 |
 |
| Anim Biosci > Volume 37(2); 2024 > Article |
|
Abstract
Objective
Pork is an important source of animal protein in many countries. Subtle physiochemical changes occur during pork postmortem aging. The changes of apoptosis and autophagy in pork at 6 h to 72 h after slaughter were studied to provide evidence for pork quality.
Methods
In this article, morphological changes of postmortem pork was observed through Hematoxylin-eosin staining, apoptotic nuclei were observed by TdT-mediated dUTP nick end labeling assay, protein related to apoptosis and autophagy expressions were tested by western blot and LC3 level were expressed according to immunofluorescence assay.
Results
In this study, we found the occurrence of apoptosis in postmortem pork, and the process was characterized by nucleus condensation and fragmentation, formation of apoptotic bodies, increase in apoptosis-related Bax/Bcl-2 levels, and activation of caspases. Autophagy reached its peak between 24 and 48 h after slaughter, accompanied by the formation of autophagosomes on the cell membrane and expression of autophagy-related proteins beclin-1, P62, LC3-I, LC3-II, and ATG5.
Conclusion
Obvious apoptosis was observed at 12 h and autophagy reached its peak at 48 h. The present work provides the evidence for the occurrence of apoptosis and autophagy during postmortem aging of pork. In conclusion, the apoptosis and autophagy of muscle cells discovered in this study have important implications for pork in the meat industry.
Pork is one of the important sources of animal protein in many countries with important nutritional and economic significance [1]. There is a growing demand for high-quality pork with desirable color, tenderness and flavor. Studies suggested that complex physiological and biochemical reactions during postmortem aging may play an important role in meat quality [2]. Transportation stress, storage temperature, slaughter methods, and nitric oxide application can affect glycolysis. Nitric oxide could promote the activation of AMP-activated protein kinase, thus accelerating glycolysis [3]. Pork refrigeration conditions and aging time have an impact on the tenderness and flavor of pork, which will directly affect consumersŌĆÖ desire to buy. In addition, changes in muscle energy metabolism and physiology processes, such as apoptosis and autophagy during postmortem aging play a key role in the regulation of meat quality [4]. However, the mechanism and regulation of such changes are still unclear.
After the pigs are slaughtered, the muscles are in a tense state, irreversible contraction occurs in the muscle fibers and lactic acid is produced in the carcass, When the meat reaches a certain state, the internal protein degrades into molecular peptides and amino acids, and the taste and tenderness of the muscles improves, which is the maturation process of meat. Apoptosis and autophagy affect the maturation process of meat. Apoptosis refers to autonomous and highly organized programmed cell death and plays a role in homeostasis [5]. It can be induced by intracellular mitochondrial signals or extracellular death receptors [6]. Hypoxia or the ischemia of muscle cells may increase the rate of anaerobic metabolism, decrease cellular ATP and pH levels, and induce calcium release. These factors are involved in endogenous mitochondrial or endoplasmic reticulum-related cell death signaling pathways and induce muscle cell apoptosis during postmortem aging [7,8]. In the process of pork sale, consumers are particularly concerned about the of pork. Postmortem tenderization improves meat quality and tenderness is one of the most important factors for consumers to evaluate the quality of pork [9]. Apoptosis plays a major role in the postmortem tenderization of meat after death [10]. Caspases act as the key effector molecules of apoptosis. Changes in caspase activity could affect muscle quality during pork aging [11]. The Caspases family such as Caspase-3 and Caspase-6 are involved in the hydrolysis of muscle fibrin after slaughter, and their activity is positively correlated with the tenderness of meat. Evans et al [12] proposed that the occurrence of cell apoptosis triggered an imbalance between the rate of muscle protein synthesis and degradation. Therefore, the molecular regulation mechanism of apoptosis is related to the quality of pork after slaughter.
