 |
 |
| Anim Biosci > Volume 35(12); 2022 > Article |
|
Abstract
Objective
The present study aimed to quantify the effects of fermented soybean meal (FSBM) on broiler chickens’ performance by employing a meta-analysis approach.
Methods
A total of 16 studies were included in the database after being systematically selected using a PRISMA protocol. Hedges’ g effect size was used to quantify pooled standardized mean difference (SMD) using random-effects models at 95% confidence intervals (95% CI). Publication bias among studies was computed with Egger’s test and visualized using funnel plots.
Results
Results indicated that dietary FSBM inclusion increased final body weight (BW) (SMD = 0.586, 95% CI: 0.221 to 0.951, p = 0.002) of broiler chickens, particularly in starter period (SMD = 0.691, 95% CL: 0.149 to 1.233, p = 0.013) while in the finisher period, the effect was weaker (SMD = 0.509, 95% CI: 0.015 to 1.004, p = 0.043). Average daily gain (ADG), feed intake (FI), and feed conversion ratio (FCR) were not affected with FSBM inclusion when compared to control. Subgroup analysis revealed that FI increased in starter period (SMD = 0.582, 95% CI: 0.037 to 1.128, p = 0.036). When considering types of microorganism as moderating variables in the subgroup analysis, we found that Aspergillus oryzae, mixed probiotics+bromelain protease, Bacillus subtilis, and Lactobacillus bacteria significantly increased ADG and FI (p<0.01). Additionally, either Bacillus subtilis+protease or Bacillus subtilis alone decreased FCR (p<0.001). However, meta-regression analysis showed that levels of FSBM inclusion had no effects on final BW (p = 0.502), ADG (p = 0.588), FI (p = 0.861), and FCR (p = 0.462).
Soybean meal (SBM) is a major contributor of protein and indispensable amino acids such as lysine, tryptophan, threonine, isoleucine, and valine in broiler diets [1]. These nutritional components play a crucial role to support the rapid metabolism and growth rate of modern broiler chickens. However, the presence of anti-nutritional factors (ANFs) such as protease inhibitor (i.e. trypsin inhibitor), antigenic protein, and phytic acid limits the nutrients’ bioavailability and digestion thus impairing the growth of broilers and their health status [2,3]. A considerable amount of undigested protein that enters the broiler hindgut is detrimental. The undigested protein can form toxic compounds such as ammonia and polyamine and it becomes fermentable for bacterial pathogens [3,4]. An excessive amount of fermentable proteins can promote an increase of pathogenic bacteria associated with the incidence of intestinal diseases such as coccidiosis and necrotic enteritis (NE) on farm [5].
In the last two decades, microbial fermentation has been proposed as an economically feasible method to enhance the nutritional content as well as eliminate the ANFs concentration of SBM [2,6,7]. Fermented-SBM (FSBM) is a product resulted from fungal or bacterial fermentation (mainly Aspergillus niger, Aspergillus oryzae, Bacillus subtilis, and Lactobacillus group) [8–11]. Previous studies reported that fermentation successfully decreased ANFs and increased protein and amino acids composition of SBM. For instance, Wu et al [7] reported considerable increase of threonine, leucine, and isoleucine by 29.8%, 34.7%, and 27.8%, respectively, by fermenting SBM with Bacillus stearothermophilus (ATCC 7953). Microbial fermentation of bacillus, lactobacillus, and yeast was also reported to increase protein composition of SBM from 8.2% to 18.9% [6,10] as well as removed antinutrient substances and enhanced nutrient utilization [2,12]. These beneficial effects led in an improvement of broiler growth performance and feed efficiency.
There is accumulating evidence of significantly increased average body weight gain (BWG), final body weight (BW), and feed efficiency [6,11,13]. Other reports also suggested that FSBM positively modulated ileal and cecal microbial composition, intestinal morphology, immune status, and carcass traits of broiler chickens [6,7,11,14]. However, inconsistent results were observed whereas several studies with FSBM inclusion did not show any effect on average daily gain (ADG) and final BW [13,15]. There was also an indication of level-dependency in which a decreasing trend on BW was detected in studies that used a used high level of FSBM [16,17]. In the experiments, FSBM was used as either total or partial replacement to SBM which may contribute to the magnitude of the effect on broiler performance and feed efficiency. Inclusion of FSBM varied among studies, ranging from less than 2% to 30% of the diet [13,15].
To our knowledge, there is no consensus regarding the threshold of FSBM inclusion in the diet. However, large variation in the inclusion levels, microbial types used as fermenters, and environmental conditions among experiments might elucidate why the outcomes differed. Thus, we hypothesized that levels of FSBM and microbial fermenters are two major factors contributing to the effect size on broiler growth performance. To generalize the effect, it is useful to integrate available studies and calculate the effect size with relevant statistical method [18]. The present study therefore aimed to quantify the effect of FSBM on broiler performance and feed efficiency by employing a meta-analysis approach.
A literature search was performed on scientific digital platforms of Science Direct ( https://www.sciencedirect.com), PubMed Central ( https://www.ncbi.nlm.nih.gov/pmc/), and Google Scholar ( https://scholar.google.com) with the keywords “fermented”, “soybean meal”, and “broiler chicken”. In the PubMed central, we used combinatory terms with specific Boolean operators as follows: ((fermented [All Fields] AND ("soybeans"[MeSH Terms] OR "soybeans"[All Fields] OR "soybean"[All Fields]) AND ("meals"[MeSH Terms] OR "meals"[All Fields] OR "meal"[All Fields])) AND (broiler [All Fields] AND ("chickens"[MeSH Terms] OR "chickens"[All Fields])). All relevant titles of articles from the platforms were imported into the reference manager to easily select the papers.
