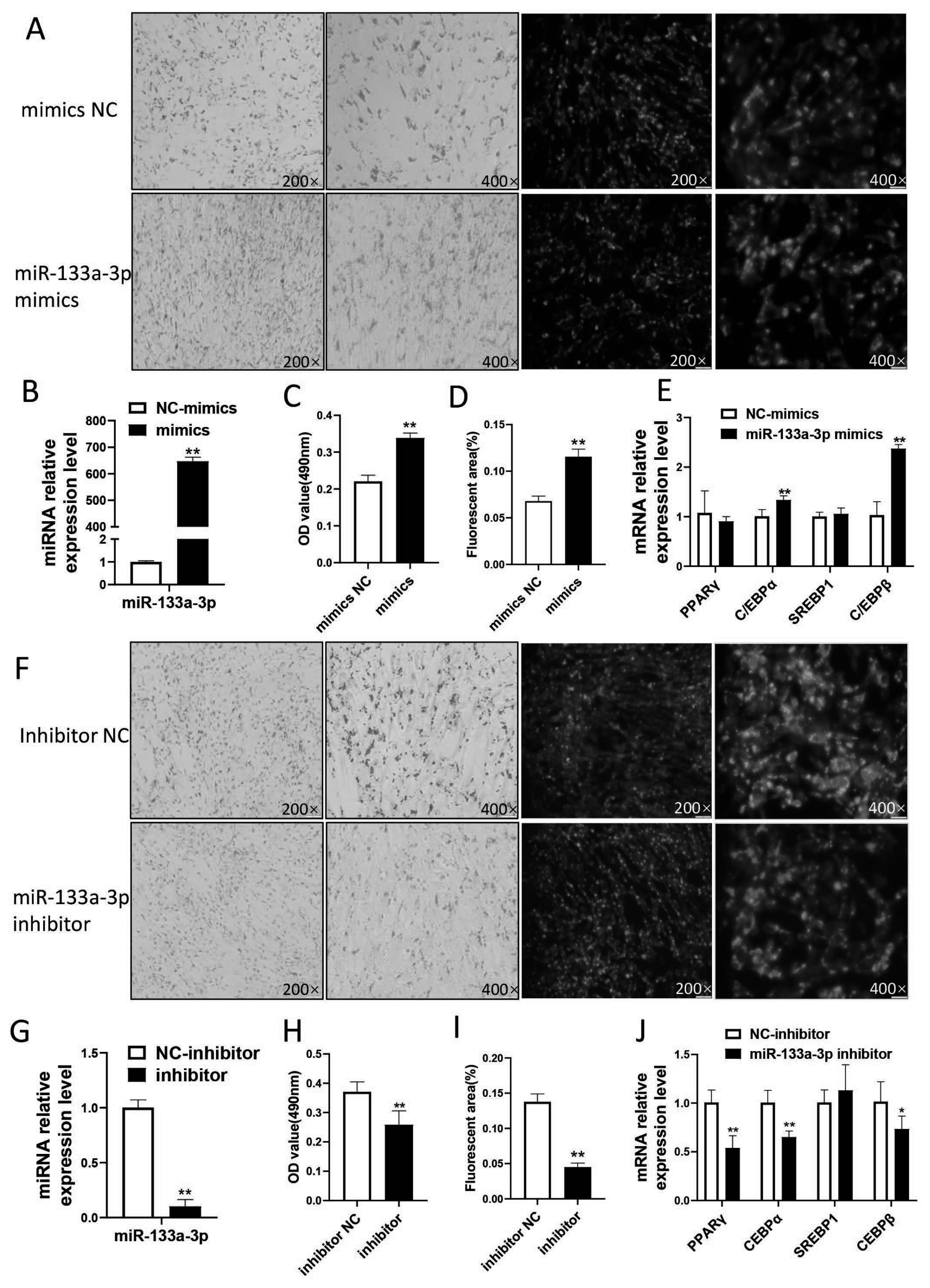
miR-133a-3p was selected for functional verification in further study. We firstly constructed an overexpression model by transfecting subcutaneous pre-adipocytes with miR-133a-3p mimics. The result showed that the expression of miR-133a-3p was up-regulated 647 times in subcutaneous pre-adipocytes that transfected with miR-133a-3p mimics when compared to the control (Figure 6B). Oil red O staining and BODIPY staining showed that the accumulation of lipid droplets in the mimics group was greater than that in the NC group (p<0.01) (Figure 6A,C,D). Besides, we found that the differentiation marker gene CCAAT enhancer binding protein ╬▒ (C/EBP╬▒) and C/EBP╬▓ was significantly increased in miR-133a-3p mimics (p<0.01) (Figure 6E).
Then we conducted a miR-133a-3p loss model by transfecting with miR-133a-3p inhibitor. After transfection of miR-133a-3p inhibitor, the expression of miR-133a-3p was down-regulated about 90% (Figure 6G). Oil red O staining and BODIPY staining showed that the lipid droplet accumulation in the inhibitor group was significantly less than that in the NC group (p<0.01 (Figure 6F,H,L)). The expression level of differentiation marker genes peroxisome proliferator activated receptor gamma (PPAR╬│) and C/EBP╬▒ was extremely significantly reduced (p<0.01), and C/EBP╬▓ was significantly reduced (p<0.05) (Figure 6J).
miRNAs played a regulatory role in many biological processes, including adipocyte differentiation and lipid metabolism [14]. The regulatory network of miRNAs was extremely complicated because one miRNA could target multiple different mRNAs. Adipocyte differentiation was the main source of fat deposit, but the regulatory role of most miRNAs in goat subcutaneous adipocyte differentiation was still unclear. RNA-seq technology has been widely used in different samples to screen key mRNAs and non-coding RNAs [15] or key signaling pathways [16]. Therefore, miRNA-seq was used in this study to obtain key miRNAs in the differentiation of goat subcutaneous adipocytes to understand molecular mechanism in goat fat deposition. As a result, differently expressed miRNAs and several pathways enriched in regulating the differentiation of adipocytes were identified through functional analyses.
From both the aspect of cell sample morphology observation and sequencing quality evaluation results, it was shown that the sequencing samples with high quality, the sequencing results with high utilization rate. We found that the length of miRNA was mainly distributed in 21 to 24 bp, which is in line with the length of mammalian miRNA sequence [4]. In this study, using |log2FoldChange|>0 and q value<0.05 as the thresholds, 218 differentially expressed miRNAs were screened, of which 120 were up-regulated and 98 were down-regulated. Among them, the five miRNAs with the largest up-regulation changes were miR-208b, miR-499-5p, miR-133a-5p, miR-133a-3p and miR-1, and the five miRNAs with the largest down-regulation changes were miR-335-3p, let-7c-3p, miR-196a, miR-16b-5p, and miR-15b-5p. It has been reported that finishing pigs fed a diet containing 0.3% butyrate had a higher intramuscular fat content than the pigs fed a basal diet, the fat deposits associated with this process resulted in a decrease in the expression level of miR-133a-3p and an increase in expression of miR-208b and miR-499-5p [17]. miR-499-5p could target PTEN, which in turn affected the PI3K/AKT/GSK signaling pathway and glycogen synthesis [18]. miR-133a could inhibit the browning of adipocytes [19], which may partly decrease adipocyte loss. Inhibition of miR-499-5p expression in mice with nonalcoholic fatty liver could reduce the accumulation of lipid and triglyceride and reduce total cholesterol in serum in individual mice [20]. miR-133a-5p was down-regulated