 |
 |
| Anim Biosci > Volume 29(4); 2016 > Article |
|
Abstract
The Sal-like 1 gene (Sall1) is essential for kidney development, and mutations in this gene result in abnormalities in the kidneys. Mice lacking Sall1 show agenesis or severe dysgenesis of the kidneys. In a recent study, blastocyst complementation was used to develop mice and pigs with exogenic organs. In the present study, transcription activator-like effector nuclease (TALEN)-mediated homologous recombination was used to produce Sall1-knockout porcine fibroblasts for developing knockout pigs. The vector targeting the Sall1 locus included a 5.5-kb 5Ⲡarm, 1.8-kb 3Ⲡarm, and a neomycin resistance gene as a positive selection marker. The knockout vector and TALEN were introduced into porcine fibroblasts by electroporation. Antibiotic selection was performed over 11 days by using 300 Îźg/mL G418. DNA of cells from G418-resistant colonies was amplified using polymerase chain reaction (PCR) to confirm the presence of fragments corresponding to the 3Ⲡand 5Ⲡarms of Sall1. Further, mono- and bi-allelic knockout cells were isolated and analyzed using PCRârestriction fragment length polymorphism. The results of our study indicated that TALEN-mediated homologous recombination induced bi-allelic knockout of the endogenous gene.
Many patients with end-stage organ failure can only be treated using organ transplantation; however, transplantation is performed only in a limited number of patients because of a shortage of organ donors (Ye et al., 1994; Sprangers et al., 2008). Therefore, one of the ultimate goals of regenerative medicine is the generation of functional organs from stem cells (Liu et al., 2013). For stem cell replacement therapy, induced pluripotent stem cells (iPSCs) specific to patients can be generated using somatic cell nuclear transfer (SCNT) and reprogramming technology (Takahashi and Yamanaka, 2006; Takahashi et al., 2007). Recently, mice with rat pancreas were developed by injecting rat pluripotent stem cells (PSCs) into mouse blastocysts lacking the pancreatic and duodenal homeobox 1 gene (Kobayashi et al., 2010). The Sal-like 1 gene (Sall1) is essential for kidney development using studying Sall1-knockout mouse (Nishinakamura and Takasato, 2005). In a recent study, PSC-derived kidney was generated via blastocyst complementation, using Sall1-knockout blastocysts and mouse iPSCs or mouse embryonic stem cells (mESCs) (Usui et al., 2012). In another study, cloned pigs with exogenic pancreas were developed using blastocyst complementation (Matsunari et al., 2013). However, generation of PSC-derived kidneys in pigs has not been reported to date. The first requirement for producing exogenic kidneys in pigs is the development of heterozygous Sall1-knockout pigs.
Knockout pigs can be produced by performing SCNT in porcine fibroblasts (Dai et al., 2002). In general, knockout fibroblast can be produced using a gene-targeting system comprising knockout vectors. However, the efficiency for producing knockout fibroblasts is lower than that for producing knockout mESCs (Denning and Priddle, 2003; Laible and Alonso-GonzĂĄlez, 2009). Moreover, ESCs from domestic animals are not available easily. To overcome these problems, zinc-finger nucleases, transcription activator-like effector nucleases (TALENs), and clustered regularly interspaced short palindromic repeats (CRISPR)/Cas9 systems have been used to develop knockout domestic animals (Carlson et al., 2012; Tan et al., 2013; Xin et al., 2013; Hai et al., 2014; Ni et al., 2014; Whitworth et al., 2014). Recently, although CRISPR/Cas9 system have been used to generate a knock-out pig, knockout pigs were developed using TALEN with high efficient (Carlson et al., 2012; Xin et al., 2013; Yao et al., 2014). Targeting efficiency is very important for producing transgenic pigs via SCNT. Generation of knockout pigs via SCNT and transfected cells is associated with more advantages than generation of knockout pigs via DNA microinjection because the former method allows the assessment of vector integration in transfected cells before they can be used as nuclear donors (Wolf et al., 2000).
The aim of this study was to determine whether TALEN-mediated homologous recombination system on the Sall1 gene locus in the porcine primary somatic cells is much more efficient than conventional homologous recombination system. We observed that use of TALEN-mediated homologous recombination resulted in the production of bi-allelic Sall1-knockout fibroblasts.