Preferred reporting items for systematic review and meta-analysis (PRISMA) protocol was employed to reduce publication bias and to ensure the quality of meta-analysis [19]. Accordingly, inclusion criteria were determined as follows: i) the article was published in English in a peer-reviewed journal; ii) reported FSBM inclusion level in the diet, processing method as well as microbes used as fermenter; iii) described the broiler strain, length of rearing periods, replication, and sample size per replicate (n); iv) used similar nutrient specification in the treatment and control groups; v) provided means and appropriate variances (standard deviation or standard error of the means) for at least the variables of BW or feed intake (FI). Experiments reporting only ADG or feed conversion ratio (FCR) were considered because they allowed to calculate final BW and FI, and vice versa. In addition to the criteria, studies that used soy protein concentrate, probiotics, or prebiotics (i.e. mannan-oligosaccharide) were not included because these materials may interfere the results. Challenged experiments with pathogens were also disregarded.
In total, 1,613 articles were detected by employing the above-mentioned strategy. A total of 365 papers were excluded because they were review articles (86), book chapters (147), Encyclopedia (23), and other non-relevant types of articles (109). Following this, 1,248 remaining papers were evaluated to meet the criteria. Two investigators (Niati Ningsih [NN] and Rahma Fitriastuti [RF]) reviewed the titles and abstracts of the papers and excluded non relevant publications from the imported database. Subsequently, two independent investigators (Agung Irawan [AI] and Adib Norma Respati [ANR]) reviewed the selected studies to assess the risk of publication bias among individual studies [20,21]. In the subsequent step, the investigators (NN and RF) extracted data from the selected articles and did calculations for analyses. In total, 16 studies representing 68 comparisons were eligible and therefore were integrated into the database (Table 1). The selection process of the studies is provided in Figure 1.
Information on the broiler chickens used (number, strain, sex), replication, study design, FSBM inclusion level including processing methods and microbes used for fermentation, country in which the experiment was carried out, and nutrient compositions of the diet in respective periods (starter, grower, finisher) such as metabolizable energy (ME), crude protein (CP), lysine, and methionine were retrieved from each paper into Microsoft Excel spreadsheet. The main outcomes used for meta-analysis were final BW (g), ADG (g/d), FI (g/bird/period), and FCR (g feed/g BW). Therefore, mean values including the corresponding variant (standard deviation [SD]) or standard error of the mean (SEM) of each unit of comparison were extracted. The SD value of each study was calculated from SEM using the formula: SD = SE×sqrt(n); where n = sample size or number of replicates, when the study only provided SE rather than SD value. When available, the values from graphical data were extracted by employing an extraction tool of WebPlotDigitizer ( https://apps.automeris.io/wpd/). The summary of the studies used for meta-analysis is presented in Table 1 while summary statistics of the nutrient specification and outcome variables are shown in Table 2.
Meta-analysis was performed using the meta and metafor packages [26] available in R statistical software. Means of control and treatment in the studies were considered as continuous outcome data. Effect size of individual study was calculated with Hedges’ g and expressed as standardized mean differences (SMD) in a forest plot following a random-effects model at 95% confidence intervals (CIs). We considered Hedges’ g because it is easy to interpret and has strong statistical power which can control study bias that may occur in small sample sizes [27]. Hedges’ g outcome estimates >0 with p≤0.05 show a significant higher on BW, ADG, FI, or FCR due to FSBM inclusion when compared to control. Heterogeneity index (I2) among studies was analyzed using the DerSimonian and the Laird test (Q-statistic) at a significance level p≤0.05. The degree of heterogeneity was categorized as no heterogeneity (0<I2≤25%), low (25%<I2≤50%), moderate (50%<I2≤75%), and high (I2>75%) [28].
Meta-regression was conducted to evaluate the relationship between FSBM inclusion levels with the effect size (Hedges’ g) of the outcome variables. In addition, subgroup analysis was also performed to quantify the effect sizes of several factors that may influence the magnitude of the effect size. In this study, rearing period (starter and finisher) and type of microbes used to ferment SBM were used in the subgroup meta-analysis using random effects models.
In addition to the risk of bias from studies, publication bias was assessed by using funnel plots and the results are presented in Figure 2. Egger’s test was conducted to know the presence of bias [29]. Bias among publications was considered to be significant at p≤0.05. In case there was a presence of bias, sensitivity analysis was carried out using the leave-one-out study analysis to compare between-study heterogeneity (I2) [28] and to determine the effect size of the studies contributing to the bias [30].