in subcutaneous fat deposition in rats fed high-fat diet (HFD), but up-regulated in HFD mice [21]. Overexpression of miR-196a induces preadipocyte differentiation by increasing the expression of adipocyte-specific markers, lipid accumulation and triglyceride content [22]. miR-196a could mediate the conversion of white adipocyte progenitor cells to brown adipocytes through Hoxc8-C/EBP╬▓ [23]. In C2C12 cell adipogenesis induced by cocktail, miR-15b-5p was down-regulated, it targeted Akt3 to stimulate adipogenic differentiation [24]. The above studies have shown that DE miRNAs also play an important role in the differentiation of goat white adipocytes.
The DE miRNA target genes were predicted, and enrichment analysis was performed. KEGG enrichment analysis showed that the common enrichment pathways for up-regulated and down-regulated miRNAs were focal adhesion kinase (FAK) and MAPK signaling pathway. Interestingly, Thermogenesis pathway was most significant enriched by up-regulate miRNAs, and the FAK pathway was most significant enriched by down-regulate miRNAs. Studies have shown that the FAK participated in adipocyte differentiation, and its cleavage by calpain was required to fulfill the final maturation of adipocytes [25]. Study had also shown that liver kinase B1 (LKB1) inhibited goat intramuscular adipogenesis through the FAK pathway, blocking this pathway rescued the observed phenotypes in LKB1 knockdown adipocytes [26]. MAPK phosphatase-1 (MKP-1) plays an essential role in adipocyte differentiation through down-regulation of p42/p44 MAPK activity [27]. C1q/TNF-related protein 6 (CTRP6) promotes porcine intramuscular and subcutaneous adipocyte differentiation through the MAPK signaling pathway [28]. Interleukin-27 (IL-27) promoted adipocyte thermogenesis and energy expenditure by activating p38 MAPK-PGC-1╬▒ signaling and stimulating uncoupling protein 1 (UCP1) [29].
Adipose tissue acts against cold through thermogenesis. Thermogenesis is initiated physiologically by cold sensation or dietary intake, which stimulates sympathetic release of norepinephrine to activate ╬▓3-adrenergic receptors (╬▓3-ARs) in adipose tissue [30,31]. This pathway promotes thermogenesis through a protein kinase A (PKA)-p38 MAPK-Ucp1 signaling axis [32,33]. Besides, pathways such as the calcium signaling pathway, FoxO signaling pathway, and insulin signaling pathway PI3K-Akt signaling pathway and Notch signaling pathway were also significantly enriched. Numerous studies have confirmed that the above-mentioned enriched pathways were involved in the differentiation process of adipocytes. For instance, Calcium signaling pathway was a key pathway in regulating obesity [34], Ca2+ promote adipocyte differentiation and metabolism [35], the PI3K/Akt signaling pathway could increase lipid accumulation [36] and promote cell adipogenic differentiation [37]. Both FoxO signaling pathway [38], insulin signaling pathway [39] and Notch signaling pathway [40] have been reported to be involved in adipocyte adipogenesis. These signaling pathways can not only function independently, but they also interact with and regulate each other, for example, in adipocytes, the activation of p38-MAPK signaling pathway can promote intracellular calcium transport to regulate adipocytes metabolism to reduce obesity [41,42]. Therefore, the enriched pathways of DEm are mostly related to adipocyte differentiation, this work will provide support for further studies revealing the regulatory network during goat adipocyte differentiation.
