 |
 |
| Anim Biosci > Volume 35(7); 2022 > Article |
|
Abstract
Objective
Methods
Results
Notes
CONFLICT OF INTEREST
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
FUNDING
This work was supported by Suranaree University of Technology (SUT), Thailand Science Research and Innovation (TSRI), National Science, Research and Innovation Fund (NSRF), and the National Research Council of Thailand (NRCT) (SUT3-303-60-24-09).
The authors would like to acknowledge the staff of FTIR team (beam line 4.1: Infrared Spectroscopy and Imaging) from Synchrotron Light Research Institute (Public Organization) in Thailand for their technical suggestions. We would also like to express our sincere thanks to Pascal Mermillod from Reproductive Physiology and Behavior unit, National Institute of Agronomical Research (INRA), France for his guiding and proofreading this manuscript with the support of the Siam Huber Curien grant n¬į 42879QC.
Figure 1
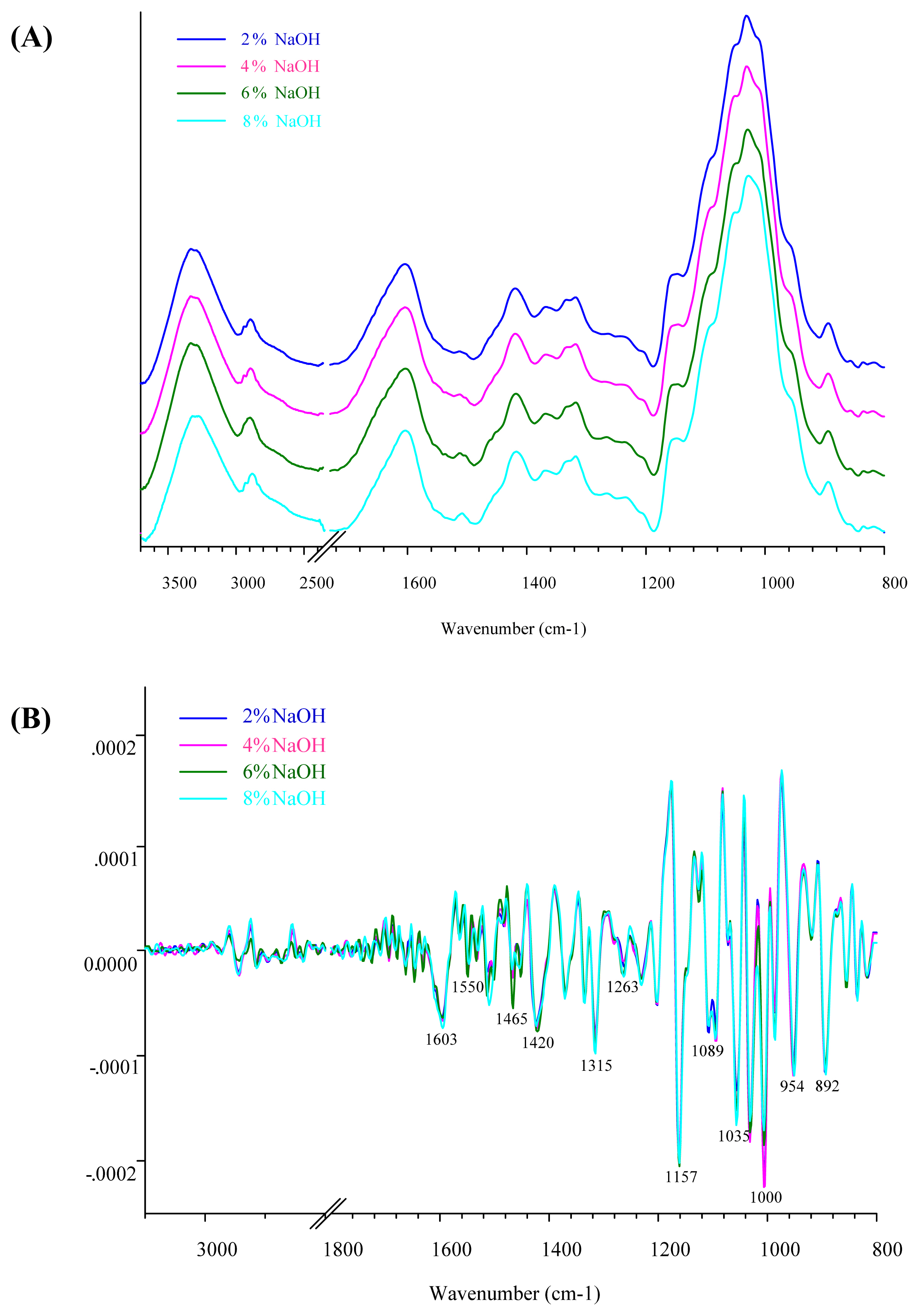
Figure 2
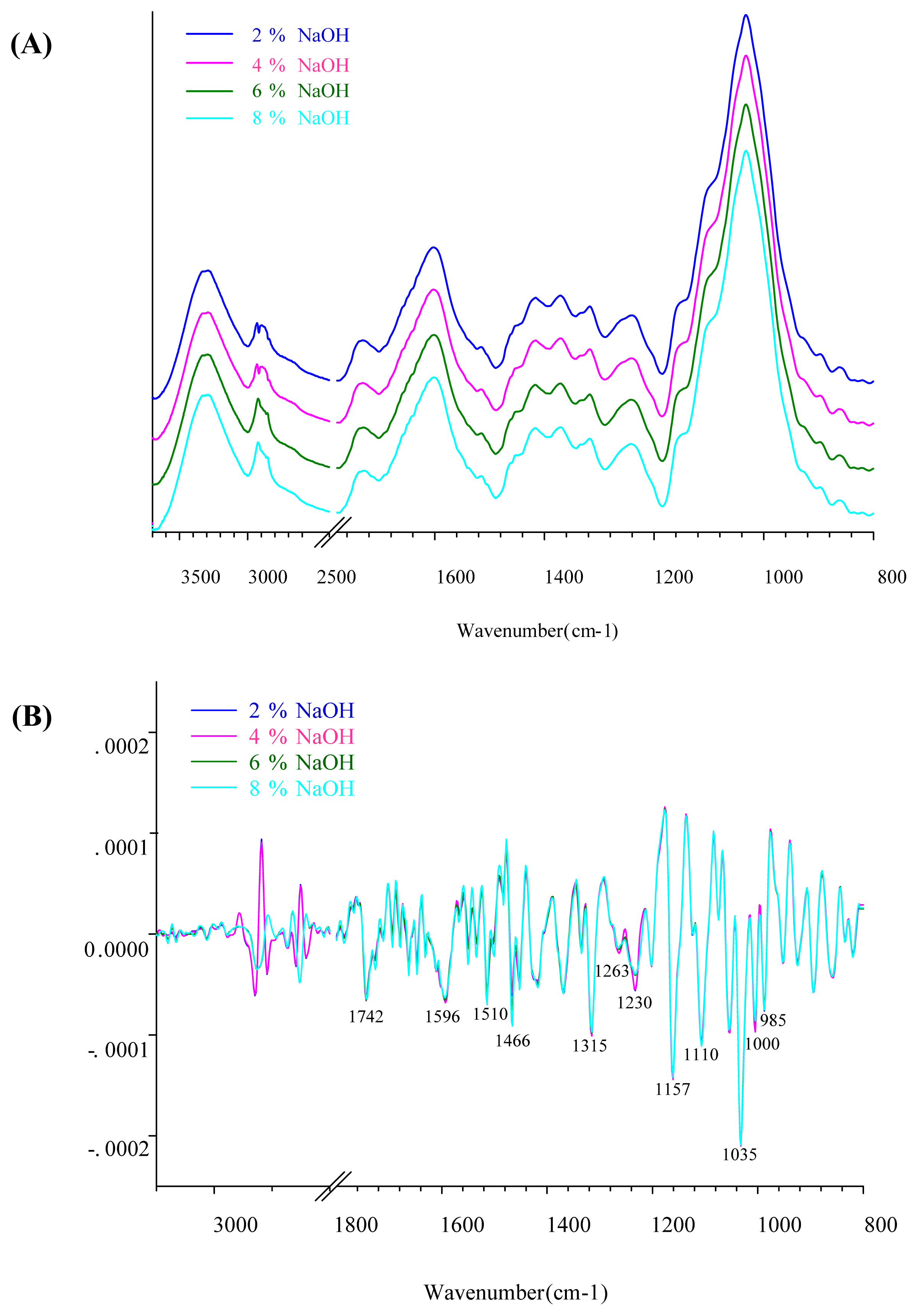
Figure 3

Figure 4
