 |
 |
Abstract
During two years, four samples per year were collected in Vietnam from rice bran, cassava residue, brewerŌĆÖs grain, tofu residue, soybean meal, coconut cake, sweet potato vines and water spinach for chemical analysis and assessment of water holding capacity (WHC). The selected feedstuffs represent fibre-rich plant sources and agro-industry co-products commonly used in pig feeding in Vietnam. The content (g/kg DM) of crude protein (CP), ether extract (EE) and non-starch polysaccharides (NSP) varied between feedstuffs and ranged from 21 to 506 for CP, from 14 to 118 for EE and from 197 to 572 for NSP. Cassava residue had a high starch content of 563 g/kg DM, while sweet potato vines, water spinach, coconut cake and soybean meal had a high content of sugars (63ŌĆō71 g/kg DM). The content of individual neutral sugars varied between feed ingredients, with the highest content of arabinose, galactose and glucose in tofu residue, the highest content of xylose in brewerŌĆÖs grain and the highest content of mannose in coconut cake. The content of uronic acid was high for cassava residue, tofu residue, sweet potato vines and water spinach (57ŌĆō88 g/kg DM). The content of soluble non-cellulosic polysaccharides (S-NCP) was positively correlated (r2 = 0.82) to the WHC. The content (g/kg DM) of CP, NDF, neutral sugars, total NSP, total NCP, S-NCP and total dietary fibre in tofu residue, water spinach and coconut cake varied (p<0.05) between years. In conclusion, diet formulation to pigs can be improved if the variation in chemical composition of the fibre fraction and in WHC between potential feed ingredients is taken into account.
The major fraction in diets for pigs is dietary carbohydrates, which can be divided according glycosidic linkages into sugars, oligosaccharides and two broad classes of polysaccharides, starch and non-starch polysaccharides. Non-starch polysaccharides (NSP) together with lignin, has been defined as the dietary fibre (DF) fraction in feedstuffs and food, and can be used as a collective measure of their fibre content (Trowell et al., 1976; Theander et al., 1994; Bach Knudsen, 1997).
Potential feed resources used for animals in Vietnam, as well as in many other countries in South-East Asia, derive primarily from the vegetable foods and agro-industry co-products, such as cassava leaves, sweet potato vines, water spinach, rice bran, cassava residue, brewerŌĆÖs grain and tofu residue. They represent under-utilized feedstuffs all having high fibre content, which may cause limitations in their use in diets for pigs, in particular for post-weaning and growing pigs, due to their bulky nature and a limited capacity to ferment fibre in pigs (Bach Knudsen and J├Ėrgensen, 2001). Thus, in order to better utilize available fibre-rich feedstuffs in diets for pigs, their chemical and physical cheracteristics has to be described and taken into account in feed formulation.
Currently, the fibre fraction in feedstuffs used in pig feed is still commonly characterized by use of crude fibre and more frequently by use of the Van Soest detergent system (Van Soest et al., 1991) in which the fibre fraction is separated into neutral detergent fibre (NDF), acid detergent fibre (ADF) and lignin. However, despite considerable refinement in describing the fibre fraction compared with crude fibre, the detergent system has the limitation of not accounting for soluble carbohydrates. In order to more accurately describe the whole fibre fraction in feed and food, including both soluble and insoluble NSP, the Uppsala method (Theander et al., 1995) is today accepted as being the most relevant method to apply (Bach Knudsen, 1997).