We selected miR-133a-3p as the object for further research. We found that simulating its expression in preadipocytes could promote the differentiation of subcutaneous adipocytes, while inhibiting its expression inhibited their differentiation, indicating that miR-133a-3p was a positive regulator of subcutaneous adipocyte differentiation. Studies had reported that the expression of miR-133a-3p was low in the subcutaneous adipose tissue of obese patients, and its expression was increased in individuals who lose weight after energy restriction, suggesting that miR-133a-3p seems to be a negative regulator of adipogenesis [43]. This was inconsistent with the results of our study, presumably due to different experimental subjects. So, how miR-133a-3p regulates goat subcutaneous adipocyte adipogenesis still needs further study.
This study identified 218 differentially expressed miRNAs between pre-adipocytes and mature adipocytes in goat subcutaneous fat. The pathway enrichment analysis of DE miRNA target genes showed that pathways related to adipocyte differentiation and fatty acid metabolism were enriched. Furthermore, we found that miR-133a-3p positively regulate the differentiation of goat adipocytes.
Notes
ACKNOWLEDGMENTS
We want to affectionately thank Prof. Bai Wenlin for his assistance in the revision of this article.
Figure┬Ā1
The flow diagram of sRNA-Seq. Total RNA extracted from samples was separated by gel, then the 15ŌĆō35 nt fragments were selected to connect 3ŌĆ▓ adaptor and 5ŌĆ▓ adaptor orderly, and these fragments were reverse transcribed and amplified by polymerase chain reaction to complete the small RNA library construction, further sequencing by Illumina HiSeq.
Figure┬Ā2
Characteristic analysis of miRNAs. (A) Bar plot of correlation coefficient analysis, the darker of the color means the greater the correlation coefficient, Pearson correlation coefficient (r) was showed in the box, r<0 represents negative correlation and r>0 represents positive correlation. (B) PCA analysis between PC and PE, PE2 showed a large distance from other samples in the PE group, and we excluded PE2 data in later analysis. (C) The composition of miRNA in PC and PE group and the ratio of each component. (D) Length distribution map of miRNAs. PCA, principal component analysis; PC, preadipocytes; PE, mature adipocytes by adipogenic differentiation.
Figure┬Ā3
DE miRNA in goat subcutaneous preadipocytes and adipocytes. (A) Volcano plot of DE miRNAs, |log2FoldChange|>0 and q value<0.05 as the threshold, log2FoldChange>0, q value<0.05 represents the up-regulated miRNA and shown as black dots, while log2FoldChange<0, q value<0.05 represents the down-regulated miRNA and shown as dark grey dots, and light gray dots means the miRNA with no significant. (B) Heatmap of DE miRNAs, the black box represents higher expression, and the grey box represents lower expression, each horizontal represents a DE miRNA. DE, differentially expressed.
Figure┬Ā4
GO and KEGG analysis of DE miRNAs target genes. (A) GO terms in BP enriched by up-regulated miRNAs target genes. (B) GO terms in CC enriched by up-regulated miRNAs target genes. (C) GO terms in MF. (D) enriched by up-regulated miRNAs target genes. (D) Up-regulated miRNAs target genes are enriched in the KEGG pathway. (E) Down-regulated miRNAs target genes are enriched in GO terms in BP. (F) Down-regulated miRNAs target genes are enriched in GO terms in CC. (G) Down-regulated miRNAs targets gene-enriched GO terms in MF. (H) KEGG pathway enriched by up-regulated miRNAs target genes. GO, gene ontology; KEGG, Kyoto encyclopedia for genes and genomes; DE, differentially expressed; BP, biological process; CC, cellular component; MF, molecular function.
Figure┬Ā5
Validating the expression trends of the 5 differentially expressed miRNAs by quantitative reverse-transcription polymerase chain reaction (qPCR). The log2Foldchange>0 indicates that the miRNA is up-regulated, while log2Foldchange<0 indicates that the miRNA is down-regulated. The qPCR results showed the same trend with RNA-seq, indicating the RNA-seq data reliable.