In apoptosis, characteristic morphological changes such as cell shrinkage, chromatin condensation, apoptotic body formation can be observed. Autophagy is also involved in the maturation process of meat after slaughter [13]. Studies had proved that autophagy affects the activity of caspases enzyme and indirectly affects the tenderness of meat. Autophagy can also activate the enzyme activity of apoptosis and regulate the mechanism of apoptosis [14]. Therefore, the study of postmortem autophagy is of significance to the quality of pork, but research on autophagy is relatively scarce. Besides, autophagy is initiated in cells in response to nutrient deficiency, metabolic imbalance, and oxidative stress [15]. During postmortem aging, oxidative stress produced by reactive oxygen species is related to the occurrence of skeletal muscle cell apoptosis, autophagy, and necrosis [16,17]. At the end of autophagy, morphological changes, including an increase in number, and enlargement, gray-white components inside autophagosomes, and minor nuclear pyknosis can be observed [18]. Regarding the autophagy-related genes, as the homologous gene of Atg6, beclin-1 is the first specific gene that mediates autophagy in mammalian cells. The levels of apoptosis and autophagy in pork tissues change with time even in low temperature environment, so we evaluated the quality of pork by measuring the changes of apoptosis and autophagy in different shelf-life periods. Our present study investigated the occurrence and mechanism of muscle cell apoptosis and autophagy during postmortem pork aging and further explored the mechanism of postmortem pork aging, so as to provide theoretical reference for the development of pork in food industry.
The 6-month-old crossbred boars with live weight of 100 ┬▒10 kg were acquired from Xiangtai Food Co., LTD (Yangzhou, Jiangsu, China). The implementation of the slaughter process was based on the Operating Procedures of Livestock and Poultry Slaughtering-Pig (GB/T 17236ŌĆō2019). Briefly, crossbred boars without feeding for 12 hours before slaughter, the longissimus thoracis muscles were taken and refrigerated at 0┬░C to 4┬░C for maturation. The related indexes were detected at 6, 12, 24, 48, and 72 h of postmortem maturation. Part of meat samples were rapidly placed in liquid nitrogen and then frozen at ŌłÆ80┬░C for biochemical analysis while other samples were fixed with 1% paraformaldehyde for morphological observation. Animal studies were approved by the Jiangsu Administrative Committee for Laboratory Animals, the approval number of IACUC is A12070201.
The 2 cm of fresh intestine was flushed and fixed in 4% paraformaldehyde solution. The intestines were buried in paraffin and 4 to 5 ╬╝m slices were prepared. The slices placed on a slide were stained by hematoxylin-eosin (HE) for immunohistochemical analysis.
Briefly, the slices were immersed in pure xylene solution for 20 min until the paraffin was completely dissolved. Then the slices were placed in the mixed solution of xylene and anhydrous ethanol (v/v, 3/4), xylene and anhydrous ethanol (v/v, 1/2), and xylene and anhydrous ethanol (v/v, 1/4), respectively. After each reaction for 10 min, the slices were rehydrated in 100%, 95%, 90%, 80%, 70%, 60%, and 50% ethanol solutions with each reaction for 10 min. Afterwards, the slices were stained with hematoxylin for 4 min, and then washed with running water. Hydrochloric acid and alcohol solution (v/v, 1/100) was added for reaction and subsequently washed with running water. The obtained sections were dyed in eosin solution for 90 s and washed with ultra-pure water. Then 25%, 50%, 75%, and 100% xylene solution was applied in the slices. Each reaction proceeded for 10 min. Finally, the slices were mounted by neutral balsam (Beyotime, Shanghai, China) and then observed under a microscope (BX53; Olympus, Tokyo, Japan) to analyze the morphology of the nuclei. Images were acquired at 400├Ś magnification.
The TdT-mediated dUTP nick end labeling (TUNEL) assay was performed for the analysis of apoptosis. The frozen sections were rinsed twice with a phosphate buffer solution (PBS) buffer and stained according to the manufacturerŌĆÖs instructions (Servicebio, Hubei, China). The tissue slices were mounted with glycerol and observed under a super-resolution laser confocal microscope (TCS SP8 STED; Leica, Wetzla, Germany). Each sample was photographed in at least three different fields at 400├Ś magnification. Apoptotic and total nuclei were recognized according to the red fluorescence of TUNEL and blue fluorescence of 4ŌĆ▓,6-diamidino-2-phenylindole (DAPI), respectively.
The pork samples at different postmortem aging times were washed with PBS and lysed with lysis buffer (Applygen, Beijing, China) including protease and phosphatase inhibitor cocktail (New Cell Molecular Biotech, Suzhou, China) for 30 min. The mixtures were centrifuged at 12,000├Śg for 10 min at 4┬░C. The protein concentration of supernatant was determined using bovine serum albumin (BSA) kit (Thermo Fisher Scientific, Waltham, MA, USA) and adjusted to the same concentration. To prepare gel samples, protein samples were mixed with loading buffer (100 mM Tris-HCl, 5 mM ethylene diamine tetraacetic acid, 20% glycerol, 4% sodium dodecyl sulfate (SDS), 1% ╬▓-mercaptoethanol, 0.01% bromophenol blue) and then boiled at 95┬░C for 5 min. Subsequently, protein samples were separated by polyacrylamide gel electrophoresis. After electrophoresis, the proteins were transferred to polyvinylidene difluoride membrane (Bio-Rad Laboratories, Hercules, CA, USA). Subsequently, 5% BSA was used for the blocking of proteins for 2 h. The membranes were then incubated with primary antibodies overnight at 4┬░C. After washing three times by Tris buffered saline with tween-20 (TBST) for 5 min each, the membranes were incubated with secondary antibody at room temperature for 1 h. After washing again, chemiluminescence reagents (New Cell Molecular Biotech, Suzhou, China) were used to develop the protein bands. The bands were imaged by the chemiluminescence imaging analysis system (ProteinSimple, Silicon Valley, CA, USA) and then quantified using NIH Image-J software.