The main objective of the study was to characterize the fibre fraction in selected fibre-rich feedstuffs, originating from green plants and agro-industry co-products and used in pig feed, with modern analytical methods, and to evaluate the variation in composition within feedstuff and between years.
A total of 64 samples, comprising rice bran (RB), cassava residue (CR), brewerŌĆÖs grain (BG), tofu residue (TFR), soybean meal (SBM), coconut cake (CC), sweet potato vine (SPV) and water spinach (WSP) were analysed. Sweet potato vines and WSP are green plants that were collected from households in Hanoi (Vietnam) and the seasonal co-product CR (Cassava Starch Factory, Ha Tay, Vietnam) was collected 4 times during the first season in 2008 and the second season in 2009. The other samples were collected every third month during 2 years from agricultural processing factories producing all year, these were RB (Rice Processing Factory, An Giang, Vietnam), BG (HABECO, Ha Noi, Vietnam), TFR (from a tofu processing household in Hanoi, Vietnam), CC (Coconut Processing Factory, Ben Tre, Vietnam) and SBM (Cai Lan Vegetable Oil Company, Quang Ninh, Vietnam). Cassava residue, BG, TFR, SPV, and WSP were collected in fresh form and were freeze-dried, whereas RB, SBM and CC were in air-dry form. All samples were milled through a 0.5 mm screen and stored at room temperature until analysis.
All analyses were performed in duplicate. Water holding capacity (WHC) was analysed using an adaptation of the filtration method proposed by Robertson and Eastwood (1981). A dry sample 0.5 g was soaked in 200 ml of distilled water for 24 h at room temperature and then filtered through Whatman No. 1 filter paper. Samples filtered rapidly and filtration was completed after 10 min. A sample of the residue was weighed (wet weight) before drying overnight in an oven at 105┬░C and reweighed (dry weight) which gave WHC.
Dry matter (DM) (967.03), crude protein (CP) (984.13), and ether extract (EE) (920.39) and ash (942.05) were analysed according to the standard AOAC methods (1990). The CP content was calculated as nitrogen├Ś6.25. Neutral detergent fibre was determined by the method of Van Soest et al. (1991). Starch and sugars were analysed by an enzymatic method (Larsson and Bengtsson, 1983).
Total, soluble and insoluble NSP and their constituent sugars were determined as alditol acetates by gas-liquid chromatography (GLC) for neutral sugars, and by a calorimetric method for uronic acids using a modification of the Uppsala method (Theander et al., 1995) as described by Bach Knudsen (1997). Starch was removed from the sample in acetate buffer using termostatable ╬▒-amylase and amyloglucosidase. Soluble polymers were precipitated with ethanol. Precipitated and insoluble polysaccharides were hydrolysed with H2SO4. Analyses of released neutral sugars were performed on an Agilent 6890 gas chromatograph equipped with flame-ionisation detector. A 30 m├Ś0.32 mm i.d. narrow-bore capillary column (Supelco SP 2380, Cat No. 2-4116) was used. Klason lignin was measured gravimetrically as the insoluble residue to 12 M H2SO4. Total DF is defined as amylase-resistant polysaccharides plus Klason lignin.
Content of cellulose was calculated as:
Total non-starch polysaccharide (T-NSP) content was calculated as:
Total non-cellulosic polysaccharide (T-NCP) content was calculated as:
Soluble NCP (S-NCP) content was calculated as:
Data on chemical composition of individual feedstuffs was described by mean values and their standard deviation (SD) using descriptive statistics. The variation between years within the individual feedstuffs was analysed by one-way analysis of variance in Minitab software using the model;