Genomic DNA was isolated from the ear tissue of Chicago miniature pigs and the Sall1 gene was amplified using polymerase chain reaction (PCR). The 5Ⲡarm of the knockout vector was amplified using a forward primer with an additional NotI site (GCGGCCGCAATTGGCCTA CGAAAGAGGAGCAGCCTGTG) and a reverse primer with an additional BamHI site (GGATCCGATGG GGGCTGGCTCTTCGGTCTTGATGAA). The 3Ⲡarm of the knockout vector was amplified using a forward primer with an additional SmaI site (CCCGGGATTCCAC AAGTCACCCAGCGCCGAGGAGAA) and a reverse primer with an additional SalI site (GTCGACGGA ATCTGTCTCAGCTCATATCCCAACACA). PGK-neo, which was used as a positive selection maker, was amplified from pKJ2-neo plasmid by using a forward primer with an additional BamHI site (GGATCCT ACCGGGTAGGGGAGGCGCTTTTC) and a reverse primer with an additional SmaI site (CCCGGGCCT CAGAAGAACTCGTCAAGAAGG). All the amplicons were subcloned into pGEM-T Easy vector (Promega Co., Madison, WI, USA), and their sequences were confirmed. The knockout vector was constructed as follows. The fragment corresponding to the 5Ⲡarm (NotIâBamHI) was subcloned into pBluescript SK- (pBSK-) to produce a pBSK-5â˛-arm plasmid. The PGK-neo fragment (BamHIâSmaI) was then ligated into the BamHIâSmaI site of the pBSK-5â˛-arm plasmid to produce a pBSK-5â˛-arm-neo plasmid. The fragment corresponding to the 3Ⲡarm was then inserted into the SmaIâSalI site of the pBSK-5â˛-arm-neo plasmid to produce a pBSK-5â˛-arm-neo-3â˛-arm plasmid. Finally, the pBSK-5â˛-arm-neo-3â˛-arm plasmid was digested using NotIâSalI and was ligated into the NotIâSalI site of pMCDT-A plasmid (Gibco BRL; Grand Island, NY, USA) to produce the Sall1-knockout vector. Thus, the knockout vector contained porcine Sall1 5Ⲡarm (5.5 kb), PGK-neo as the positive selectable marker, porcine Sall1 5Ⲡarm (1.8 kb), and a gene fragment encoding diphtheria toxin A (DT-A) as a negative selectable marker. For transfection into porcine fibroblasts, the Sall1 knockout vector was digested with NotI.
TALEN expression vector containing the CMV promoter that was designed to cleave the exon 2 of porcine Sall1 was purchased from ToolGen, Inc. (Seoul, Korea). TALEN binding sites are shown in Figure 1A. For the validation of TALEN-mediated homologous recombination via a T7 endonuclease assay, porcine fibroblasts were transfected with the TALEN expression vector by electroporation and the genomic DNA of the transfected fibroblasts was isolated using GenElute Mammalian Genomic DNA Miniprep Kit (Sigma-Aldrich, St. Louis, MO, USA). The target region of TALEN was amplified from the genomic DNA by using gene-specific primers (sense: AGAAACCCAGGTGGGCTCCCGGAGGAGGCA; antisense: TGCATGCGGATGTGCTGCTGTAGGACCACA). The amplicons were purified using QIAquick Gel Extraction Kit (Qiagen; Hilden, Germany), denatured by heating, and annealed in hybridization buffer (10 mM Tris-HCl [pH 8.5], 75 mM KCl, and 1.5 mM MgCl2) to produce heteroduplex DNAs. The heteroduplex DNAs were treated with T7 endonuclease I (4 U; New England Biolabs; Ipswich, MA, USA) at 37°C for 15 min. The resultant fragments were electrophoresed on a 2% agarose gel and were visualized by staining the gel with ethidium bromide (EtBr).
Porcine ear fibroblasts were prepared from tissues biopsied from the ear of specific pathogen-free Minnesota male miniature pigs maintained at the Seoul National University (Ahn et al., 2011). For electroporation, the fibroblasts were cultured in DMEM supplemented with 15% defined fetal bovine serum (FBS), 1Ă non-essential amino acids, 1Ă sodium pyruvate, 10â4 M β-mercaptoethanol, 100 units/mL penicillin, and 100 Îźg/mL streptomycin at 37°C in a humidified atmosphere of 5% CO2. Transfection was performed according to a previously reported method (Kim et al., 2012). Briefly, the fibroblasts were harvested by treatment with 0.25% trypsin and were resuspended in Hamâs F10 medium (HyClone Co.; Logan, UT, USA) at a density of 5Ă106 cells per 0.4 mL of the medium. The fibroblast suspension (400 ÎźL) was electroporated with 5 Îźg of the linearized knockout vector and each 2.5 Îźg of left and right TALEN expression vectors in a 4-mm cuvette by using four 1-ms pulses and 450-V capacitive discharges with BTX Electro-cell Manipulator (ECM 2001, BTX; Holliston, MA, USA). After electroporation, the cuvette was placed on ice for 10 min. The cells in the cuvette were resuspended in 10 mL of the medium, dispensed in a 48-well plate (800 cells/well), and cultured for 48 h. Cells harboring the knockout vector were selected over 10 to 12 days by culturing them in a medium containing 300 Îźg/mL geneticin-G418 (Gibco/Life Technologies, Gaithersburg, MD, USA). After selection, single colonies were passaged in 48-well plates and were cultured further for analysis.