The primary antibodies include BCL2-associated X (Bax, ab104156), B-cell lymphoma-2, (Bcl-2, ab117115), glyceraldehyde-3-phosphate dehydrogenase (GAPDH, ab9484), Beclin-1 (ab231341), sequestosome-1 (P62, ab233207), and autophagy-related 5 (ATG5, ab228668) were acquired from abcam (Cambridge, Britain), Caspase-3 (#14220), Caspase-8 (#4790), microtuble-associated protein light chain 3-I/II (LC3-I/II, #12741) were acquired from Cell Signaling Technology (Boston, MA, USA). The secondary antibodies include Anti-rabbit immunoglobulin G (IgG), horseradish peroxidase-linked (HRP-linked, Antibody (#7074) and Anti-mouse IgG, HRP-linked Antibody (#7076) sourced from Cell Signaling Technology (USA).
The optimal cutting temperature compound adhesive coated part of the tissue was placed into liquid nitrogen, then removed and placed on the frozen microtome (CM1950; Leica, Germany). The tissues were cut to 8 ╬╝m and adsorbed to an adhesive slide, allowed to stand at room temperature for 5 min. Paraformaldehyde solution (4%) was added to the drops and fixed at room temperature for 10 to 15 min, then dried in a fume cupboard, and stored in a section box at ŌłÆ20┬░C.
The prepared frozen sections were washed with PBS and blocked in 5% bovine serum at room temperature for 1.5 h and incubated with an LC3B (-II) rabbit antibody (ab229327; abcam, China) overnight at 4┬░C. Fluorescent secondary antibodies (ab150080; abcam, China) were applied to the tissue sections in the dark for incubation at 37┬░C for 2 h. After incubation, the slices were stained with DAPI for 10 min. After fixation, the sections were observed under an ultra-high resolution laser confocal microscope (TCS SP8 STED; Leica, Germany).
All tests were carried out in triplicate. The results were represented as mean┬▒standard error and analyzed with GraphPad Prism 8.0 software. In the statistical model, storage time was the fixed variable, and muscle was considered as random variable. One-way analysis of variance was performed using SPSS 19.0 software. A p-value of <0.05 was considered statistically significant.
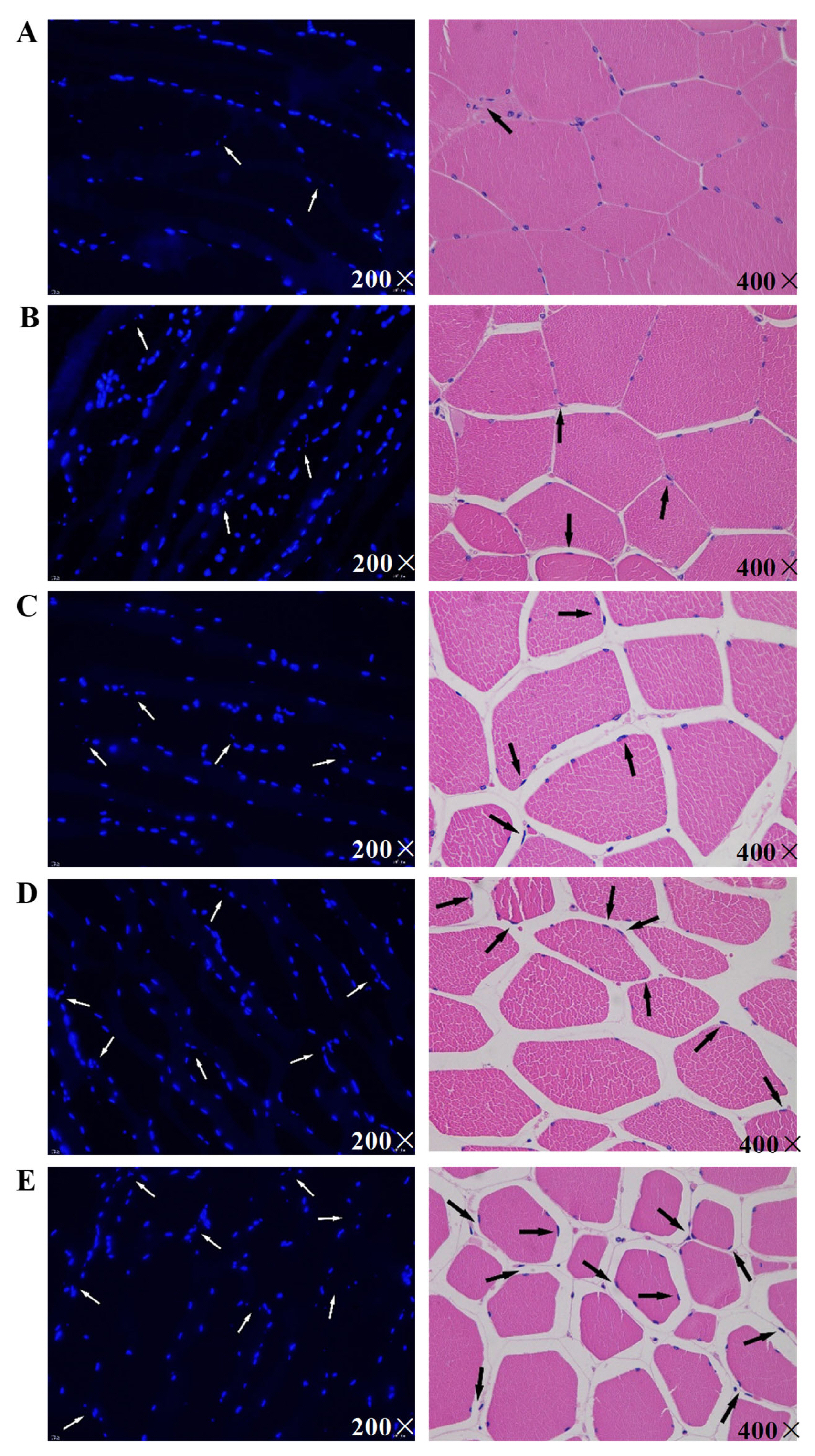
Observing the changes of nuclear morphology is an effective method for detecting cell apoptosis. Generally, when cells undergo apoptosis, cell shrinkage, chromatin condensation, and apoptotic bodies formation are detected [19]. In this study, morphological changes of postmortem pork were observed through DAPI staining (Figure 1AŌĆō1E) and HE staining (Figure 1FŌĆō1J).
The nuclei of skeletal muscle cells were stained blue by DAPI. The nuclei at 6 h postmortem was basically intact and uniform in size. The level of nuclei pyknosis and irregularity increased over time. At 12 h postmortem, the nuclear staining became brighter, and particles increased. The white arrows in Figure 1AŌĆō1E point to typical apoptotic nuclei, which gradually disintegrated and ruptured to form multiple apoptotic bodies or fragments.
Through HE staining, the nucleic structures of pork muscle cells at 6 h postmortem showed retained integrity, clear outlines, and uniform nuclear and cytoplasm distributions. However, after 6 h postmortem, the intercellular space gradually increased with the extended postmortem aging time. As indicated by the black arrows in Figure 1FŌĆō1J, at 12, 24, 48, and 72 h postmortem, some parts of the nucleus and cytoplasm gradually migrated to edge, half-moon-shaped apoptotic bodies formed, and the nuclei shrunk. These phenomena implied that apoptosis occurred in postmortem pork muscle cells. Apoptosis in pork occurred 6 to 12 h after slaughter. These results are consistent with the results of Dirks and Leeuwenburgh [20] and Ouali et al [21] who indicated that apoptosis might occur in the early stage of muscle aging.