Where Yij is the observed dependent variable, ╬╝ the overall mean, ╬▒i the effect of the year (1, 2), ╔øj is the residual error. Mean values were considered different at a p-value Ōēż0.05.
The correlation between WHC and S-NCP in the whole data set was analysed using linear regression analysis.
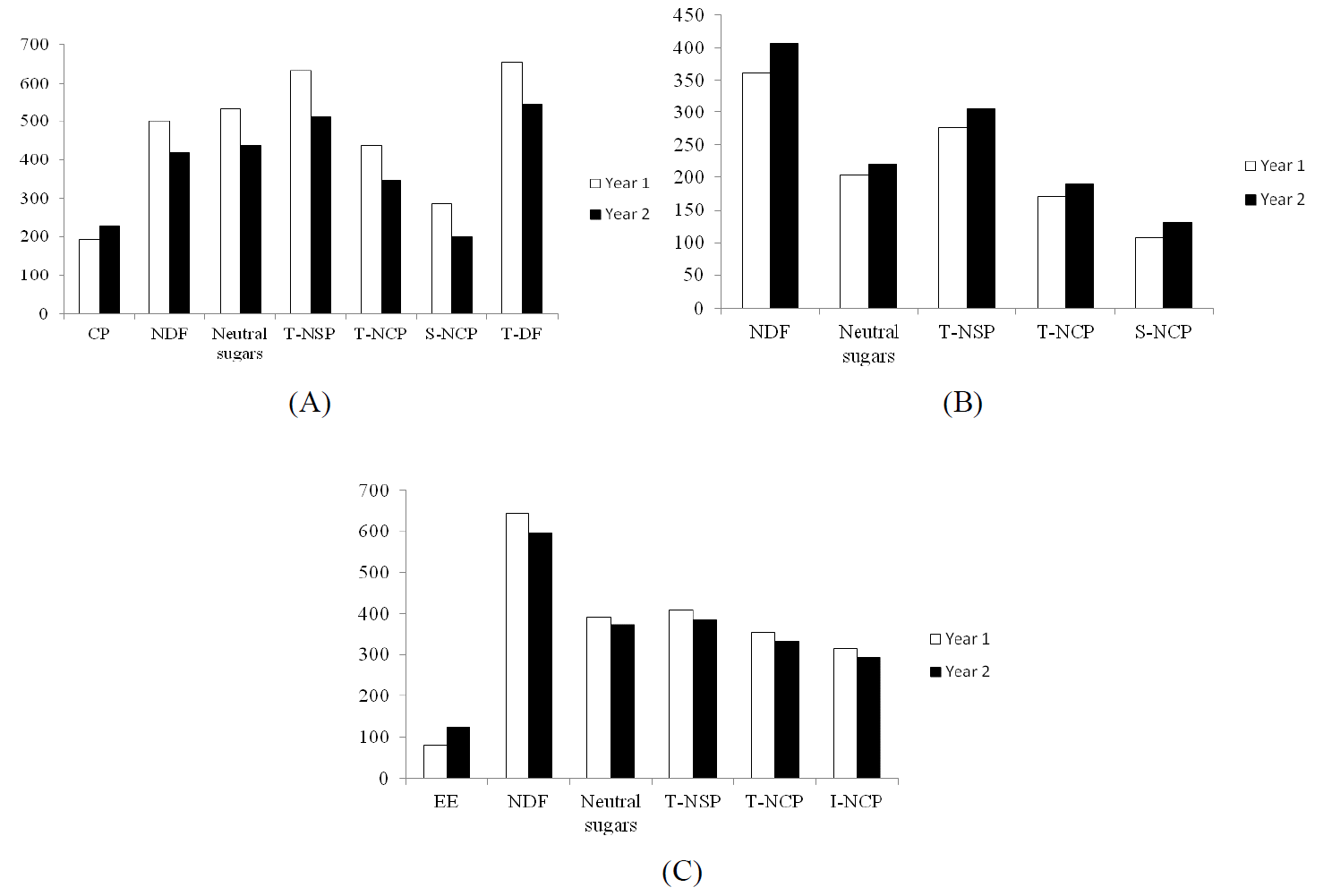
Soybean meal had the highest CP content (506 g/kg DM), followed by BG and WSP (264ŌĆō308 g/kg DM). Tofu residue, CC, SPV and RB had a CP content in the range 116ŌĆō211 g/kg DM, and CR had the lowest CP content (21 g/kg DM) (Table 1). It was only the TFR which showed a variable CP content between years (p<0.05) (Figure 1).
BrewerŌĆÖs grain, RB, CC and TFR were high in EE (58ŌĆō118 g/kg DM), whereas CR, SPV and WSP had a low content of EE (15ŌĆō36 g/kg DM). In general, CR and the green plants (SPV and WSP) were lower in EE than the agro-industry co-products. It was only CC that showed a variation in EE between years (p<0.005) (Figure 1).
The NDF content was high in the green plants and in the agro-industry co-products (384ŌĆō619 g/kg DM), with the highest NDF value recorded for CC (Table 1). All green plants and agro-industry co-products were higher in NDF than SBM. There was variation between years (p<0.05) in the NDF content in TFR, WSP and CC (p<0.05) (Figure 1).
Ash content was low for CR, TFR and BG (20ŌĆō40 g/kg DM), while high values were recorded for RB, SPV, WSP, CC and SBM (71ŌĆō131 g/kg DM) (Table 1).
The agro-industry co-products CR, TFR, BG and RB were all low in sugar content (1ŌĆō20 g/kg DM), while the sugar content of the agro-industry co-product CC and the green plants was high (63ŌĆō71 g/kg DM) and comparable to SBM (Table 1).
The content of starch varied a lot between the potential feed resources. Low to negligible starch content was recorded for TFR, SPV, CC, WSP and SBM (5ŌĆō28 g/kg DM), while intermediate starch content was found for BG and high starch content in CR (Table 1).
All vegetables and agro-industry co-products were higher in T-NSP content (254ŌĆō572 g/kg DM) than SBM (197 g/kg DM) (Table 1). Cellulose accounted for more than 50% of the T-NSP in RB, followed by approximately 45% cellulose in CR and SPV, and 31ŌĆō38% cellulose in TFR, BG and WSP. The lowest value was found for CC, where cellulose accounted for 13% of the T-NSP and the corresponding figure for SBM was 28% (Table 1).
The proportion of I-NCP was high in BG, RB and CC where it accounted for 88ŌĆō94% of the T-NCP (Table 1). In SPV, the I-NCP proportion of T-NCP was 54%, followed by values in the range 34ŌĆō43% for CR, TR and WSP, and 62% for SBM.
The content of DF, followed the same pattern as the T-NSP content with high values for the vegetables and the agro-industry co-products (357ŌĆō599 g/kg DM) compared to SBM (222 g/kg DM) (Table 2). The proportion of I-DF was high in BG, RB and CC where it accounted for 91ŌĆō97% of the T-DF (Table 2). In SPV, the I-DF proportion of T-DF was 84%, followed by values in the range 59ŌĆō74% for CR, TFR and WSP, and 76% for SBM.
The neutral sugars were the most abundant constituent of T-NSP accounting for 73% of the T-NSP in WSP and 81ŌĆō96% of the T-NSP in the other feed ingredients (Table 2). The content of individual neutral sugars varied substantially between feed ingredients. The content of arabinose, galactose and glucose were highest in TFR, the highest content of xylose was in BG and mannose was highest in CC. The content of uronic acid was high for CR, TFR, SPV and WSP (57ŌĆō88 g/kg DM), while low values were found in BG, RB and CC (11ŌĆō14 g/kg DM) and intermediate in SBM (26 g/kg DM).