This study included 5,176 broiler chickens from various strains predominantly by Ross 308 (57.14%) while the rest were Arbor Acres (21.43%), Cobb 500 (14.29%), and Vencob (7.14%). A total of 35.71% of studies used either male or mixed sexes of broiler chickens while others were female (14.30%) and not given (14.30%). All studies used corn and soybean-based diet for their experiments. Inclusion levels of FSBM varied among studies, from 15 g/kg diet to 361 g/kg diet (Table 1). In each study, microbial strain for SBM fermentation also varied in which Bacillus subtilis was the most frequently used, corresponded to 57.14% of the studies, either in a single strain or mixed with other microorganisms. Other microorganisms applied for fermentation were fungi (Aspergillus niger and Aspergillus oryzae), yeast (Saccharomyces cerevisiae), and from lactic acid bacteria group such as Bacillus subtilis, Lactobacillus spp., Lactobacillus acidophilus, Lactobacillus plantarum, and Bacillus stearothermophilus. Information on the nutrient composition of each study was summarized and is presented in Table 2 for ME (kcal/kg), CP (%), lysine (%), and methionine (%) both in starter and finisher phases. As indicated in Table 2, the nutritional specifications were appropriate to the nutrient recommendation of NRC [31]. Additionally, descriptive statistic of the chemical compositions of SBM and FSBM are provided in Table 3. Processing SBM to FSBM increased CP by 14.3% while decreased trypsin inhibitor, Glycinin, mg/g, and β-Conglycinin, mg/g by 83.1%, 82.2%, and 79.7%, respectively.
Summary of meta-analysis is presented in Table 4 and the forest plots for subgroup analysis are presented in the Supplementary materials 1. Comparisons between studies with or without FSBM in the present meta-analysis suggested that replacing SBM with FSBM had a significant positive effect on final BW of broiler chickens as shown from the pooled SMD (SMD = 0.586, 95% CI: 0.221 to 0.951, p = 0.002), regardless of inclusion levels. Since there were high variations of between-study heterogeneity (83.5% of I2 statistic), subgroup analyzes were performed to indicate which factors mediating the effect sizes. Results from subgroup analysis based on rearing period showed that significant increase was more pronounced in the starter period (≤21 d) (SMD = 0.691, 95% CL: 0.149 to 1.233, p = 0.013) compared to finisher period (>21) (SMD = 0.509, 95% CI: 0.015 to 1.004, p = 0.043) with considerable between-study variations (84.9% and 82.2% of I2 statistics, respectively). In addition, most microbial fermentation resulted in a significant increase of final BW except for Aspergillus niger (SMD = 0.259, 95% CI: −0.322 to 0.839, p = 0.382) and multiple strain microbes containing lactic acid bacteria and yeast (SMD = 0.191, 95% CI: −0.274 to 0.656, p = 0.420).
Results from the Egger’s test indicated that there was no publication bias (p>0.05) for BW, FI, and FCR among studies which was supported by symmetrical funnel plots (Figure 2). However, there was a significant publication bias on ADG (p = 0.047). Thus, we performed a sensitivity analysis to detect source of bias. As a result, Wang et al [32] was eliminated from the dataset because it had the highest effect size among others and contributed to increase the heterogeneity index of the outcome. After excluding Wang et al [32] as the major source of variance, there was an improvement in publication bias (p = 0.069) and therefore we decided to use this final dataset for meta-analysis. However, there was no different on the SMD before and after removing the data of Wang et al [32]. Forest plots of subgroup analysis on FI are given in the Supplementary Figures 1 to 8. Overall SMD of ADG as a result of new dataset was 0.294 (95% CI = −0.060 to 0.648, p>0.10) with high heterogeneity (I2 = 66.3%, Q<0.001). Although there was no effect both in starter and finisher period, however, positive effects were detected corresponding to the FSBM with Aspergillus oryzae (SMD = 0.258, 95% CI: −0.322 to 0.838, p = 0.002), Mixed probiotics+bromelain (SMD = 0.612, 95% CI: 0.027 to 1.196, p<0.001), Bacillus subtilis (SMD = 1.640, 95% CI: 0.712 to 2.567, p = 0.006), and Lactobacillus microbes (SMD = 0.591, 95% CI: 0.172 to 1.009, p<0.001) (Table 4) in which no heterogeneity was detected (I2 = 0.00%, Q>0.10). By contrast, SBM fermented with Bacillus stearothermophilus significantly decreased the ADG (SMD = −1.654, 95% CI: −2.323 to −0.985, p<0.001) with low level of heterogeneity (I2 = 32.6%, Q = 0.191).
Of the 51 comparisons satisfying the inclusion criteria, treatment with FSBM did not alter FI (SMD = 0.115, 95% CI: −0.272 to 0.502, p = 0.560) at the degree of heterogeneity of 85.9% (Q = 0.001) (Table 4). However, when it turned to rearing period, FSBM utilization significantly increased the FI in the starter period (SMD = 0.582, 95% CI: 0.037 to 1.128, p = 0.036) with high heterogeneity (I2 = 80.3%, Q = 0.001), but it did not affect FI in finisher period (p = 0.223). Subgroup analysis on microbes used in the fermentation process indicated that groups of Aspergillus oryzae, Bacillus subtilis +protease, and mixed of probiotics+bromelain were responsible for increasing the FI. Contrary effect was found on the use of Bacillus stearothermophilus which significantly reduced cumulative FI (SMD = −2.008, 95% CI: −3.654 to −0.362, p = 0.017) with high heterogeneity (I2 = 86.1%, Q = 0.001).
Grand estimates obtained from SMD suggested that SBM fermentation had no effect on FCR of broiler chickens (SMD = −0.219, 95% CI: −0.501 to 0.064, p = 0.129). Restricted subgroup analysis on rearing periods also showed that FSBM did not influence FCR both in starter and finisher phase (Table 4). However, when observed on type of microorganisms used for fermentation, we found a significant decreased on FCR with either Bacillus subtilis+protease (SMD = −1.183, 95% CI: −1.799 to −0.567, p<0.001) or Bacillus subtilis alone (SMD = −1.277, 95% CI: −2.159 to −0.396, p = 0.005) with no evidence of heterogeneity from these treatments (I2 = 0.00%, Q>0.10). In contrast, treatment with probiotics containing LAB, yeast, and Bromelain protease significantly increased FCR of broiler chickens (SMD = 1.044, 95% CI: 0.436 to 1.652, p<0.001) with no heterogeneity (I2 = 0.00%, Q = 0.914).