Figure┬Ā6
The effect of miR-133a-3p in goat subcutaneous adipocytes differentiation (A), (F): Lipid droplet accumulation after simulating/inhibiting miR-133a-3p expression by oil red O and BODIPY staining. (B), (G): efficiency of miR-133a-3p overexpression/interference. (C), (H): the OD value by oil red O staining. (D), (I): fluorescence quantification by BODIPY staining. (E), (J): changes in the expression of marker genes by overexpression/inhibition of 133. * means p<0.05, ** means p<0.01. 3 biological replicates were set for each treatment group.
Table┬Ā1
miRNAs 5ŌĆ▓ primer information
Table┬Ā2
qPCR primer information for genes
qPCR, quantitative reverse-transcription polymerase chain reaction; TM, melting-out temperature; PPAR╬│, peroxisome proliferator-activated receptor ╬│; C/EBP╬▒, CCAAT enhancer binding protein ╬▒; C/EBP╬▓, CCAAT enhancer binding protein ╬▓; SREBP1, Sterol-regulatory element binding protein 1; UXT, ubiquitously expressed transcript gene.
Table┬Ā3
Quality assessment of sequencing data
Table┬Ā4
Result of sRNA sequence reference genome comparison
REFERENCES
1. Ripoll G, Alcalde MJ, Arg├╝ello A, C├│rdoba MG, Panea B. Effect of rearing system on the straight and branched fatty acids of goat milk and meat of suckling kids. Foods 2020; 9:471
https://doi.org/10.3390/foods9040471