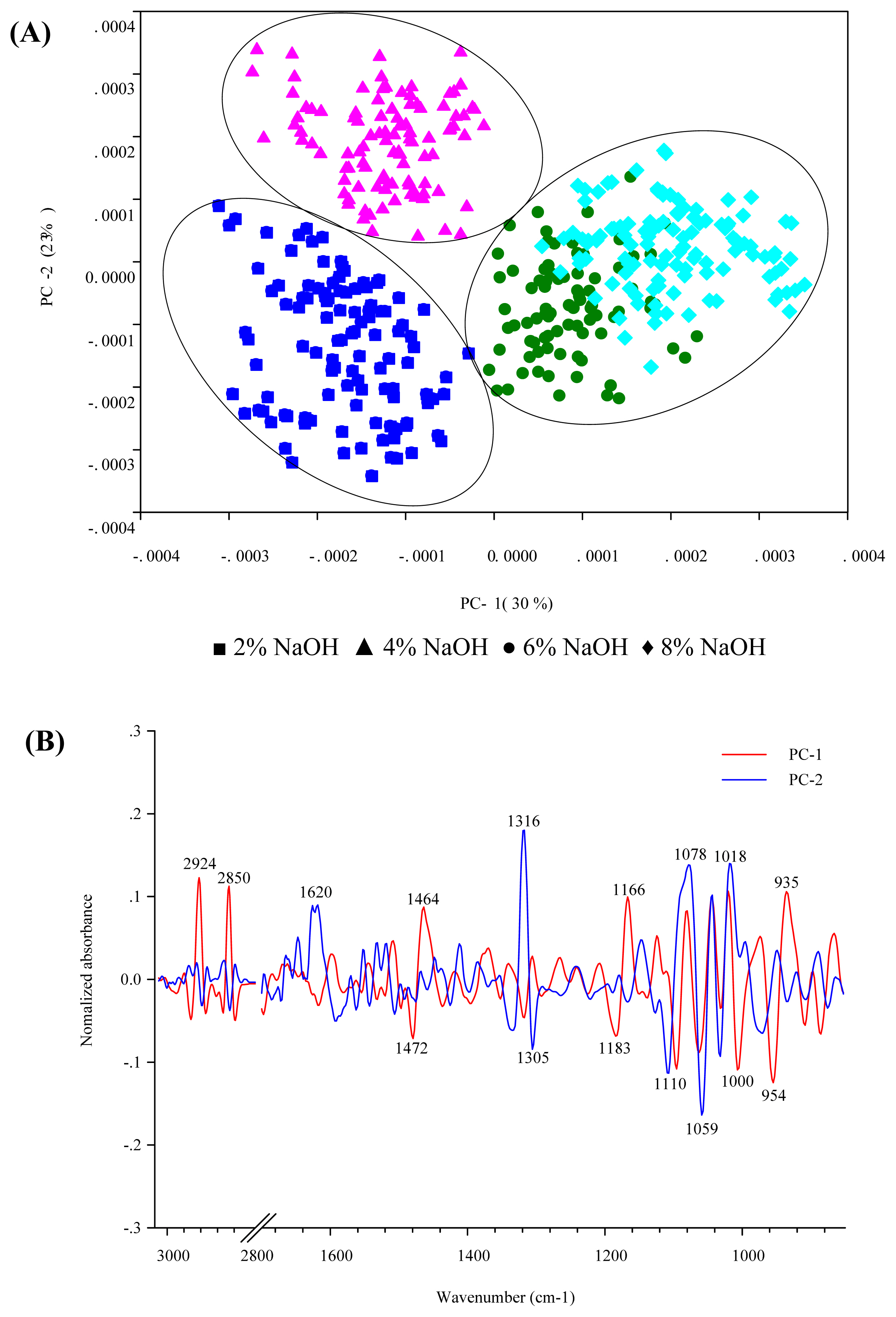
Table 1
Table 2
| Item | Concentration of NaOH levels | SEM | p-values | ||||||
|---|---|---|---|---|---|---|---|---|---|
|
|
|
||||||||
| 2% | 4% | 6% | 8% | ANOVA | Linear1) | Quadratic1) | Cubic1) | ||
| Cassava pulp extracted dietary fiber | |||||||||
| ‚ÄÉTotal dietary fiber | 23.60b | 24.29b | 27.93a | 27.60a | 0.57 | 0.001 | 0.001 | 0.285 | 0.032 |
| ‚ÄÉSoluble dietary fiber | 3.65 | 2.57 | 3.19 | 2.84 | 0.19 | 0.501 | 0.501 | 0.188 | 0.365 |
| ‚ÄÉInsoluble dietary fiber | 19.95b | 21.72ab | 24.73a | 24.76a | 0.63 | 0.001 | 0.001 | 0.117 | 0.089 |
| Cassava distiller’s dried grains extracted dietary fiber | |||||||||
| ‚ÄÉTotal dietary fiber | 22.85b | 25.12a | 25.33a | 25.35a | 0.30 | <0.001 | <0.001 | <0.001 | 0.009 |
| ‚ÄÉSoluble dietary fiber | 2.38 | 2.59 | 2.33 | 2.32 | 0.06 | 0.313 | 0.368 | 0.356 | 0.152 |
| ‚ÄÉInsoluble dietary fiber | 20.46b | 22.53a | 23.00a | 23.03a | 0.31 | <0.001 | <0.001 | <0.001 | 0.186 |
Table 3
| Wavenumber (cm‚ąí1) | Assignments | Reference |
|---|---|---|