When apoptosis occurs, chromosomal DNA is initially cleaved by endonucleases, and then two strands of DNA break to form 3ŌĆ▓-OH ends, thereby forming nucleosomal DNA multimers [22]. Through TUNEL, fluorescently labeled deoxynucleotides can covalently link to 3ŌĆ▓-OH, which has the advantages of high sensitivity and fast response, and thus being widely applied in detecting cell apoptosis [23]. Combined with DAPI staining, TUNEL accurately reflects DNA breakage and staining location in the nuclei, and the contrast between positive cells and negative cells is clearly identified. Blue-red staining in the nuclei indicated apoptotic nuclei, and normal nuclei were only stained blue by DAPI (Figure 2A). At 6 h postmortem, apoptotic nuclei appeared, and the number of apoptotic nuclei significantly increased (p<0.05). At 12 h postmortem, the number of apoptotic nuclei rapidly increased and reached its maximum. Subsequently, the number of apoptotic nuclei gradually decreased (Figure 2B). However, the results are not consistent with the apoptosis in yak meat, which showed the highest apoptotic state at 6 h after slaughter [20]. The differences between these results may attribute to the differences in the types of muscle fibers in yak meat and pork, and differences in mitochondria, cytochrome-C content, and energy. Pistilli et al [24] indicated that fiber type could affect early energy metabolism in postmortem muscles, and energy metabolism disorders are closely related to apoptosis [25].
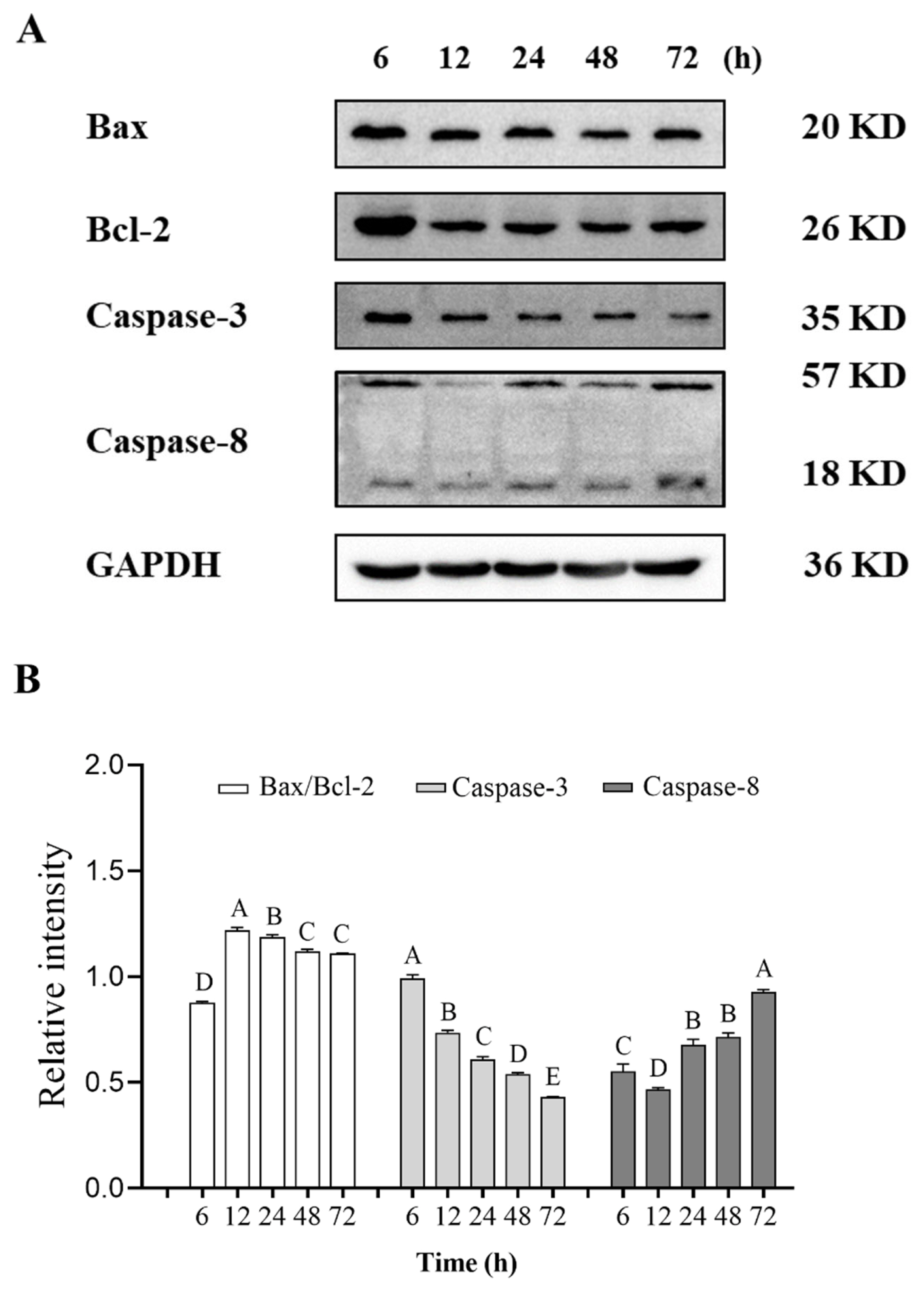
Apoptosis is closely related to the Bcl-2 family, which can regulate apoptotic processes by maintaining the balance between the level of Bcl-2, the apoptosis inhibitor, and Bax, the apoptosis promoter [26]. The relative content of Bax/Bcl-2 in pork significantly increased at 6 h and 12 h postmortem and reached the maximum value at 12 h postmortem (Figure 3). This result is consistent with that of Guo et al [27] who reported that the Bax/Bcl-2 level decreased gradually with the increasing of aging time. Manna et al. proposed that the ratio of Bax to Bcl-2 in fish was calculated and used in determining whether cells underwent apoptosis [28]. In this experiment, changes in Bax/Bcl-2 level during pork aging indicated that the apoptosis of pork muscle cells occurred after slaughter, and the highest rate of the process was observed at 12 h postmortem.
Caspase-3, one of the essential members of the caspase family, is considered a key effector molecule and an important executive protein in apoptosis [29]. Its activation marks the beginning of irreversible apoptosis. Caspase-3 was produced in pork muscle cells after slaughter, and its expression obviously decreased at 6 h postmortem (Figure 3). During postmortem aging of pork, the apoptosis signal was amplified after the release of mitochondrial cytochrome C and the formation of apoptotic bodies. Then a caspase cascade reaction was initiated, with caspase-3 being activated and then acting on the degradation of downstream proteins [30]. Zhang et al [31] reported that caspase-3 was degraded into 23 and 17 kDa fragments during postmortem beef aging. This process was considered in the present study.
Caspase-8 is involved in the initiation of apoptosis and activated caspase-8 can participate in the activation of the downstream effector caspase, such as caspase-3 and caspase-7. In postmortem pork, caspase-8 was cleaved into 18 kDa activated fragments, and the relative content generally showed a trend of first decreasing (6 to 12 h postmortem) and then increasing (12 to 72 h postmortem). Similarly, an 18 kDa fragment of caspase-8 was found in the comparative research of different pork skeletal muscles [32]. The result suggests that caspase-8 is activated under hypoxia in the early postmortem stage of pork.
LC3 is a marker for autophagosome membranes, and red fluorescence indicates the formation of autophagosomes. The results showed that at 6 and 24 h postmortem, red fluorescence appeared as puncta in pork muscle cells, and the fluorescence intensity gradually increased, suggesting that autophagosomes were formed on the cell membrane at early postmortem, and the level of autophagy was significant increased (Figure 4). However, the amount of red fluorescence spots decreased at 48 and 72 h, and then red fluorescence distributed diffusely, indicating that the level of autophagy decreased over time.
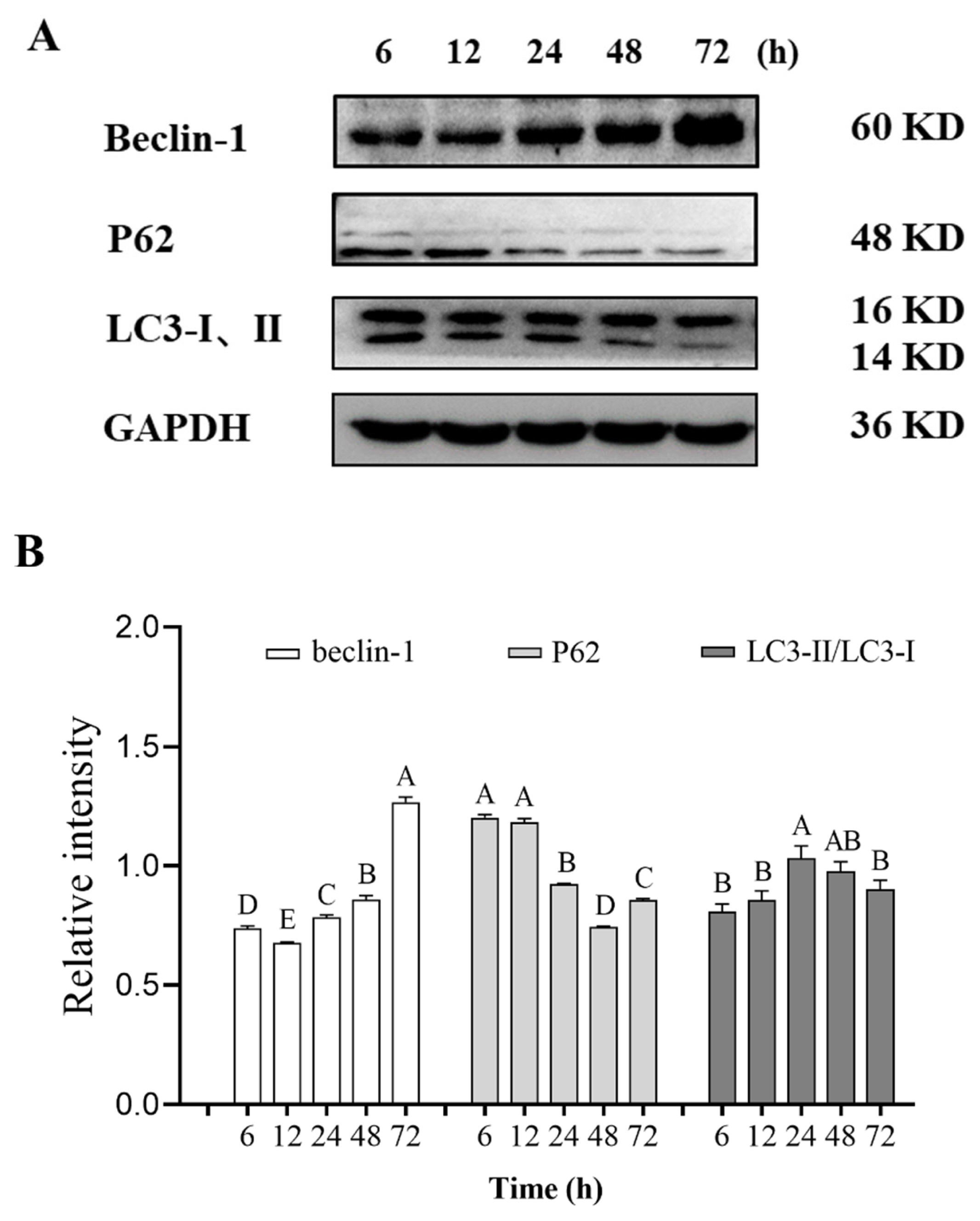
Autophagy is a multi-step process involving multiple proteins. Beclin-1 is one of the important proteins in the initial stage of autophagy, which plays a role in autophagy regulation and participates in the formation of autophagosomes [33]. At 12 and 24 h postmortem, the relative content of beclin-1 decreased and then increased compared with 6 h postmortem. When aging time was extended from 12 to 72 h after slaughter, the relative content of beclin-1 in pork showed an overall upward trend (Figure 5). These results indicate that the occurrence of autophagy proceeded gradually and persisted in the entire postmortem aging period.
Another important protein is the autophagy substrate P62 protein, which is a multi-domain ubiquitin-binding protein encoded by the recombinant sequestosome 1 (SQSTM1) gene and plays an important role in the ubiquitin-proteasome system and autophagy [34]. In autophagy, the expression of P62 gradually decreased with the enhancement of autophagy, which was usually negatively correlated with the activity of autophagy. The relative expression of P62 in this experiment showed a significant downward trend at 12 and 48 h postmortem, indicating that autophagy was enhanced during this period (Figure 5).