In the meta-regression analysis, we analyzed the relationship between the Hedges’ g effect size from the outcome variables with the inclusion levels of FSBM in the diet as predictor variable considering there were wide ranges of inclusion rates of FSBM in the broiler diet (g/kg diet, Table 1). As displayed in Figure 3, the results of the meta-regression revealed that inclusion levels had no effects on final BW (p = 0.502), ADG (p = 0.588), FI (p = 0.861), and FCR (p = 0.462) (Table 5; Figure 3).
There are growing interests in utilizing fermented SBM as a partial or total substituting ingredient for SBM in the broiler diet due to their beneficial effects on broiler chickens’ performance. The present meta-analysis confirmed that fermented-SBM substituting diet can effectively improve final BW of broiler chickens. Results also emphasized that improvement of BW was more pronounced in the starter period (≤21 d of rearing period). This is in line with the fact that in the starter period, FI substantially increased in broiler chickens fed a diet containing FSBM (Table 4). More specifically, the discrepancies in the effect sizes were mainly influenced by the microorganisms used for fermentation because they have a different activity to degrade and utilize the SBM substrates thus may produce different fermentation products such as amino acids (AAs) and peptides [7].
Major explanations regarding beneficial effects of FSBM on broiler chickens were due to their effectiveness eliminating ANFs present in the SBM [6,7,12,14,17], as indicated in Table 3. Soybean meal is widely known to have a considerable amount of ANFs, particularly trypsin inhibitor, glycinin, and β-conglycinin that can impair the growth performance of broiler chickens especially in their early life [33]. This can be a logical reason to explain the increasing effect of utilizing FSBM in the starter phase of broiler chickens. Although there is less information on this, however, it could be speculated that feeding easily digestible materials with more nutritious material such as fermented SBM is beneficial to facilitate higher metabolism rate as well as to accommodate the immature digestive tract of broilers in this phase [34–36]. In addition, it was suggested that these ANFs also had negative effects on the immune status of broiler chickens. Elimination of glycinin and β-conglycinin in the diet can enhance the immune function of broiler chickens [6]. Several studies have demonstrated that most microbial strains used for fermentation were able to substantially remove ANFs. For instance, SBM fermented with B. subtilis, B. stearothermophilus, and Lactobacillus bacteria significantly reduced trypsin inhibitor by 33.6% to 100%, 74.0%, and >80%, respectively [2,7,14,37]. In addition, concentrations of glycinin, and β-conglycinin in the SBM were significantly reduced by more than 80% when fermented with B. subtilis+Enterococcus faecium and multiple strains of bacteria and yeast [6,12].
In addition, fermentation also significantly increased protein, peptide molecules, and AAs of SBM [6,7,12,13]. These nutritional improvements were associated with increasing the bioavailability of nutrients and further promoted better nutrients digestion and utilization. In regard with our findings, [9] reported that while increasing final BW, DM, CP, energy, and phosphorus digestibility were also increased. There was also evidence that supplementing FSBM in the broiler diet enhanced digestive enzyme activity [22], that may be a factor in increasing nutrient digestibility. Additionally, since small molecule peptides were produced during fermentation, it could also promote a higher nutrient absorption because small peptides can be absorbed directly and they do not require hydrolysis [38]. Furthermore, it is reasonable that the production of antimicrobial peptides created a suitable condition for intestinal flora while modulating microbial composition and improving gut integrity. Accordingly, studies reported that FSBM inclusion suppressed salmonella pathogens colonization and improved immune status and intestinal morphology [6,15,22]. In particular, Li et al [6] and Soumeh et al [11] demonstrated a reducing effect on the relative abundance of undesirable bacteria belonging to the phylum of Proteobacteria either with partial or total FSBM replacing SBM in broiler diets. In their study, Li et al [6] also reported increasing microbial richness as indicated by OS and Chao percentage in which Firmicutes phylum was predominantly presented. Abundance of Firmicutes is a good indicator for increasing non-starch carbohydrates degrading enzymes activities and it was positively associated with improvement of animal performances [39,40].
In addition to our findings, we found that strains of microorganisms performed different effects on the outcome variables including BW, ADG, FI, and FCR. In case of fermented SBM in broiler chickens, there is no evidence comparing the efficacy of microbial strains on SBM fermented products in a single study so far. However, to our knowledge, there is evidence that microbial strains have specific activity in utilizing substrates [7]. It can be explained by previous studies reporting the ability of different types of microbes in degrading ANFs and elevating nutrient compositions of FSBM [2,12–14,17]. As indicated in our results, SBM fermented with Aspergillus oryzae consistently increased BW, ADG, and FI of broiler chickens but had no effect on FCR. Meanwhile, Bacillus subtilis and Bacillus subtilis+protease were effective to reduce FCR although the sample sizes with these groups were small. The main factors that cause these effects were due to an increase in DM, CP, and energy retention and a decreased of indigestible fractions (ANFs) after fermentation [2,10].