The content of Klason lignin was high (103ŌĆō160 g/kg DM) in BG, RB and SPV, followed by WSP, CC and CR (59ŌĆō87 g/kg DM) (Table 2). Tofu residue and SBM had the lowest content of Klason lignin (25ŌĆō27 g/kg DM).
For TFR, WSP and CC there was variation between years (p<0.05) in the content of neutral sugars, T-NSP, T-NCP, S-NCP, I-NCP, T-DF and S-DF (Figure 1).
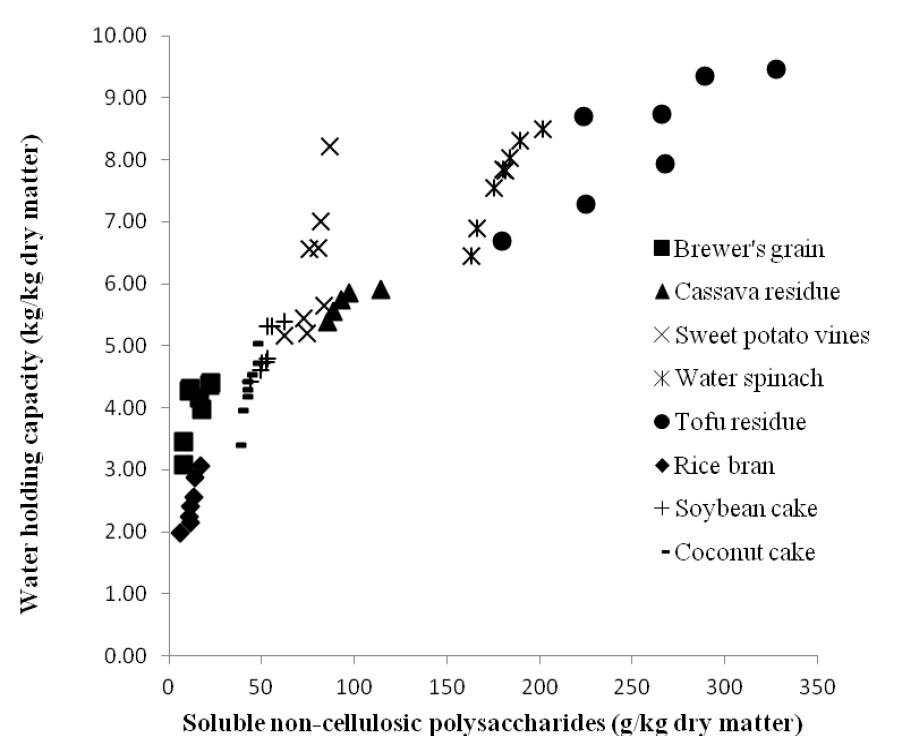
The WHC varied greatly between feed ingredients with the highest values recorded for TFR and WSP, and the lowest value for RB (Figure 2). Soybean meal had an intermediate WHC. Overall, the WHC of the feed ingredients studied was closely correlated (R2 = 0.82; p<0.001) to the content of S-NCP (Figure 3). No other correlation was found between WHC and the components of the DF fraction.
Generally, the content of CP, EE and ash in green plants and agro-industry co-products were similar to those previously reported from Vietnam (NIAH, 2001) and other tropical countries (Gohl, 1981; Lekule et al., 1990). However, for some of the feed ingredients (e.g. tofu residue, coconut cake and water spinach) there was variation between years.
The plant cell wall can be divided into three layers: middle lamella, primary and secondary cell wall (Kunzek et al., 1999). The middle lamella connects single cells to form tissues. The main components of the middle lamella are pectin substances and it acts as a cementing substance in the middle lamella. In the primary cell walls, a cellulose-xyloglucan framework (cellulose-hemicellulose network) is embedded in a pectin matrix and exists as microfibrils. When the cell growth has finished the cellulose microfibrils aggregate in the secondary wall and lignification can cause cell wall fixation. Thus, a change in chemical composition of the plant material and their co-products not only depends on the botanical origin of the plants and the type of processing applied, but also on the tissue type and the maturity at the plant at harvesting time. For example, while the nature of cellulose varies little between plants, the composition of the amorphous matrix usually shows considerable variation from tissue to tissue within plant and between plants. In cereals, which are monocotyledonous plants, the main cell wall NSP of whole grain cereals are arabinoxylans, cellulose and ╬▓-glucan with some variation between cereals (Bach Knudsen, 2001). In contrast, the amorphous matrix in dicotyledonous plants can differ markedly from that in monocotyledonous plants due to different tissue types, exemplified by the huge difference in content and properties of pectic substances (Bacic et al., 1988; McDougall et al., 1996).
BrewerŌĆÖs grain in the current study was higher in CP (308 vs. 215 g/kg DM) and lower in T-NSP (366 vs. 463 g/kg DM) than reported by Serena and Bach Knudsen (2007). The chemical composition of BG will vary depending on barley variety, harvest time, malting and mashing conditions, and the quality and type of adjuncts added in the brewing process (Huige, 1994; Santos et al., 2003). BrewerŌĆÖs grain basically consists of the husk-pericarp-seed coat layers that covered the original barley grain and depending on the evenness of the malting process more or less starchy endosperm and walls of empty aleurone cells may be included in the co-product. In consequence, the starch content will be negligible and the major components of BG will be the walls of the husk-pericarp-seed coat, which are rich in cellulose and non-cellulosic polysaccharides and lignin (Mussatto et al., 2006; Serena and Bach Knudsen, 2007), and protein, lipids and ash.