Knockout colonies were identified by performing the first and second PCRs. For the first PCR, 100 ΟL cell suspension from the 48-well plates was recovered by centrifugation. The cells were resuspended in 36 ΟL 50 mM NaOH. For genome extraction, the cells were incubated at 95°C for 10 min and were neutralized by treatment with 4 ΟL 1 M Tris-HCl (pH 8.0). The cells were then centrifuged at 10,000 rpm and 4°C for 10 min to obtain a crude genome extract. The first PCR was performed in a 50-ΟL reaction mixture containing 20 ΟL crude genome extract, 0.1 M each of Neo 3-2 sense (primer A: GCCTGCTTGCC GAATATCATGGTGGAAAAT) and Sall1 Sc 5-2 antisense (primer B: GGGGAGAGGAAGGGAGAAGCTTAATAG TGG) primers for regions outside the 3Ⲡarm, and KOD SYBR qPCR Mix (TOYOBO Co. Ltd., Osaka, Japan). Amplification was performed for 32 cycles of denaturation at 98°C for 30 s, annealing at 62°C for 30 s, and extension at 68°C for 2.5 min by using the Mx3000P qPCR System (Agilent Technologies, La Jolla, CA, USA).
Positive colonies from the first PCR were subcultured in 10-cm dishes. The cells were frozen in media containing 10% demethyl sulfoxide (DMSO) for further analyses. Genomic DNA of positive clones identified in the first PCR was isolated using GenElute Mammalian Genomic DNA Miniprep Kit and was used as the template for the second PCR to accurately identify the Sall1-knockout cells. The second PCR amplication was conducted using the 3Ⲡarm, 5Ⲡarms, and internal PCR. The 3Ⲡarm was amplified using 100 ng genomic DNA under the same conditions as the first PCR. The 5Ⲡarm was amplified using 100 ng genomic DNA, Sall1 Sc Inl S primer (primer C: AAGCTGATTCAGATGCAGGCTTTTCCC) and Neo 5â˛-2 antisense primer (primer D: TGCTAAAGCGCATGCT CCAGACTGCCTTGG), and i-MAX II DNA Polymerase (iNtRON Biotechnology, Seongnam, Korea). Amplification was performed for 32 cycles of denaturation at 94°C for 30 s, annealing at 66°C for 30 s, and extension at 72°C for 6 min. To identify colonies with bi- and mono-allelic mutations, an internal PCR was performed using 100 ng genomic DNA, primers (primer E: GCCACCCTCCAGTGGCAAAAGCGAAGACAG; primer F: TCATCGAAGGAGCCCGTGTCGGACTCCATG) against a part of exon 3 that was excluded from the knockout vector, and KOD FX Neo Polymerase (TOYOBO Co. Ltd., Japan). Amplification was conducted using a 2-step PCR involving 29 cycles of denaturation at 94°C for 10 s and annealing/extension at 68°C for 1 min. PCR products (20 ÎźL) were validated by electrophoresis on a 0.8% agarose gel.
To validate the colonies showing mono- and bi-allelic mutations, PCRârestriction fragment length polymorphism (PCRâRFLP) was performed against a part of exon 3 that was excluded from the knockout vector. The PCR mixture included 100 ng genomic DNA, Sall1 E2 Sc sense primer (primer H: AATCGTAAATGAGAGTCCAGCCTCTCCCCC) and Sall1 Sc 5-2 antisense primer (primer B: GGGAGAGGAAGGGAGAAGCTTAATAGTGG), and KOD FX Neo Polymerase. Amplification was performed using a 2-step PCR involving 30 cycles of denaturation at 94°C for 30 s and annealing/extension at 68°C for 6 min. The PCR products were purified using QIAquick Gel Extraction Kit and were digested using SmaI. The digested PCR products (20 ÎźL) were electrophoresed on 1% agarose gel to confirm the presence of targeted (2.8 and 1.8 kb) and non-targeted alleles (2.6 and 1.5 kb).
In this study, we used TALEN to produce knockout fibroblasts. The target sequences of TALEN are located in exon 2 of Sall1 (Figure 1A). To examine whether TALEN could be used for the targeted cleavage of endogenous Sall1 in porcine fibroblasts, genomic DNA isolated from fibroblasts transfected with the TALEN expression vector was treated with T7 endonuclease I, which specifically cleaves heteroduplexes formed by the hybridization of wild-type and mutant DNA sequences. We observed that the T7 endonuclease I cleaved a fragment with an indel mutation in the genomic DNA isolated from fibroblasts transfected with the TALEN expression vector compared with genomic DNA isolated from negative controls, which were not transfected with the TALEN expression vector (Figure 1B). We examined the activity of TALEN at 30°C and 37°C because previous studies reported that the activity of TALENs is higher at 30°C than at 37°C. However, we observed no difference in the activity of the TALENs at these 2 temperatures.
Figure 2 shows a schematic diagram of Sall1 knockout strategy in porcine fibroblasts transfected with the Sall1 knockout vector. The knockout vector contained a 5Ⲡhomologous arm (5.5 kb), PGK-neo as the positive selection marker, 3Ⲡhomologous arm (1.8 kb), and DT-A as the negative selection marker. The selection markers PGK-neo and DT-A did not contain a polyA signal and were only expressed if they were inserted at a gene locus before the polyA signal. This strategy allowed easy selection of knockout cells because cells with random integration of the knockout vector did not express PGK-neo and DT-A because of the absence of the polyA signal.