| 2,800 ‚Äď 3,000 | C‚ÄďH stretching (aliphatic compounds) |
Lammers et al [22] Abidi et al [27] Pouzet et al [12] |
| 1,600 ‚Äď 1,680 | O‚ÄďH bending of adsorbed water |
Abidi et al [27] Pouzet et al [12] |
| 1,514, 1,595 | C‚ÄďH deformation of lignin | Lammers et al [22] |
| 1,300 ‚Äď 1,500 | C‚ÄďH bending (crystalline versus amorphous structure of cellulose) |
Abidi et al [27] Ying et al [28] |
| 1,200 ‚Äď 1,260 | C‚ÄďO stretching of hemicellulose |
Corredor et al [23] Abidi et al [27] Cheikh Rouhou et al [29] |
| 1,035 ‚Äď 1,160 | C‚ÄďO‚ÄďC glycoside in cellulose, C‚ÄďO vibration of crystalline cellulose, C‚ÄďO stretching and C‚ÄďC stretching of cellulose | Corredor et al [23] |
|
Abidi et al [27] Ying et al [28] Cheikh Rouhou et al [29] |
||
| 980 ‚Äď 1,000 | C‚ÄďO and ring stretching modes, C‚ÄďO stretching of starch |
Lammers et al [22] Abidi et al [27] |
| 800 ‚Äď 960 | Vibration of the pyranose ring, glucose ring stretch |
Lammers et al [22] Corredor et al [23] |
Table 4
| Item | Concentration of NaOH levels | SEM | p-values | ||||||