The chemical composition of RB is variable due to varieties used and in particular to the milling process (Bor et al., 1991). In Vietnam, the RB is classified into type 1, 2 and 3, based on CP and crude fibre (CF) content (NIAH, 2001) with CP descending and CF increasing going from type 1 to type 3. The RB included in the current study was classified as type 2. Rice bran, a by-product of rice polishing, constitutes about 10% of paddy rice and consists of the seed coat, the major part of the germ and the outer layer of the endosperm, and some small fragments of endosperm. Thus, due to the endosperm present RB can contain a fair amount of starch (100ŌĆō550 g starch/kg DM) (Saunders, 1986; Bor et al., 1991). The RB analyzed in the current study had a starch content of 219 g/kg DM. Free sugars in RB are concentrated in the aleurone layer and were low in the current study compared with values (3ŌĆō5%) reported earlier (Saunders, 1986). Most grains and their co-products contain a large amount of arabinoxylans and cellulose (Bach Knudsen, 1997; Choct, 2006). The T-NSP and cellulose content in RB in the current study were in line with earlier data (Saunders, 1986).
The co-products (SBM and TFR) from soybean, a grain legume, differed markedly in chemical compositions due to different processing. Thus, while TFR is obtained from the extraction of soybean milk, SBM is the residue obtained from the extraction of soybean oil. To produce soybean milk, soybean seeds are soaked for a day and then ground with water before filtering. Consequently, TFR is lower in CP and sugars, and higher in ether extract and NSP fractions than SBM. Cellulose and xylans, which are the major NSP in cereal grains, are only found in the hulls or husks of most grain legumes (Choct, 1997). The major NSP in the cotyledon of legumes are pectic polysaccharides. The starch content in TFR and SBM was low and was lower than reported by others (Lekule et al., 1990; Bach Knudsen, 1997).
The T-NSP content in CC in the current study was lower than reported by Bach Knudsen (1997). However, composition of individual sugars in the NSP fraction was comparable, with predominance of mannans and with about 5% cellulose and trace amounts of other neutral sugars (rhamnose, fucose, arabinose and xylose). Coconut cake is the co-product obtained after milk extraction from the fresh coconut. After shelling and paring the residue is disintegrated and pressed in a screw press to separate the coconut milk from the solid materials. The coconut milk is used fresh or preserved and can be used for the preparation of virgin coconut oil, while the solid coconut residue is used in animal feeding.
Cassava residue, a co-product from the extraction of starch from cassava roots, was low in protein and high in fibre and starch. The fibre fraction was high in uronic acids and more than 50% of the T-NCP fraction was soluble. The predominant sugars in the NSP fraction were glucose, galactose and xylose. The high content of starch (560 g/kg DM) could due to entrapment in the cellulose-hemicellulose matrix (Divya Nair et al., 2011). Lower values (500 g/kg DM) were reported by Carta et al. (1999) and higher values (620 g/kg DM) by John et al. (2007). The variation in starch content in CR between studies is probably because most of the processing is done under poorly controlled technological conditions (Pandey et al., 2000).