Efficiency of Sall1 knockout by using TALEN-mediated homologous recombination is presented in Table 1. In all, 110 and 126 G418-resistant colonies transfected with and without the TALEN expression vector, respectively, and with the Sall1 knockout vector were analyzed using the first PCR. Of these, 6 and 71 colonies, respectively, yielded positive results. The first PCR showed that TALEN-mediated homologous recombination along with the Sall1 knockout vector had higher targeting efficiency (55.5%) than the knockout vector alone. In order to accurate analysis of homologous recombination, we analyzed 4 colonies in the without TALEN and 9 colonies in with TALEN by the 3â˛, 5Ⲡand internal PCR analysis. Four colonies transfected with the Sall1 knockout vector but not with the TALEN expression vector were all wild-type colonies. Of the 9 colonies transfected with both the Sall1 knockout vector and the TALEN expression vector, 6 colonies (colony no. 22, 24, 27, 30, 46, and 58) were found to be mono-allelic knockout colonies and 2 colonies (colony No. 19 and 83) were found to be wild-type colonies. In addition, Colony number 5 in the Figure 3 in 9 colonies showed bi-allelic mutation (Table 1 and Figure 3). PCRâRFLP was conducted to validate the colonies showing bi- and mono-allelic mutations (Figure 4). Results of PCRâRFLP confirmed that only colony number 5 had bi-allelic mutations.
Recently, exogenic pancreas was developed in mice and pigs by performing blastocyst complementation with PSCs (Kobayashi et al., 2010; Matsunari et al., 2013). In another study, exogenic kidneys were developed in mice via blastocyst complementation, using injection of mouse iPSCs or mESCs into the Sall1-knockout blastocysts (Usui et al., 2012). These results raise the possibility that exogenic human kidney may be develop via blastocyst complementation using injection of human ES cell or iPSCs into the Sall1-knockout porcine blastocysts. Sall1 is involved in kidney development (Nishinakamura and Takasato, 2005). To develop exogenic organs in pigs, it is important to develop heterozygous knockout pigs lacking specific genes required for kidney organogenesis.
In the present study, we produced Sall1-knockout porcine fibroblasts by using TALEN-mediated homologous recombination and Sall1 knockout vector to using donor cells for producing Sall1-knockout pigs. The targeting efficiency (55.5%) was higher in the TALEN-mediated homologous recombination than in the conventional homologous recombination using knockout vector alone. Further, we confirmed that of the 7 Sall1-knockout fibroblast colonies, 6 and 1 colonies showed mono-allelic and bi-allelic mutations, respectively. Targeting efficiency of a conventional knockout vector for targeting the porcine Îą-1,3-galactosyltransferase locus was 0% to 9.3% in the absence of TALENs (Dai et al., 2002; Lai et al., 2002; Klumiuk et al., 2010). Moreover, the knockout efficiency of TALENs alone is higher than that of conventional methods (Carlson et al., 2012; Xin et al., 2013; Yao et al., 2014). Carson et al. (2012) reported 64% knockout efficiency by using TALENs alone in primary cells. Further, Xin et al. (2013) reported 89.5% knockout efficiency by using TALENs alone in porcine fetal fibroblasts. However, Yao et al. (2014) reported only 5% to 22% knockout efficiency by using TALENs alone in porcine fibroblasts. TALENs induce double-stranded DNA breaks in a specific target sequence, produce frameshift mutations, and silence the expression of target genes. The above authors used only TALENs against target genes to produce knockout cells. In the present study, we used TALEN-mediated homologous recombination along with a knockout vector to produce knockout fibroblasts. We observed low targeting efficiency because of the use of the knockout vector as a DNA donor for TALEN-mediated homologous recombination.
However, these results indicated that TALEN-mediated homologous recombination occurred more efficiently on the Sall1 gene locus in the porcine primary somatic cells compare to that of conventional homologous recombination system. Also, fibroblasts with mono- and bi-allelic mutations obtained in this study could be used as nuclear donors for SCNT to develop Sall1-knockout pigs for producing exogenic kidneys by using PSCs.
ACKNOWLEDGMENTS
This study was supported by a grant for the Woo Jang-Choon projects (PJ00784904) from the Rural Development Administration, Republic of Korea.
Figure 1
Binding site for TALEN and validation of TALEN activity. (A) The binding site of TALEN in exon 2 of porcine Sall1. (B) T7 endonuclease assay of porcine fibroblasts transfected with TALEN. TALEN, transcription activator-like effector nuclease; M, size markers; NC, negative control. Fibroblasts transfected with TALEN expression vector were incubated at 30°C and 37°C for 24 h.