2. Watkins PJ, Frank D. Heptadecanoic acid as an indicator of BCFA content in sheep fat. Meat Sci 2019; 151:33ŌĆō5.
https://doi.org/10.1016/j.meatsci.2019.01.005


3. Gregoire FM, Smas CM, Sul HS. Understanding adipocyte differentiation. Physiol Rev 1998; 78:783ŌĆō809.
https://doi.org/10.1152/physrev.1998.78.3.783


4. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116:281ŌĆō97.
https://doi.org/10.1016/s0092-8674(04)00045-5


5. Ambros V. The functions of animal microRNAs. Nature 2004; 431:350ŌĆō5.
https://doi.org/10.1038/nature02871


6. Ha M, Kim VN. Regulation of microRNA biogenesis. Nat Rev Mol Cell Biol 2014; 15:509ŌĆō24.
https://doi.org/10.1038/nrm3838


7. Sun YM, Qin J, Liu SG, et al. PDGFR╬▒ Regulated by miR-34a and FoxO1 Promotes Adipogenesis in Porcine Intramuscular Preadipocytes through Erk Signaling Pathway. Int J Mol Sci 2017; 18:2424
https://doi.org/10.3390/ijms18112424



8. Chen L, Song J, Cui J, et al. microRNAs regulate adipocyte differentiation. Cell Biol Int 2013; 37:533ŌĆō46.
https://doi.org/10.1002/cbin.10063


9. Hu X, Tang J, Hu X, et al. MiR-27b impairs adipocyte differentiation of human adipose tissue-derived mesenchymal stem cells by targeting LPL. Cell Physiol Biochem 2018; 47:545ŌĆō55.
https://doi.org/10.1159/000489988


10. Qi R, Wang J, Wang Q, et al. MicroRNA-425 controls lipogenesis and lipolysis in adipocytes. Biochim Biophys Acta Mol Cell Biol Lipids 2019; 1864:744ŌĆō55.
https://doi.org/10.1016/j.bbalip.2019.02.007


11. Zhang M, Li DH, Li F, et al. Integrated analysis of MiRNA and genes associated with meat quality reveals that gga-mir-140-5p affects intramuscular fat deposition in chickens. Cell Physiol Biochem 2018; 46:2421ŌĆō33.
https://doi.org/10.1159/000489649


12. Hu Y, Wang K, He X, Chiang DY, Prins JF, Liu J. A probabilistic framework for aligning paired-end RNA-seq data. Bioinformatics 2010; 26:1950ŌĆō7.
https://doi.org/10.1093/bioinformatics/btq336



13. Li X, Zhang H, Wang Y, et al. RNA-seq analysis reveals the positive role of KLF5 in the differentiation of subcutaneous adipocyte in goats. Gene 2022; 808:145969
https://doi.org/10.1016/j.gene.2021.145969


14. Peng Y, Yu S, Li H, Xiang H, Peng J, Jiang S. MicroRNAs: emerging roles in adipogenesis and obesity. Cell Signal 2014; 26:1888ŌĆō96.
https://doi.org/10.1016/j.cellsig.2014.05.006