LC3-II is a key protein in autophagy and localized on the surface of pre-autophagic vacuoles and autophagic vesicles, which is considered a specific marker of autophagic activity [35]. When autophagy occurs, cytoplasmic LC3 (LC3-I) enzymatically degrades small segments of polypeptides into the autophagosome membrane LC3-II. The LC3-II/I ratio is usually estimated according to the level of phagocytosis. Within 72 h after slaughter, the relative content of LC3-II/LC3-I in pork increased first and then decreased. This result is consistent with the expression of LC3-II/LC3-I in Figure 5. The early increase may be inseparable from the mediating effect of beclin-1 [36]. Moreover, during autophagosome membrane formation, prophase LC3 may be cleaved into LC3-I, which binds to phosphatidylethanolamine to form LC3-II and thus prolonging autophagy [37]. Upon the formation of autophagosomes, LC3-II gradually aggregated and localized on the autophagosome membrane. When autophagosomes fuse with lysosomes and are degraded, LC3-II can be re-lipidated to LC3-I, which may be involved in the formation of autophagy [38].
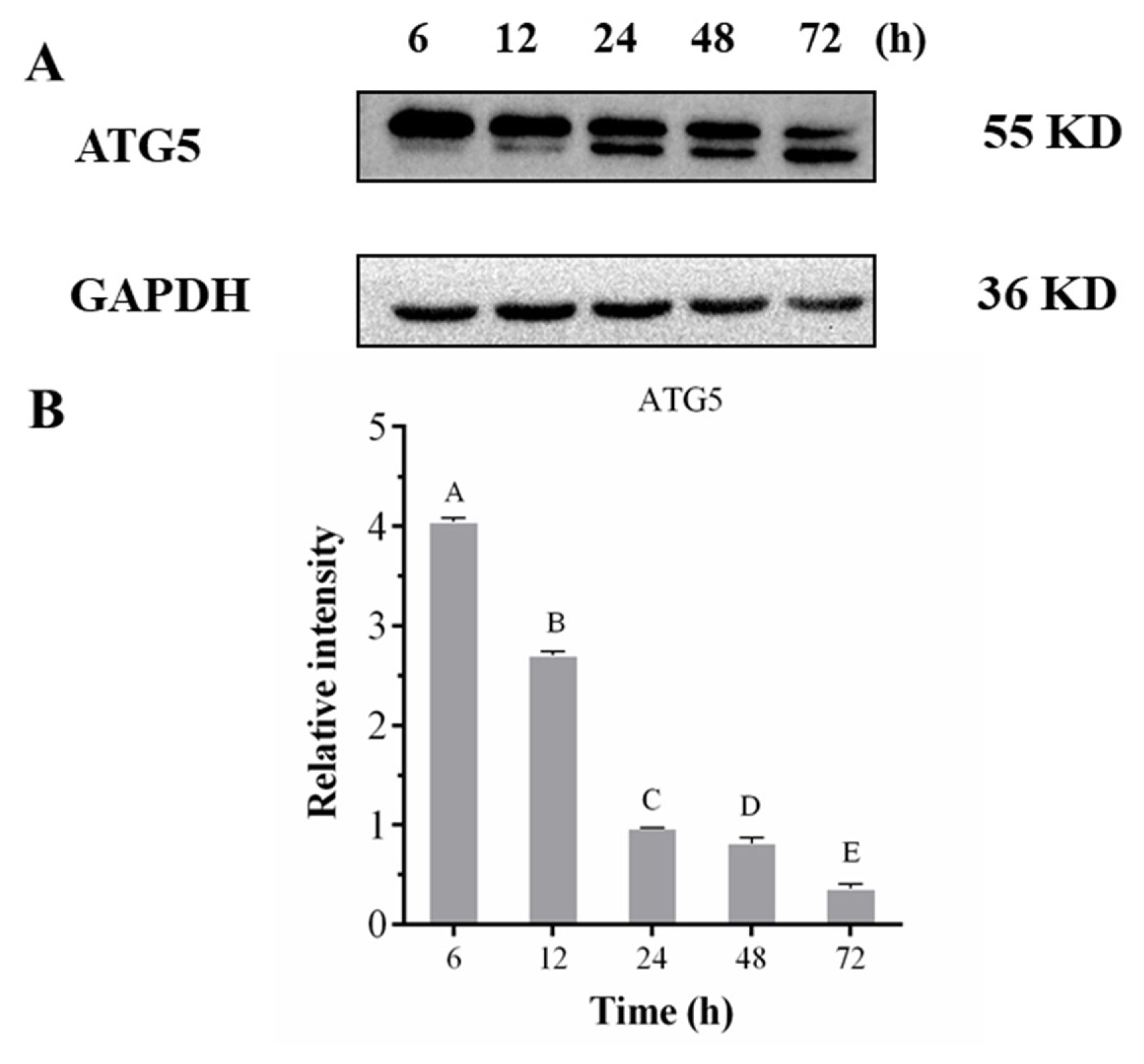
ATG5 is an essential protein for autophagosome formation. Complexes conjugated with ATG12, which can determine the bending direction of pre-autophagosome membrane, promote the extension and expansion of the membrane, and further produce esterification and activate autophagy. Besides, it plays an essential role in autophagy extension [39]. Western blot results showed that ATG5 exhibited two bands (Figure 6). One band was attributed to the endogenous level of the ATG5 total protein, and another was attributed to the conjugation of ATG5 ATG12. The relative expression of total protein showed a significant downward trend (p< 0.05) during postmortem aging, and the level of ATG5 that conjugated with ATG12 showed an obvious upward trend. The results demonstrate that autophagy occurred in postmortem pork.
The identification of pork quality is helpful to the development of the pork meat industry. Apoptosis and autophagy of pork are directly related to pork quality. A series of complex physiological and biochemical reactions occur in the muscle after death, mainly including glycolysis, muscle contraction, apoptosis, protein degradation, etc. These reactions largely determine the formation of meat quality. The improvement of pork quality can be achieved by influencing the biochemical reaction during postmortem aging [40,41]. The present work provides evidence for the occurrence of apoptosis and autophagy during postmortem aging of pork. It was characterized by the gradual disintegration and rupturing of the nucleus, formation of multiple apoptotic bodies, as well as the activation of apoptosis-related proteins. The number of apoptotic nuclei and the expression of related proteins show that apoptosis is intensive at 12 h postmortem. Subsequently, autophagy reaches its peak at 48 h after slaughter, accompanied by the gradual formation of autophagosomes on the cell membrane, and changes in the expression levels of autophagy-related proteins (beclin-1, P62, LC3-I, LC3-II, and ATG5). Previous studies have found that oxidative stress occurred, and reactive oxygen produced after slaughter, which leads to mitochondrial lipid peroxidation, changes in mitochondrial permeability and ATP levels, and caspase family activation, thus affecting the tenderness of meat. At the same time, the BCL family is also involved in muscle postmortem apoptosis, which is consistent with our studies [42]. Zhang et al [43] showed that cell membrane permeability increased, calcium ion level increased firstly and then decreased, pork muscle exhibited apoptosis in the early stage of postmortem aging and tenderness in the late stage of postmortem aging, and protein phosphorylation regulated cells apoptosis by regulating the combination of ATP and calcium ion. The apoptosis level was higher at 12 h and autophagy level was higher at 24 h. Our research is beneficial to the application of pork in practical industry and provides theoretical reference for the change of pork quality in different periods.
Notes
Figure┬Ā1
Morphological changes during pork postmortem aging through hematoxylin-eosin (HE) staining. Morphological changes were marked with arrowheads.
Figure┬Ā2
Changes of apoptosis levels during pork postmortem aging through the TdT-mediated dUTP nick end labeling (TUNEL). (A) Apoptotic and total nuclei were recognized by red fluorescence of TUNEL and blue fluorescence of DAPI, respectively. (B) The ratio of the red/blue fluorescence was used to calculate the apoptosis rate. DAPI, 4ŌĆ▓,6-diamidino-2-phenylindole. AŌĆōE Different letters indicate significant differences (p<0.05, n = 3).