Figure 2
Strategy for the knockout of the porcine Sall1 locus. (A) The Sall1 locus, (B) the knockout vector, (C) the targeted locus. Solid black boxes indicate the exon. Arrows indicate polymerase chain reaction (PCR) primer pairs used for detecting homologous recombination.
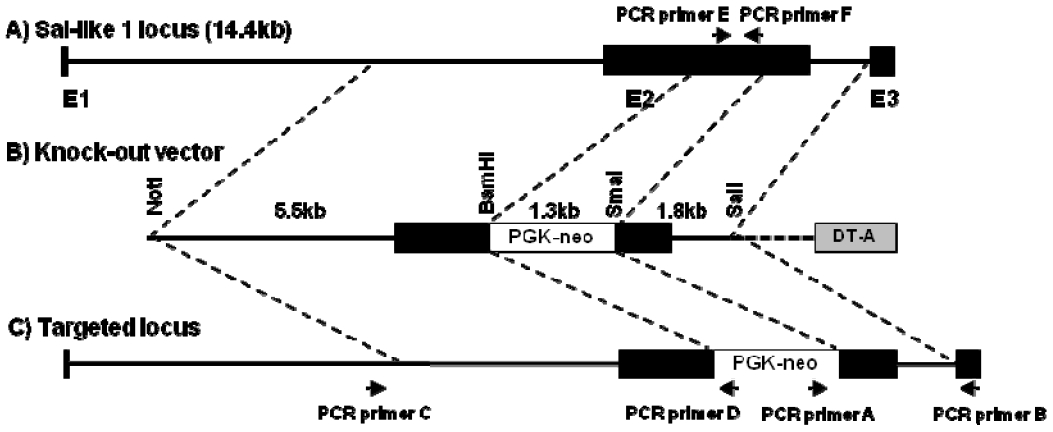
Figure 3
Analysis of Sall1-knockout fibroblasts via PCR. Representative EtBr-stained agarose gel showing PCR amplicons of (A) the 3Ⲡarm, (B) the 5Ⲡarm, and (C) internal PCR. PCR, polymerase chain reaction; EtBr, ethidium bromide; M, size marker (1-kb ladder); NC, negative control; PC, positive control; number, G418-resistant colonies.
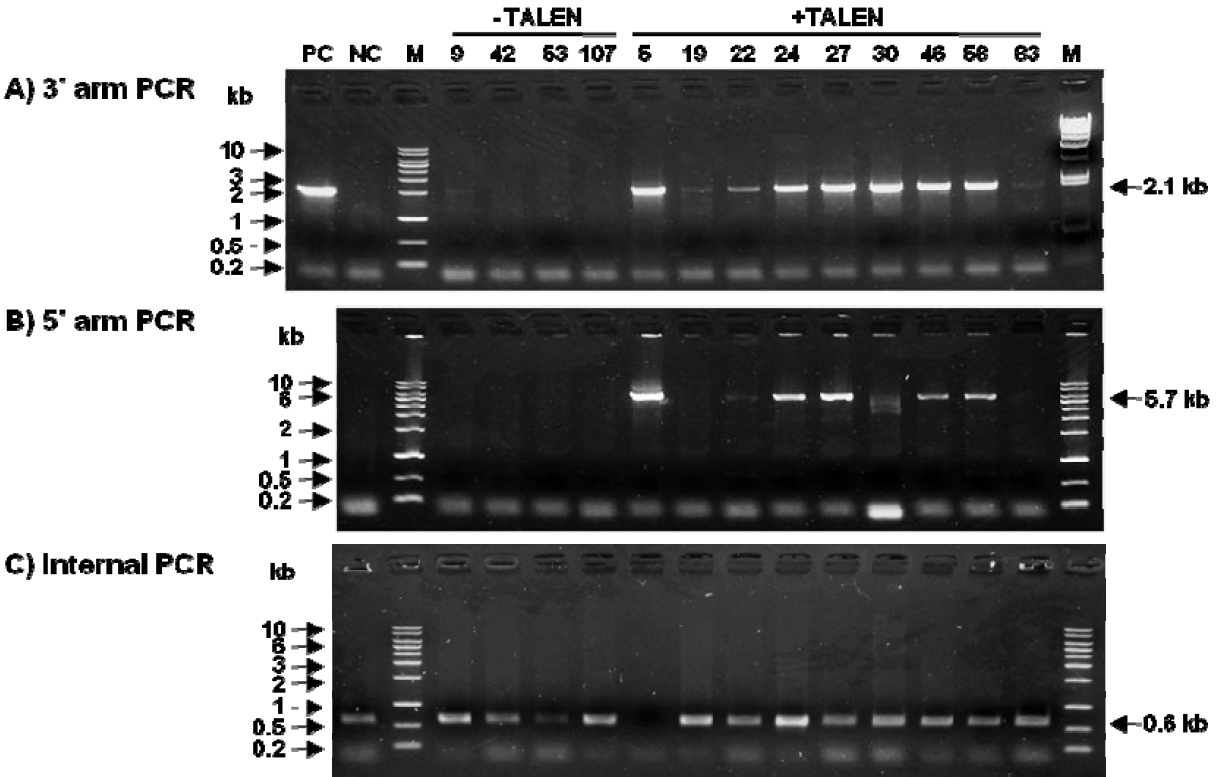
Figure 4