Interestingly, among the large variability of FSBM inclusion levels in the diet, there was no evidence of relationship with all the outcome variables. This finding was in agreement with previous studies either with minimum FSBM use of 30 g/kg diet or with maximum inclusion of 150 g/kg diet [7,13]. This finding indicated that using low levels of FSBM aiming to improve broiler performance was sufficient. Other possible explanations may relate to the presence free of amino acids in the diet. When used in an excessive amount, free amino acids produced from fermentation of SBM concurrently increased. Since absorption of free amino acids requires higher energy than that of oligopeptides [41], increasing levels of FSBM in the diet might not contribute to further improvement in broiler chickens’ performance.
This study suggests that broiler feed containing fermented SBM is beneficial to improve growth performance of broiler chickens, especially when it is substituted in the diet of starter period. The effects of FSBM on ADG, FI, and FCR are apparently depended on microbial strains used for fermentation. The meta-regression suggests that increasing the inclusion of FSBM did not affect broiler chickens’ performance, thus it may be used either partially or entirely to substitute the conventional SBM.
Notes
SUPPLEMENTARY MATERIAL
Supplementary file is available from: https://doi.org/10.5713/ab.21.0546
Supplementary Figure S1. Forest plot of subgroup meta-analysis of the effect of fermented-SBM on final body weight of broiler chickens based on rearing periods
Supplementary Figure S2. Forest plot of subgroup meta-analysis based on inoculant used for fermentation of fermented-SBM on final body weight of broiler chickens
Supplementary Figure S3. Forest plot of subgroup meta-analysis of the effect of fermented-SBM on average daily gain of broiler chickens based on rearing periods
Supplementary Figure S4. Forest plot of subgroup meta-analysis based on inoculant used for fermentation of fermented-SBM on average daily gain of broiler chickens
Supplementary Figure S5. Forest plot of subgroup meta-analysis of the effect of fermented-SBM on feed intake of broiler chickens based on rearing periods
Supplementary Figure S6. Forest plot of subgroup meta-analysis based on inoculant used for fermentation of fermented-SBM on feed intake of broiler chickens
Supplementary Figure S7. Forest plot of subgroup meta-analysis of the effect of fermented-SBM on feed conversion ratio of broiler chickens based on rearing periods
Supplementary Figure S8. Forest plot of subgroup meta-analysis based on inoculant used for fermentation of fermented-SBM on feed conversion ratio of broiler chickens
ab-21-0546-suppl.pdf
Figure 2
Funnel plots analysis on (a) body weight, (b) average daily gain, (c) feed intake, and (d) feed conversion ratio to detect publication bias between-study.
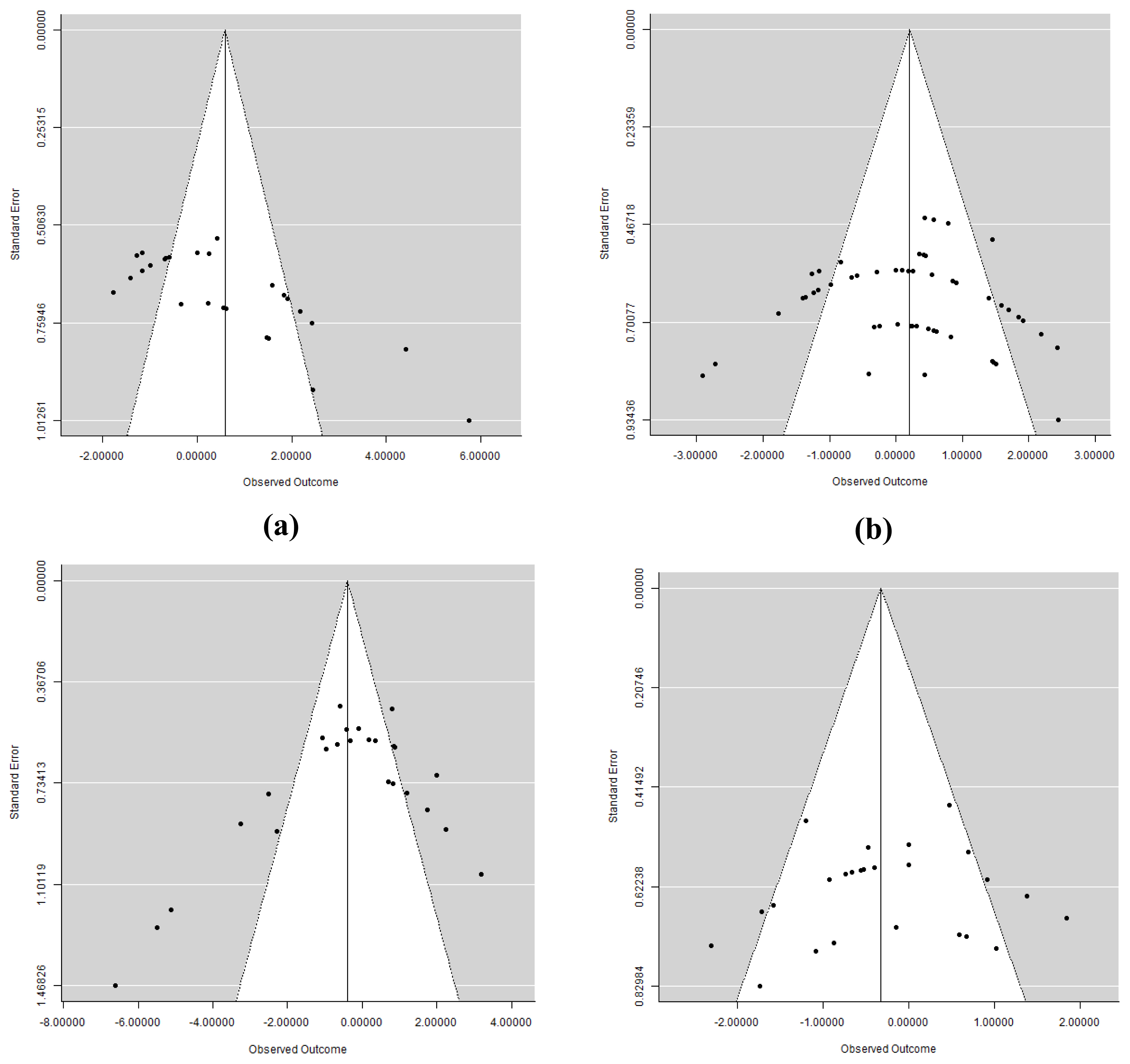
Figure 3
Meta-regression analysis to evaluate the relationship between fermented soybean meal (FSBM) inclusion levels in the broiler diet as predictor variable with outcome variables (a) body weight, (b) average daily gain, (c) feed intake, and (d) feed conversion ratio.
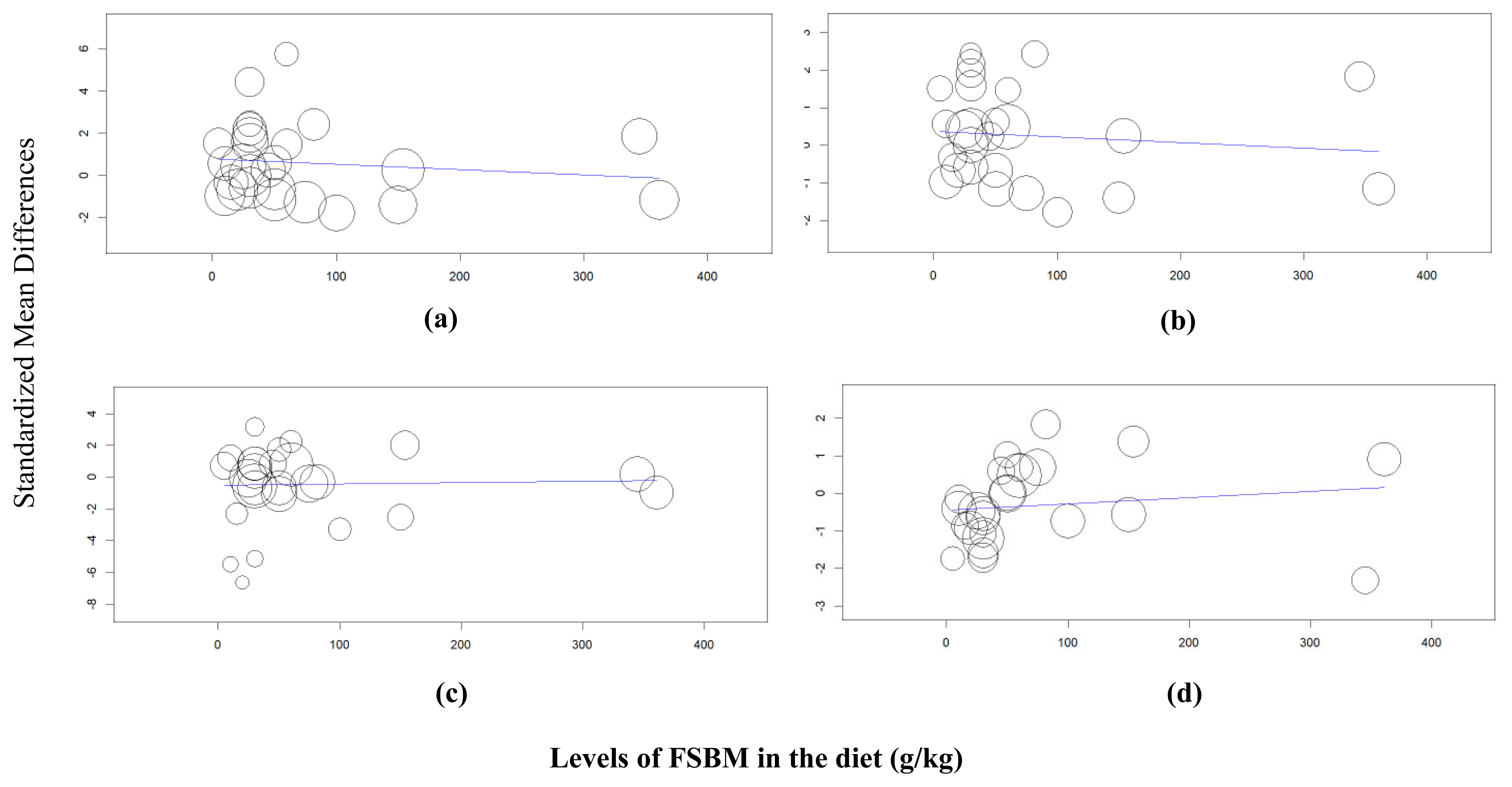
Table 1
Description of the studies included in the database
| No | Reference | Strain | Sex | Type of diet | Period (d) | Microbial starter | Fermentation time | Levels (g/kg diet) |
|---|---|---|---|---|---|---|---|---|
| 1 | [8] | Vencob | Mixed | Corn-SBM | 42 | Aspergillus niger | 48 h | 0–15 |
| 2 | [22] | Ross x Ross | Male | Corn-SBM | 42 | Aspergillus oryzae | 48 h | 0–295 |
| 3 | [9] | Ross 308 | Mixed | Corn-SBM | 42 | Aspergillus oryzae | 0–30 | |
| 4 | [2] | Cobb 500 | Female | Corn-SBM | 21 | Bacillus subtilis and protease | 24 h | 0–150 |
| 5 | [13] | Arbor Acres | n/a | Corn-SBM | 42 | Bacillus Subtilis, Aspergillus niger, and Saccharomyces cerevisiae | n/a | 0–30 |
| 6 | [16] | Arbor Acres | Mixed | Corn-SBM | 35 | Mixed probiotics fermented solution and bromelain | 36 h | 0–45 |
| 7 | [10] | Ross 308 | Male | Corn-SBM | 35 | Bacillus subtilis | n/a | 0–30 |
| 8 | [11] | Ross 308 | Male | Corn-SBM | 42 | Lactobacillus acidophilus, Lactobacillus plantarum, Bacillus subtilis, and Aspergillus oryzae | 7 d | 0–345 |
| 9 | [15] | Ross 308 | Male | Corn-SBM | 24 | Lactobacillus acidophilus, Lactobacillus plantarum, Bacillus subtilis, and Aspergillus oryzae | 7 d | 0–361 |
| 10 | [23] | Ross 308 | Female | Corn-SBM | 21 | Bacillus subtilis and protease | 24 | 0–100 |
| 11 | [14] | Ross 308 | n/a | Corn + Wheat and SBM | 40 | Lactobacillus (unspecific) | n/a | 0–60 |
| 12 | [17] | Ross 308 | Mixed | Corn-SBM | 42 | Bacillus subtilis, Lactobacillus spp., and yeasts | n/a | 0–75 |
| 13 | [6] | Cobb 500 | Male | Corn-SBM | 36 | Bacillus amyloliquefaciens, Lactobacillus acidophilus, and Saccharomyces cerevisiae | 24 h | 0–154 |
| 14 | [7] | Arbor Acres | Mixed | Corn-SBM | 42 | Bacillus stearothermophilus | 48 h | 0–150 |
| 15 | [24] | Ross 308 | Mixed | Corn-SBM | 35 | Bacillus velezensis Lactobacillus brevis | 36 h | 0–60 |
| 16 | [25] | Indian river | Mixed | Corn-SBM | 35 | Saccharomyces cerevisiae | 72 h | 0–40 |
Table 2
Descriptive statistics of nutrient specifications of the diets used in the meta-analysis
Table 3
Descriptive statistics to compare chemical composition profile between soybean meal and fermented soybean meal of the studies included in the meta-analysis
Table 4
The effect size summary of meta-analysis and sub-group analyses
Table 5
Meta-regression analysis for moderator variable that influenced the effect of fermented soybean meal inclusion on broiler chickens’ performance (standardized mean differences)
REFERENCES
1. Ravindran V, Abdollahi MR, Bootwalla SM. Nutrient analysis, metabolizable energy, and digestible amino acids of soybean meals of different origins for broilers. Poult Sci 2014; 93:2567–77.
https://doi.org/10.3382/ps.2014-04068