Water spinach and SPV in the current study were high in CP and fibre (NDF and T-NSP). However, the CP values were lower and NDF values were higher than reported by Dung et al. (2002). This was probably due to differences between studies in time of harvest and in the sampling protocol (i.e. the cutting height of WSP and the length of SPV). The content of total sugars (63ŌĆō71 g/kg DM) was high in both WSP and SPV, while starch content was low (13ŌĆō27 g/kg DM). These results were in line with earlier data (Woolfe, 1992). The T-NSP fraction was composed of 38% cellulose and 62% T-NCP in WSP, and 46% cellulose and 54% T-NCP in SPV, respectively. Woolfe (1992) reported that the NSP fraction in SPV was composed of 34% cellulose and 66% T-NCP.
Hydration properties of the feed is very important for effective digestion to occur in the animal and can be characterized by the swelling capacity, solubility, WHC and water binding capacity (WBC). The terms WHC and WBC have been used differently in the literature since both reflect the ability of the fibre source to incorporate water within its matrix. According to Kunzek et al. (1999), the WBC describes the ability of a sample to bind water when exposed to an external stress, while WHC describes the ability to hold water within the feed matrix under atmospheric pressure. The WHC is usually obtained by centrifugation at high speed (McConnell et al., 1974; Robertson and Eastwood, 1981) and sometimes by filtration (Robertson and Eastwood, 1981). The WHC values reported in the current study were obtained by filtration. The filtration method is robust and easy to perform, and has been suggested to follow more closely the condition likely found in the gastro-intestinal tract and should resemble normal physiological conditions (Robertson and Eastwood, 1981). Results from the current study showed a strong correlation between content of S-NCP and WHC in plant material and agro-industry co-products. This could be due to the occurrence of more gaps within the cell matrix that can retain excess water in feed ingredients which are high in S-NCP. In agreement with our findings, Serena and Bach Knudsen (2007) have shown that the S-NCP fraction in co-products from vegetable food and agro industries was linearly related to selected hydration properties, such as swelling and WBC.
There are marked differences in the chemical composition and water holding capacity between potential feed ingredients derived from green plants and agro-industry co-products. In particular, there were major differences in the composition and properties of carbohydrate fraction of the potential feed ingredients. It appears reasonable to assume that diet formulation to pigs can be improved if the variation in chemical composition, in particular of the fibre fraction, is taken into account.
ACKNOWLEDGEMENTS
This study was financed by Sida-SAREC (Swedish International Development Cooperation Agency - Department for Research Cooperation), through the regional MEKARN program and the Swedish University of Agricultural Sciences (SLU). The authors would like to thank the researchers at the Department of Animal Nutrition and Forage of the National Institute of Animal Science and Anna Greta Haglund who is the staff at the Department of Animal Nutrition and Management of SLU for their help in carrying out the study.
Figure┬Ā1
Average chemical composition (g/kg DM) of tofu residue (A), water spinach (B) and coconut cake (C) collected during two separate years. CP = Crude protein; EE = Ether extract; NDF = Neutral detergent fibre; T-NSP = Total non-starch polysaccharides; T-NCP = Total non-cellulosic polysaccharides; I-NCP = Insoluble non-cellulosic polysaccharides; S-NCP = Soluble non-cellulosic polysaccharides.
Figure┬Ā2
Water holding capacity (kg/kg dry matter) in selected fibre-rich plant sources and agro-industry co-products (n = 8). The standard error is shown by vertical bars.