15. Wang Z, Gerstein M, Snyder M. RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 2009; 10:57ŌĆō63.
https://doi.org/10.1038/nrg2484



16. Bakhtiarizadeh MR, Salehi A, Alamouti AA, Abdollahi-Arpanahi R, Salami SA. Deep transcriptome analysis using RNA-Seq suggests novel insights into molecular aspects of fat-tail metabolism in sheep. Sci Rep 2019; 9:9203
https://doi.org/10.1038/s41598-019-45665-3



17. Zhang Y, Yu B, Yu J, et al. Butyrate promotes slow-twitch myofiber formation and mitochondrial biogenesis in finishing pigs via inducing specific microRNAs and PGC-1╬▒ expression. J Anim Sci 2019; 97:3180ŌĆō92.
https://doi.org/10.1093/jas/skz187



18. Wang L, Zhang N, Pan HP, Wang Z, Cao ZY. MiR-499-5p contributes to hepatic insulin resistance by suppressing PTEN. Cell Physiol Biochem 2015; 36:2357ŌĆō65.
https://doi.org/10.1159/000430198


19. Liu W, Bi P, Shan T, et al. miR-133a regulates adipocyte browning in vivo. PLoS Genet 2013; 9:e1003626
https://doi.org/10.1371/journal.pgen.1003626



20. Liu H, Wang T, Chen X, et al. Inhibition of miR-499-5p expression improves nonalcoholic fatty liver disease. Ann Hum Genet. 2020. Jan. 20[Epub]. https://doi.org/10.1111/ahg.12374

21. Youssef EM, Elfiky AM, Soliman Bangly, Abu-Shahba N, Elhefnawi MM. Expression profiling and analysis of some miRNAs in subcutaneous white adipose tissue during development of obesity. Genes Nutr 2020; 15:8
https://doi.org/10.1186/s12263-020-00666-0



22. Ning X, Liu S, Qiu Y, et al. Expression profiles and biological roles of miR-196a in swine. Genes (Basel) 2016; 7:5
https://doi.org/10.3390/genes7020005



23. Mori M, Nakagami H, Rodriguez-Araujo G, Nimura K, Kaneda Y. Essential role for miR-196a in brown adipogenesis of white fat progenitor cells. PLoS Biol 2012; 10:e1001314
https://doi.org/10.1371/journal.pbio.1001314



24. Hou Y, Fu L, Li J, et al. Transcriptome analysis of potential miRNA involved in adipogenic differentiation of C2C12 myoblasts. Lipids 2018; 53:375ŌĆō86.
https://doi.org/10.1002/lipd.12032


25. Li JJ, Xie D. Cleavage of focal adhesion kinase (FAK) is essential in adipocyte differentiation. Biochem Biophys Res Commun 2007; 357:648ŌĆō54.
https://doi.org/10.1016/j.bbrc.2007.03.184


26. Xiong Y, Wang YX, Xu Q, et al. LKB1 regulates goat intramuscular adipogenesis through focal adhesion pathway. Front Physiol 2021; 12:755598
https://doi.org/10.3389/fphys.2021.755598



27. Sakaue H, Ogawa W, Nakamura T, Mori T, Nakamura K, Kasuga M. Role of MAPK phosphatase-1 (MKP-1) in adipocyte differentiation. J Biol Chem 2004; 279:39951ŌĆō7.
https://doi.org/10.1074/jbc.M407353200


28. Wu W, Zhang J, Zhao C, Sun Y, Pang W, Yang G. CTRP6 regulates porcine adipocyte proliferation and differentiation by the AdipoR1/MAPK signaling pathway. J Agric Food Chem 2017; 65:5512ŌĆō22.
https://doi.org/10.1021/acs.jafc.7b00594


29. Wang Q, Li D, Cao G, et al. IL-27 signalling promotes adipocyte thermogenesis and energy expenditure. Nature 2021; 600:314ŌĆō8.
https://doi.org/10.1038/s41586-021-04127-5