Figure┬Ā3
Expression of apoptosis-related proteins including Bax, Bcl-2, caspase-3, and caspase-8 in postmortem pork during aging. (A) Representative western blot graphs of Bax, Bcl-2, caspase-3, and caspase-8. (B) The relative contents changes of Bax, Bcl-2, caspase-3, and caspase-8. AŌĆōE Different letters indicate significant differences (p<0.05, n = 3).
Figure┬Ā4
LC3 (red) level and DAPI (blue) changes during pork postmortem aging. LC3, microtuble-associated protein light chain 3; DAPI, 4ŌĆ▓,6-diamidino-2-phenylindole

REFERENCES
1. Xiao HB, Chen Q, Wang JM, et al. The puzzle of the missing meat: Food away from home and ChinaŌĆÖs meat statistics. J Integr Agric 2015; 14:1033ŌĆō44.
https://doi.org/10.1016/S2095-3119(14)60987-4

2. Lebret B, Candek-Potokar M. Review: Pork quality attributes from farm to fork. Part II. Processed pork products. Animal 2022; 16:Suppl 1100383
https://doi.org/10.1016/j.animal.2021.100383


3. Shen QW, Means WJ, Thompson SA, et al. Pre-slaughter transport, AMP-activated protein kinase, glycolysis, and quality of pork loin. Meat Sci 2006; 74:388ŌĆō95.
https://doi.org/10.1016/j.meatsci.2006.04.007