2. Wang JP, Liu N, Song MY, Qin CL, Ma CS. Effect of enzymolytic soybean meal on growth performance, nutrient digestibility and immune function of growing broilers. Anim Feed Sci Technol 2011; 169:224–9.
https://doi.org/10.1016/j.anifeedsci.2011.06.012

3. Qaisrani SN, Van Krimpen MM, Kwakkel RP, Verstegen MWA, Hendriks WH. Dietary factors affecting hindgut protein fermentation in broilers: a review. Worlds Poult Sci J 2015; 71:139–60.
https://doi.org/10.1017/S0043933915000124

4. Bryan DDLS, Abbott DA, Classen HL. The influence of indigestible protein on the performance and meat quality of broilers vaccinated for coccidiosis. Poult Sci 2019; 98:4815–28.
https://doi.org/10.3382/ps/pez216


5. Rodgers NJ, Swick RA, Geier MS, Moore RJ, Choct M, Wu SB. A multifactorial analysis of the extent to which eimeria and fishmeal predispose broiler chickens to necrotic enteritis. Avian Dis 2015; 59:38–45.
https://doi.org/10.1637/10774-011614-Reg.1


6. Li Y, Guo B, Wu Z, et al. Effects of fermented soybean meal supplementation on the growth performance and cecal microbiota. Animals 2020; 10:1098
https://doi.org/10.3390/ani10061098