Figure┬Ā3
Correlation between water holding capacity (WHC, kg/kg DM) and soluble non-starch polysaccharides (S-NCP, g/kg DM) in selected fibre-rich plant sources and agro-industry co-products (WHC = 3.50+0.0214 S-NCP, R2 = 0.82, p<0.001).
Table┬Ā1
Chemical composition of selected fibre-rich plant sources and agro-industry co-products (n = 8) (g/kg DM)
Table┬Ā2
Content of dietary fibre components in selected fibre-rich plant sources and agro-industry co-products (n = 8) (g/kg DM)
REFERENCES
AOAC. 1990. Official methods of analysis. 15th editionAssociation of Official Analytical Chemists; Arlington, Virgina, USA:
Bach Knudsen KE. 1997. Carbohydrate and lignin contents of plant materials used in animal feeding. Anim Feed Sci Technol 67:319ŌĆō338.

Bach Knudsen KE. 2001. The nutritional significance of ŌĆ£dietary fibreŌĆØ analysis. Anim Feed Sci Technol 90:3ŌĆō20.

Bach Knudsen KE, J├Ėrgensen H. 2001. Intestinal degradation of dietary carbohydrates-from birth to maturity. Digestive Physiology in Pigs. Lindberg JE, Ogle B, editorsCABI Publishing; Wallingford: p. 109ŌĆō120.