4. Suman SP, Joseph P. Myoglobin chemistry and meat color. Annu Rev Food Sci Technol 2013; 4:79ŌĆō99.
https://doi.org/10.1146/annurev-food-030212-182623


5. Ueda S, Nakamura H, Masutani H, et al. Redox regulation of caspase-3 (-like) protease activity: regulatory roles of thioredoxin and cytochrome c. J Immunol 1998; 161:6689ŌĆō95.
https://doi.org/10.4049/jimmunol.161.12.6689


6. Tosa N, Murakami M, Jia WY, et al. Critical function of T cell death-associated gene 8 in glucocorticoid-induced thymocyte apoptosis. Int Immunol 2003; 15:741ŌĆō9.
https://doi.org/10.1093/intimm/dxg070


7. Becila S, Herrera-Mendez CH, Coulis G, et al. Postmortem muscle cells die through apoptosis. Eur Food Res Technol 2010; 231:485ŌĆō93.
https://doi.org/10.1007/s00217-010-1296-5

8. Herrera-Mendez CH, Becila S, Boudjellal A, Ouali A. Meat ageing: Reconsideration of the current concept. Trends Food Sci Technol 2006; 17:394ŌĆō405.
https://doi.org/10.1016/j.tifs.2006.01.011

9. Zhang JY, Ma JY, Guo ZB, et al. Study on the apoptosis mediated by apoptosis-inducing-factor and influencing factors of bovine muscle during postmortem aging. Food Chem 2018; 266:359ŌĆō67.
https://doi.org/10.1016/j.foodchem.2018.06.032


10. Chen L, Feng XC, Lu F, et al. Effects of camptothecin, etoposide and Ca2+ on caspase-3 activity and myofibrillar disruption of chicken during postmortem ageing. Meat Sci 2011; 87:165ŌĆō74.
https://doi.org/10.1016/j.meatsci.2010.10.002


11. Kemp CM, Bardsley RG, Parr T. Changes in caspase activity during the postmortem conditioning period and its relationship to shear force in porcine longissimus muscle. J Anim Sci 2006; 84:2841ŌĆō6.
https://doi.org/10.2527/jas.2006-163


12. Evans WJ. Skeletal muscle loss: cachexia, sarcopenia, and inactivity. Am J Clin Nutr 2010; 91:1123SŌĆō7S.
https://doi.org/10.3945/ajcn.2010.28608A


13. Lana A, Zolla L. Apoptosis or autophagy, that is the question: Two ways for muscle sacrifice towards meat. Trends Food Sci Technol 2015; 46:231ŌĆō41.
https://doi.org/10.1016/j.tifs.2015.10.001

14. Wang XY, Zhang YM, Liang RR, et al. Advances in understanding the regulation of meat tenderness by peroxiredoxin 6. Food Sci 2021; 42:236ŌĆō43. (in Chinese) https://doi.org/10.7506/spkx1002-6630-20200427-355

15. Levine B, Yuan J. Autophagy in cell death: an innocent convict? J Clin Invest 2005; 115:2679ŌĆō88.
https://doi.org/10.1172/JCI26390



16. Kraft C, Martens S. Mechanisms and regulation of autophagosome formation. Current Opinion in Cell Biology 2012; 24:496ŌĆō501.
https://doi.org/10.1016/j.ceb.2012.05.001


17. Lana A, Zolla L. Apoptosis or autophagy, that is the question: Two ways for muscle sacrifice towards meat. Trends Food Sci Technol 2015; 46:231ŌĆō41.
https://doi.org/10.1016/j.tifs.2015.10.001

18. Gonz├Īlez-Polo RA, Niso-Santano M, Ort├Łz-Ort├Łz MA, et al. Inhibition of paraquat-induced autophagy accelerates the apoptotic cell death in neuroblastoma SH-SY5Y cells. Toxicol Sci 2007; 97:448ŌĆō58.
https://doi.org/10.1093/toxsci/kfm040


19. Hacker G. The morphology of apoptosis. Cell Tissue Res 2000; 301:5ŌĆō17.
https://doi.org/10.1007/s004410000193


20. Dirks A, Leeuwenburgh C. Apoptosis in skeletal muscle with aging. Am J Physiol Regul Integr Comp Physiol 2002; 282:R519ŌĆō27.
https://doi.org/10.1152/ajpregu.00458.2001