Analysis of colonies showing bi- and mono-allelic mutations by performing PCRâRFLP. (A) Diagram indicating the restriction enzyme site in the Sall1 locus and (B) the targeted locus. (C) Representative EtBr-stained agarose gel showing amplicons obtained by PCRâRFLP. PCRâRFLP, polymerase chain reactionârestriction fragment length polymorphism; EtBr, ethidium bromide; M, size marker (1-kb ladder); WT, wild type; Mo, mono-allelic mutation; Bi, bi-allelic mutation.
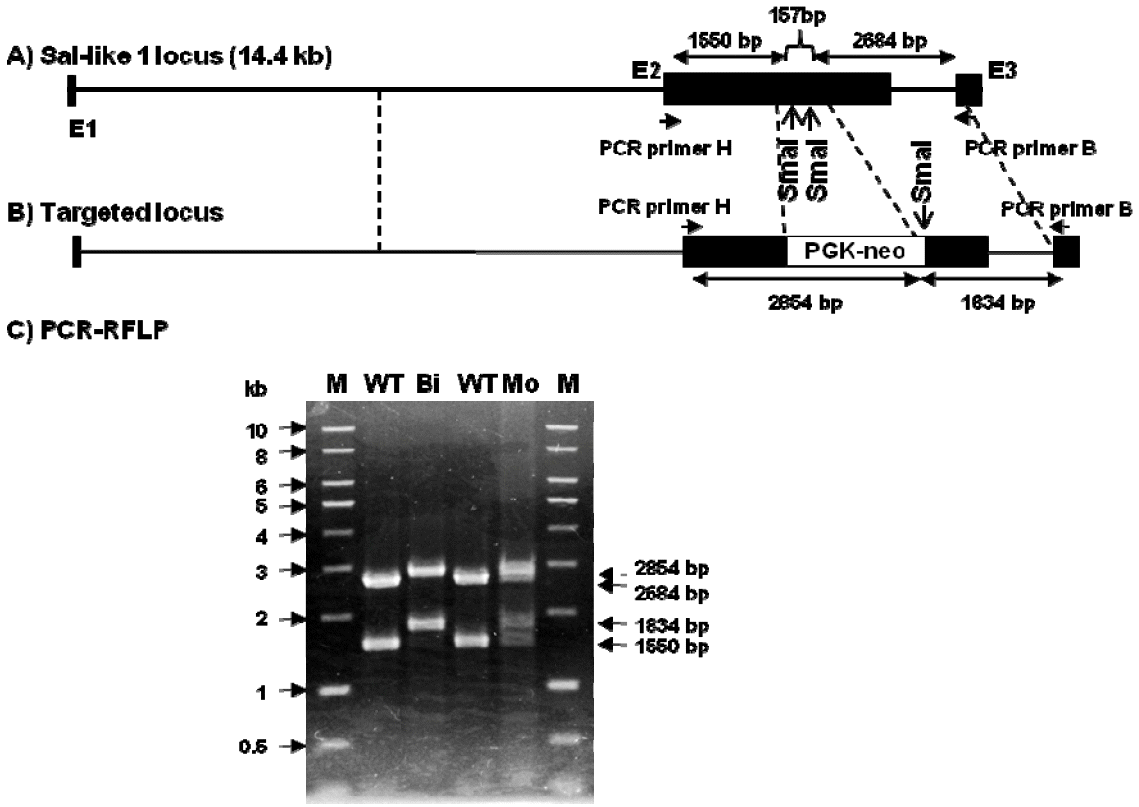
Table 1
Gene targeting efficiency at the Sall1 locus by TALEN-mediated homologous recombination using a knockout vector
REFERENCES
Ahn KS, Kim YJ, Kim M, Lee BH, Heo SY, Kang MJ, Kang YK, Lee JW, Lee KK, Kim JH, Nho WG, Hwang SS, Woo JS, Park JK, Park SB, Shim H. 2011. Resurrection of an alpha-1,3-galactosyltransferase gene-targeted miniature pig by recloning using postmortem ear skin fibroblast. Theriogenology 75:933â939.