7. Wu P, Golly MK, Guo Y, et al. Effect of partial replacement of soybean meal with high-temperature fermented soybean meal in antibiotic-growth-promoter-free diets on growth performance, organ weights, serum indexes, intestinal flora and histomorphology of broiler chickens. Anim Feed Sci Technol 2020; 269:114616
https://doi.org/10.1016/j.anifeedsci.2020.114616

8. Mathivanan R, Selvaraj P, Nanjappan K. Feeding of fermented soybean meal on broiler performance. Int J Poult Sci 2006; 5:868–72.
https://doi.org/10.3923/ijps.2006.868.872

9. Feng J, Liu X, Xu ZR, Liu YY, Lu YP. Effects of Aspergillus oryzae 3.042 fermented soybean meal on growth performance and plasma biochemical parameters in broilers. Anim Feed Sci Technol 2007; 134:235–42.
https://doi.org/10.1016/j.anifeedsci.2006.08.018

10. Kim SK, Kim TH, Lee SK, et al. The use of fermented soybean meals during early phase affects subsequent growth and physiological response in broiler chicks. Asian-Australas J Anim Sci 2016; 29:1287–93.
https://doi.org/10.5713/ajas.15.0653



11. Soumeh EA, Mohebodini H, Toghyani M, Shabani A, Ashayerizadeh A, Jazi V. Synergistic effects of fermented soybean meal and mannan-oligosaccharide on growth performance, digestive functions, and hepatic gene expression in broiler chickens. Poult Sci 2019; 98:6797–807.
https://doi.org/10.3382/ps/pez409



12. Shi C, Zhang Y, Lu Z, Wang Y. Solid-state fermentation of corn-soybean meal mixed feed with Bacillus subtilis and Enterococcus faecium for degrading antinutritional factors and enhancing nutritional value. J Anim Sci Biotechnol 2017; 8:50
https://doi.org/10.1186/s40104-017-0184-2



13. Wang LC, Wen C, Jiang ZY, Zhou YM. Evaluation of the partial replacement of high-protein feedstuff with fermented soybean meal in broiler diets. J Appl Poult Res 2012; 21:849–55.
https://doi.org/10.3382/japr.2012-00563

14. Chachaj R, Sembratowicz I, Krauze M, Ognik K. The effect of partial replacement of soybean meal with fermented soybean meal on chicken performance and immune status. J Anim Feed Sci 2019; 28:263–71.
https://doi.org/10.22358/jafs/110777/2019

15. Jazi V, Mohebodini H, Ashayerizadeh A, Shabani A, Barekatain R. Fermented soybean meal ameliorates Salmonella Typhimurium infection in young broiler chickens. Poult Sci 2019; 98:5648–60.
https://doi.org/10.3382/ps/pez338


16. Li CY, Lu JJ, Wu CP, Lien TF. Effects of probiotics and bremelain fermented soybean meal replacing fish meal on growth performance, nutrient retention and carcass traits of broilers. Livest Sci 2014; 163:94–101.
https://doi.org/10.1016/j.livsci.2014.02.005

17. Guo S, Zhang Y, Cheng Q, et al. Partial substitution of fermented soybean meal for soybean meal influences the carcass traits and meat quality of broiler chickens. Animals 2020; 10:225
https://doi.org/10.3390/ani10020225