Bacic A, Harris PJ, Stone BA. 1988. Structure and function of plant cell walls. Biochem Plant 14:297ŌĆō372.

Bor SL, Barber S, Benedito de Barber C. 1991. Rice bran: Chemistry and technology. Rice Utilization. II:2nd EdBor SL, editorVan Nostrand Reinhold; New York, USA: p. 313ŌĆō362.
Carta FS, Soccol CR, Ramos LP, Fontana JD. 1999. Production of fumaric acid by fermentation of enzymatic hydrolysates derived from cassava bagasse. Bioresource Technol 68:23ŌĆō28.

Choct M. 1997. Feed non-starch polysaccharides: Chemical structures and nutritional significance. Feed Milling Intern June13ŌĆō26.
Choct M. 2006. Enzyme for the feed industry: Past, present and future. WorldŌĆÖs Poult Sci J 62:5ŌĆō15.

Divya Nair MP, Padmaja G, Moorthy SN. 2011. Biodegradation of cassava starch factory residue using a combination of cellulases, xylanases and hemicellulases. Biomass Bioenergy 35:1211ŌĆō1218.

Dung NN, Manh XLH, Ud├® P. 2002Tropical fibre sources for pigs-digestibility, digesta retention and estimation of fibre digestibility in vitro. Anim Feed Sci Technol 102:109ŌĆō124.

Gohl B. 1981. Tropical feeds. Feed information summaries and nutritive values. FAO; Rome:
Huige NJ. 1994. Brewery by-products and effluents. Handbook of Brewing. Hardwick WA, editorMarcel Dekker; New York: p. 501ŌĆō550.

Kopinski JS, Kinh LV, Vinh D, Ninh PH, Burren B. 2007. Digestible energy, starch and cyanide content of sundried cassava residue in Vietnam. Manipulating Pig Production XI. Paterson JE, Barker JA, editorsAustralasian Pig Science Association; Werribee, Australia: p. 118
Kunzek H, Kabbert R, Gloyna D. 1999. Aspects of material science in food processing: Changes in plant cell walls of fruits and vegetables. Z Lebensm Unters Forsch A 208:233ŌĆō250.

Larsson K, Bengtsson S. 1983. Best├żmning av l├żtt tillg├żngliga kolhydrater i v├żxtmaterial. Determination of non-structural carbohydrates in plant materialMethod description no 22. Uppsala, Sweden: National Laboratory of Agricultural Chemistry.
Lekule FP, J├Ėrgensen H, Fern├Īndez JA, Just A. 1990. Nutritive value of some tropical feedstuffs for pigs: Chemical composition, digestibility and metabolizable energy content. Anim Feed Sci Technol 28:91ŌĆō101.

McConnell AA, Eastwood MA, Mitchell WD. 1974. Physical characteristics of vegetable foodstuffs that could influence bowel function. J Sci Food Agric 25:1457ŌĆō1464.


McDougall GJ, Morrison IM, Stewart D, Hillman JR. 1996. Plant cell walls as dietary fibre: Range, structure, processing and function. J Sci Food Agric 70:133ŌĆō150.

Minitab. 2000. Statistical software version 13.31. UserŌĆÖs Guide to Statistics. Minitab; PA, USA:
Mussatto SI, Dragone G, Roberto IC. 2006. BrewersŌĆÖ spent grains: Generation, characteristics and potential applications. J Cereal Sci 43:1ŌĆō14.

NIAH. 2001. Composition and nutritive value of animal feed in Vietnam. National Institute of Animal Husbandry, Agricultural Publishing House; Hanoi, Vietnam:
Pandey A, Soccol CR, Nigam P, Soccol VT, Vandenberghe PSL, Mohan R. 2000. Biotechnological potential of agro-industrial residues: 2. Cassava bagasse. Bioresour Technol 74:81ŌĆō87.

Robertson JA, Eastwood MA. 1981. An investigation of the experimental conditions which could affect water-holding capacity of dietary fibre. J Sci Food Agric 32:819ŌĆō825.

Santos M, Jim├®nez JJ, Bartolom├® B, G├│mez-Cordov├®s C, del Nozal MJ. 2003. Variability of brewersŌĆÖ spent grain within a brewery. Food Chem 80:17ŌĆō21.

Serena A, Bach Knudsen KE. 2007. Chemical and physicochemical characterisation of co-products from vegetable food and agro industries. Anim Feed Sci Technol 139:109ŌĆō124.

Theander O, ├ģman P, Westerlund E, Graham H. 1994. Enzymatic/chemical analysis of dietary fibre. J AOAC Int 77:703ŌĆō709.


Theander O, ├ģman P, Westerlund E, Andersson R, Pettersson D. 1995. Total dietary fibre determined as neutral sugar residues, uronic acid residues, and Klason lignin (The Uppsala Method): Collaborative study. J AOAC Int 78:1030ŌĆō1044.


Trowell HC, Southgate DAT, Wolever TMS, Leeds AR, Gassull MA, Jenkins DJA. 1976. Dietary fibre redefined. Lancet 967.

Van Soest PJ, Robertson JB, Lewis BA. 1991. Methods for dietary fibre, neutral detergent fibre and non-starch polysaccharides in relation to animal nutrition. J Dairy Sci 74:3583ŌĆō3597.


Woolfe JA. 1992. Sweet potato: Untapped food resource. Cambridge University Press; Cambridge:
- TOOLS





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print