Carlson DF, Tan W, Lillico SG, Stverakova D, Proudfoot C, Christian M, Voytas DF, Long CR, Whitelaw CB, Fahrenkrug SC. 2012. Efficient TALEN-mediated gene knockout in livestock. Proc Natl Acad Sci USA 109:17382â17387.



Dai Y, Vaught TD, Boone J, Chen SH, Phelps CJ, Ball S, Monahan JA, Jobst PM, McCreath KJ, Lamborn AE, Cowell-Lucero JL, Wells KD, Colman A, Polejaeva IA, Ayares DL. 2002. Targeted disruption of the alpha 1,3-galactosyltransferase gene in cloned pigs. Nat Biotechnol 20:251â255.


Denning C, Priddle H. 2003. New frontiers in gene targeting and cloning: success, application and challenges in domestic animals and human embryonic stem cells. Reproduction 126:1â11.


Hai T, Teng F, Guo R, Li W, Zhou Q. 2014. One-step generation of knockout pigs by zygote injection of CRISPR/Cas system. Cell Res 24:372â375.



Kim JW, Kim HM, Lee SM, Kang MJ. 2012. Porcine knock-in fibroblasts expressing hDAF on Îą-1,3-galactosyltransferase (GGTA1) gene locus. Asian Australas J Anim Sci 25:1473â1480.