|---|---|---|---|---|---|---|---|---|---|
|
|
|
||||||||
| 2% | 4% | 6% | 8% | ANOVA | Linear1) | Quadratic1) | Cubic1) | ||
| Cassava pulp extracted dietary fiber | |||||||||
| ‚ÄÉC‚ÄďH stretching | 5.33 | 6.22 | 4.52 | 5.08 | 0.38 | 0.486 | 0.486 | 0.832 | 0.180 |
| ‚ÄÉO‚ÄďH bending of adsorbed water | 8.86 | 8.87 | 7.79 | 8.52 | 0.17 | 0.065 | 0.116 | 0.237 | 0.052 |
| ‚ÄÉC‚ÄďH deformation of lignin | 0.66 | 0.79 | 0.75 | 0.73 | 0.04 | 0.612 | 0.595 | 0.272 | 0.574 |
| ‚ÄÉC‚ÄďH bending of crystalline cellulose | 18.20bc | 17.06c | 19.54a | 18.57ab | 0.27 | 0.001 | 0.018 | 0.769 | <0.001 |
| ‚ÄÉC‚ÄďO stretching of hemicellulose | 5.40a | 5.04b | 5.41a | 5.32a | 0.05 | 0.006 | 0.686 | 0.046 | 0.001 |
| ‚ÄÉC‚ÄďO‚ÄďC glycoside, C‚ÄďO and C‚ÄďC stretching of cellulose | 25.28b | 25.51b | 26.56a | 25.69ab | 0.17 | 0.015 | 0.060 | 0.046 | 0.029 |
| ‚ÄÉC‚ÄďO stretching of starch | 17.96 | 18.21 | 18.75 | 19.11 | 0.24 | 0.346 | 0.082 | 0.905 | 0.831 |
| ‚ÄÉVibration of the pyranose ring | 18.15 | 17.97 | 16.99 | 17.15 | 0.23 | 0.178 | 0.055 | 0.679 | 0.318 |