30. Okla M, Kim J, Koehler K, Chung S. Dietary factors promoting brown and beige fat development and thermogenesis. Adv Nutr 2017; 8:473ŌĆō83.
https://doi.org/10.3945/an.116.014332



31. Sakamoto T, Takahashi N, Goto T, Kawada T. Dietary factors evoke thermogenesis in adipose tissues. Obes Res Clin Pract 2014; 8:e533ŌĆō9.
https://doi.org/10.1016/j.orcp.2013.12.002


32. Rosen ED, Spiegelman BM. What we talk about when we talk about fat. Cell 2014; 156:20ŌĆō44.
https://doi.org/10.1016/j.cell.2013.12.012



33. Zhang Y, Li R, Meng Y, et al. Irisin stimulates browning of white adipocytes through mitogen-activated protein kinase p38 MAP kinase and ERK MAP kinase signaling. Diabetes 2014; 63:514ŌĆō25.
https://doi.org/10.2337/db13-1106


34. Song Z, Wang Y, Zhang F, Yao F, Sun C. Calcium signaling pathways: key pathways in the regulation of obesity. Int J Mol Sci 2019; 20:2768
https://doi.org/10.3390/ijms20112768



35. He YH, He Y, Liao XL, et al. The calcium-sensing receptor promotes adipocyte differentiation and adipogenesis through PPAR╬│ pathway. Mol Cell Biochem 2012; 361:321ŌĆō8.
https://doi.org/10.1007/s11010-011-1118-5


36. 36 Xiao J, Bai XQ, Liao L, et al. Hydrogen sulfide inhibits PCSK9 expression through the PI3K/Akt-SREBP-2 signaling pathway to influence lipid metabolism in HepG2 cells. Int J Mol Med 2019; 43:2055ŌĆō63.
https://doi.org/10.3892/ijmm.2019.4118



37. Song BQ, Chi Y, Li X, et al. Inhibition of notch signaling promotes the adipogenic differentiation of mesenchymal stem cells through autophagy activation and PTEN-PI3K/AKT/mTOR pathway. Cell Physiol Biochem 2015; 36:1991ŌĆō2002.
https://doi.org/10.1159/000430167


38. Lee JH, Jung HA, Kang MJ, Choi JS, Kim GD. Fucosterol, isolated from Ecklonia stolonifera, inhibits adipogenesis through modulation of FoxO1 pathway in 3T3-L1 adipocytes. J Pharm Pharmacol 2017; 69:325ŌĆō33.
https://doi.org/10.1111/jphp.12684


39. Krycer JR, Quek LE, Francis D, et al. Insulin signaling requires glucose to promote lipid anabolism in adipocytes. J Biol Chem 2020; 295:13250ŌĆō66.
https://doi.org/10.1074/jbc.RA120.014907



40. Shan T, Liu J, Wu W, Xu Z, Wang Y. Roles of notch signaling in adipocyte progenitor cells and mature adipocytes. J Cell Physiol 2017; 232:1258ŌĆō61.
https://doi.org/10.1002/jcp.25697


41. Bl├╝thgen N, Legewie S. Systems analysis of MAPK signal transduction. Essays Biochem 2008; 45:95ŌĆō108.
https://doi.org/10.1042/bse045095


42. Han HJ, Lee YJ. Insulin stimulates Ca2+ uptake via PKC, cAMP, and p38 MAPK in mouse embryonic stem cells. Life Sci 2005; 76:2903ŌĆō19.
https://doi.org/10.1016/j.lfs.2004.10.060


43. Oliveira MS, Rheinheimer J, Moehlecke M, et al. UCP2, IL18, and miR-133a-3p are dysregulated in subcutaneous adipose tissue of patients with obesity. Mol Cell Endocrinol 2020; 509:110805
https://doi.org/10.1016/j.mce.2020.110805


- TOOLS






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