21. Ouali A, Herrera-Mendez CH, Coulis G, et al. Revisiting the conversion of muscle into meat and the underlying mechanisms. Meat Sci 2006; 74:44ŌĆō58.
https://doi.org/10.1016/j.meatsci.2006.05.010


22. Darzynkiewicz Z, Galkowski D, Zhao H. Analysis of apoptosis by cytometry using TUNEL assay. Methods 2008; 44:250ŌĆō4.
https://doi.org/10.1016/j.ymeth.2007.11.008



23. Luo CL, Chen XP, Ni H, et al. Comparison of labeling methods and time course of traumatic brain injury-induced cell death in mice. Neural Regen Res 2010; 5:706ŌĆō9.
https://doi.org/10.3969/j.issn.1673-5374.2010.09.012

24. Pistilli EE, Jackson JR, Alway SE. Death receptor-associated pro-apoptotic signaling in aged skeletal muscle. Apoptosis 2006; 11:2115ŌĆō26.
https://doi.org/10.1007/s10495-006-0194-6



25. Zhu XD, Zhang Y, Wang YQ, et al. Agrimoniin sensitizes pancreatic cancer to apoptosis through ROS-mediated energy metabolism dysfunction. Phytomedicine 2022; 96:153807
https://doi.org/10.1016/j.phymed.2021.153807


26. Zhao QF, Yuan XM, Xia H, Wu Q, Jiang KT. Pristimerin inhibits growth and induces apoptosis of gastric cancer MGC803 and SGC7901 cells. World Chinese J Digestol 2016; 24:1064ŌĆō9.
https://doi.org/10.11569/wcjd.v24.i7.1064

27. Guo B, Zhang W, Tume RK, et al. Disorder of endoplasmic reticulum calcium channel components is associated with the increased apoptotic potential in pale, soft, exudative pork. Meat Sci 2016; 115:34ŌĆō40.
https://doi.org/10.1016/j.meatsci.2016.01.003


28. Manna S, Chakraborty T, Ghosh B, et al. Dietary fish oil associated with increased apoptosis and modulated expression of Bax and Bcl-2 during 7,12-dimethylbenz(alpha)anthracene-induced mammary carcinogenesis in rats. Prostaglandins Leukot Essent Fatty Acids 2018; 79:5ŌĆō14.
https://doi.org/10.1016/j.plefa.2008.05.005

29. Boatright KM, Salvesen GS. Mechanisms of caspase activation. Curr Opin Cell Biol 2003; 15:725ŌĆō31.
https://doi.org/10.1016/j.ceb.2003.10.009


30. Xu JJ, Dai XM, Liu HL, et al. A novel 7-azaisoindigo derivative-induced cancer cell apoptosis and mitochondrial dysfunction mediated by oxidative stress. J Appl Toxicol 2011; 31:164ŌĆō72.
https://doi.org/10.1002/jat.1577


31. Zhang J, Yu Q, Han L, Chen C, Li H, Han G. Study on the apoptosis mediated by cytochrome c and factors that affect the activation of bovine longissimus muscle during postmortem aging. Apoptosis 2017; 22:777ŌĆō85.
https://doi.org/10.1007/s10495-017-1374-2


32. Kemp CM, Parr T, Bardsley RG, Buttery PJ. Comparison of the relative expression of caspase isoforms in different porcine skeletal muscles. Meat Sci 2006; 73:426ŌĆō31.
https://doi.org/10.1016/j.meatsci.2005.12.009


33. Garcia-Macia M, Sierra V, Palanca A, et al. Autophagy during beef aging. Autophagy 2014; 10:137ŌĆō43.
https://doi.org/10.4161/auto.26659



34. Katsuragi Y, Ichimura Y, Komatsu M. p62/SQSTM1 functions as a signaling hub and an autophagy adaptor. FEBS J 2015; 282:4672ŌĆō8.
https://doi.org/10.1111/febs.13540


35. Yoshioka A, Miyata H, Doki YL, et al. C3, an autophagosome marker, is highly expressed in gastrointestinal cancers. Int J Oncol 2008; 33:461ŌĆō8.
https://doi.org/10.3892/ijo_00000028


36. Zoncu R, Bar-Peled L, Efeyan A, Wang S, Sancak Y, Sabatini DM. mTORC1 senses lysosomal amino acids through an inside-out mechanism that requires the vacuolar H(+)-ATPase. Science 2011; 334:678ŌĆō83.
https://doi.org/10.1126/science.1207056



37. Potes Y, Olivan M, Rubio-Gonzalez A, et al. Pig cognitive bias affects the conversion of muscle into meat by antioxidant and autophagy mechanisms. Animal 2017; 11:2027ŌĆō35.
https://doi.org/10.1017/S1751731117000714


38. Tanida I. Autophagy basics. Microbiol Immunol 2011; 55:1ŌĆō11.
https://doi.org/10.1111/j.1348-0421.2010.00271.x


39. Vij A, Yennamalli RM, Changotra H. Non-synonymous single nucleotide polymorphisms of ATG5 destabilize ATG12ŌĆōATG5/ATG16L1 complex: An enzyme with E3 like activity of ubiquitin conjugation system. Meta Gene 2017; 13:38ŌĆō47.
https://doi.org/10.1016/j.mgene.2017.04.008

40. Qian SY, Li X, Wang H, et al. Contribution of calpain to protein degradation, variation in myowater properties and the water-holding capacity of pork during postmortem ageing. Food Chem 2020; 324:126892
https://doi.org/10.1016/j.foodchem.2020.126892


41. Liu R, Warner RD, Zhou GH, Zhang W. Contribution of nitric oxide and protein S-nitrosylation to variation in fresh meat quality. Meat Sci 2018; 144:135ŌĆō48.
https://doi.org/10.1016/j.meatsci.2018.04.027


42. Zhang JY, Ma DY, Kim YHB. Mitochondrial apoptosis and proteolytic changes of myofibrillar proteins in two different pork muscles during aging. Food Chem 2020; 319:126571
https://doi.org/10.1016/j.foodchem.2020.126571


43. Zhang JY, Wang SS, Ge WP. Mechanisms of mitochondrial apoptosis-mediated meat tenderization based on quantitative phosphoproteomic analysis. Foods 2022; 11:3751
https://doi.org/10.3390/foods11233751



- TOOLS






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