| Cassava distiller’s dried grains extracted dietary fiber | |||||||||
| ‚ÄÉC‚ÄďH stretching | 3.56ab | 3.89a | 2.43bc | 2.10c | 0.25 | 0.007 | 0.002 | 0.345 | 0.087 |
| ‚ÄÉO‚ÄďH bending of adsorbed water | 7.52 | 6.84 | 6.98 | 6.41 | 0.18 | 0.134 | 0.040 | 0.878 | 0.315 |
| ‚ÄÉC‚ÄďH deformation of lignin | 1.03 | 0.68 | 0.81 | 0.87 | 0.08 | 0.528 | 0.648 | 0.252 | 0.506 |
| ‚ÄÉC‚ÄďH bending of crystalline cellulose | 29.03a | 27.30ab | 27.71ab | 26.38b | 0.37 | 0.049 | 0.015 | 0.746 | 0.189 |
| ‚ÄÉC‚ÄďO stretching of hemicellulose | 5.60 | 6.02 | 5.80 | 5.34 | 0.12 | 0.222 | 0.330 | 0.081 | 0.707 |
| ‚ÄÉC‚ÄďO‚ÄďC glycoside, C‚ÄďO and C‚ÄďC stretching of cellulose | 20.57b | 22.58a | 21.95a | 22.28a | 0.26 | 0.004 | 0.008 | 0.024 | 0.033 |
| ‚ÄÉC‚ÄďO stretching of starch | 22.67c | 23.23bc | 24.36ab | 25.64a | 0.35 | 0.001 | <0.001 | 0.356 | 0.807 |
| ‚ÄÉVibration of the pyranose ring | 10.03b | 9.44b | 9.94b | 10.98a | 0.18 | 0.003 | 0.005 | 0.004 | 0.593 |
REFERENCES
- TOOLS






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print