18. Sauvant D, Schmidely P, Daudin JJ, St-Pierre NR. Meta-analyses of experimental data in animal nutrition. Animal 2008; 2:1203–14.
https://doi.org/10.1017/s1751731108002280


19. Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med 2009; 6:e1000100
https://doi.org/10.1371/journal.pmed.1000100



20. Higgins JPT, Altman DG, Gøtzsche PC, et al. The cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011; 343:d5928
https://doi.org/10.1136/bmj.d5928



21. Xu B, Li Z, Wang C, et al. Effects of fermented feed supplementation on pig growth performance: a meta-analysis. Anim Feed Sci Technol 2020; 259:114315
https://doi.org/10.1016/j.anifeedsci.2019.114315

22. Feng J, Liu X, Xu ZR, Wang YZ, Liu JX. Effects of fermented soybean meal on digestive enzyme activities and intestinal morphology in broilers. Poult Sci 2007; 86:1149–54.
https://doi.org/10.1093/ps/86.6.1149


23. Cheng YH, Hsiao FSH, Wen CM, Wu CY, Dybus A, Yu YH. Mixed fermentation of soybean meal by protease and probiotics and its effects on the growth performance and immune response in broilers. J Appl Anim Res 2019; 47:339–48.
https://doi.org/10.1080/09712119.2019.1637344

24. Tsai CF, Lin LJ, Wang CH, Tsai CS, Chang SC, Lee TT. Assessment of intestinal immunity and permeability of broilers on partial replacement diets of two-stage fermented soybean meal by bacillus velezensis and lactobacillus brevis ATCC 367. Animals 2021; 11:2336
https://doi.org/10.3390/ani11082336



25. Premathilaka KT, Nawarathne SR, Nambapana MN, et al. Partial or complete replacement of fishmeal with fermented soybean meal on growth performance, fecal composition, and meat quality in broilers. J Anim Sci Technol 2020; 62:824–39.
https://doi.org/10.5187/jast.2020.62.6.824



26. Harrer M, Cuijpers P, Furukawa TA, Ebert DD. Doing meta-analysis in R: a hands-on guide. London, UK: Chapman & Hall/CRC Press; 2019.
27. Galkanda-Arachchige HSC, Wilson AE, Davis DA. Success of fishmeal replacement through poultry by-product meal in aquaculture feed formulations: a meta-analysis. Rev Aquac 2020; 12:1624–36.
https://doi.org/10.1111/raq.12401

28. Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003; 327:557
https://doi.org/10.1136/bmj.327.7414.557



29. Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997; 315:629
https://doi.org/10.1136/bmj.315.7109.629



30. Nakagawa S, Noble DWA, Senior AM, Lagisz M. Meta-evaluation of meta-analysis: Ten appraisal questions for biologists. BMC Biol 2017; 15:18
https://doi.org/10.1186/s12915-017-0357-7



31. National Research Council. Nutrient requirements of poultry. 9th rev. edWashington, DC, USA: National Academy Press; 1994.
32. Wang JP, Liu N, Song MY, Qin CL, Ma CS. Effect of enzymolytic soybean meal on growth performance, nutrient digestibility and immune function of growing broilers. Anim Feed Sci Technol 2011; 169:224–9.
https://doi.org/10.1016/j.anifeedsci.2011.06.012

33. Pacheco WJ, Stark CR, Ferket PR, Brake J. Effects of trypsin inhibitor and particle size of expeller-extracted soybean meal on broiler live performance and weight of gizzard and pancreas. Poult Sci 2014; 93:2245–52.
https://doi.org/10.3382/ps.2014-03986


34. Barekatain MR, Swick RA. Composition of more specialised pre-starter and starter diets for young broiler chickens: a review. Anim Prod Sci 2016; 56:1239–47.
https://doi.org/10.1071/AN15333

35. Noy Y, Uni Z. Early nutritional strategies. Worlds Poult Sci J 2010; 66:639–46.
https://doi.org/10.1017/S0043933910000620

36. Leeson S. Predictions for commercial poultry nutrition. J Appl Poult Res 2008; 17:315–22.
https://doi.org/10.3382/japr.2007-00101

37. Bi H, Zhao H, Lu F, Zhang C, Bie X, Lu Z. Improvement of the Nutritional Quality and Fibrinolytic Enzyme Activity of Soybean Meal by Fermentation of Bacillus subtilis. J Food Process Preserv 2015; 39:1235–42.
https://doi.org/10.1111/jfpp.12340

38. Zambonino Infante JL, Cahu CL, Peres A. Partial substitution of di- and tripeptides for native proteins in sea bass diet improves Dicentrarchus labrax larval development. J Nutr 1997; 127:608–14.
https://doi.org/10.1093/jn/127.4.608


39. Crisol-Martínez E, Stanley D, Geier MS, Hughes RJ, Moore RJ. Understanding the mechanisms of zinc bacitracin and avilamycin on animal production: linking gut microbiota and growth performance in chickens. Appl Microbiol Biotechnol 2017; 101:4547–59.
https://doi.org/10.1007/s00253-017-8193-9


40. Singh KM, Shah T, Deshpande S, et al. High through put 16S rRNA gene-based pyrosequencing analysis of the fecal microbiota of high FCR and low FCR broiler growers. Mol Biol Rep 2012; 39:10595–602.
https://doi.org/10.1007/s11033-012-1947-7


41. Ali Tauqir N. Absorption and transportation of amino acids in animals: a review. J Environ Agric Sci 2016; 9:96–109.
- TOOLS
-
METRICS

- Related articles
-
Effects of wilting on silage quality: a meta-analysis2024 July;37(7)







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Supplement
Supplement Print
Print