Klumiuk N, Aigner B, Brem G, Wolf E. 2010. Genetic modification of pigs as organ donors for xenotransplantation. Mol Reprod Dev 77:209â221.


Kobayashi T, Yamaguchi T, Hamanaka S, Kato-Itoh M, Yamazaki Y, Ibata M, Sato H, Lee YS, Usui J, Knisely AS, Hirabayashi M, Nakauchi H. 2010. Generation of rat pancreas in mouse by interspecific blastocyst injection of pluripotent stem cells. Cell 142:787â799.


Laible G, Alonso-GonzĂĄlez L. 2009. Gene targeting from laboratory to livestock: Current status and emerging concepts. Biotechnol J 4:1278â1292.


Lai L, Kolber-Simonds D, Park KW, Cheong HT, Greenstein JL, Im GS, Samuel M, Bonk A, Rieke A, Day BN, Murphy CN, Carter DB, Hawley RJ, Prather RS. 2002. Production of alpha-1,3-galactosyltransferase knockout pigs by nuclear transfer cloning. Science 295:1089â1092.


Matsunari H, Nagashima H, Watanabe M, Umeyama K, Nakano K, Nagaya M, Kobayashi T, Yamaguchi T, Sumazaki R, Herzenberg LA, Nakauchi H. 2013. Blastocyst complementation generates exogenic pancreas in vivo in apancreatic cloned pigs. Proc Natl Acad Sci USA 110:4557â4562.



Ni W, Qiao J, Hu S, Zhao X, Regouski M, Yang M, Polejaeva IA, Chen C. 2014. Efficient gene knockout in goats using CRISPR/Cas9 system. PLoS One 9:e106718



Nishinakamura R, Takasato M. 2005. Essential roles of Sall1 in kidney development. Kidney Int 68:1948â1950.


Sprangers B, Waer M, Billiau AD. 2008. Xenotransplantation: Where are we in 2008? Kidney Int 74:14â21.


Takahashi K, Yamanaka S. 2006. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663â676.


Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. 2007. Induction of pluriotent stem cells from adult human fibroblasts by defined factors. Cell 131:861â872.


Tan W, Carlson DF, Lancto CA, Garbe JR, Webster DA, Hackett PB, Fahrenkrug SC. 2013. Efficient nonmeiotic allele introgression in livestock using custom endonucleases. Proc Natl Acad Sci USA 110:16526â16531.



Usui J, Kobayashi T, Yamaguchi T, Knisely AS, Nishinakamura R, Nakauchi H. 2012. Generation of kidney from pluripotent stem cells via blastocyst complementation. Am J Pathol 180:2417â2426.


Ye Y, Niekrasz M, Kosanke S, Welsh R, Jordan HE, Fox JC, Edwards WC, Maxwell C, Cooper DK. 1994. The pig as a potential organ donor for man. A study of potentially transferable disease from donor pig to recipient man. Transplantation 57:694â703.


Whitworth KM, Lee K, Benne JA, Beaton BP, Spate LD, Murphy SL, Samuel MS, Mao J, OâGorman C, Walters EM, Murphy CN, Driver J, Mileham A, McLaren D, Wells KD, Prather RS. 2014. Use of the CRISPR/Cas9 system to produce genetically engineered pigs from in vitro-derived oocytes and embryos. Biol Reprod 91:78



Wolf E, Schernthaner W, Zakhartchenko V, Prelle K, Stojkovic M, Brem G. 2000. Transgenic technology in farm animals - progress and perspectives. Exp Physiol 85:615â625.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print





