The Effect of Plant Extracts on In-vitro Ruminal Fermentation, Methanogenesis and Methane-related Microbes in the Rumen
Article information
Abstract
The effect on methanogens attached to the surface of rumen ciliate protozoa by the addition of plant extracts (pine needles and ginkgo leaves) was studied with particular reference to their effectiveness for decreasing methane emission. The plant extracts (pine needles and ginkgo leaves) were added to an in vitro fermentation incubated with rumen fluid. The microbial population including bacteria, ciliated-associated methanogen, four different groups of methanogens and Fibrobacter succinogenes were quantified by using the real-time PCR. Gas profiles including methane, carbon dioxide and hydrogen, and runinal fermentation characteristics were observed in vitro. The methane emission from samples with an addition of individual juices from pine needles, ginkgo leaves and 70% ethanol extract from ginko leaves was significantly lower (p<0.05, 27.1, 28.1 and 28.1 vs 34.0 ml/g DM) than that of the control, respectively. Total VFAs in samples with an addition of any of the plant extracts were significantly lower than that of the control (p<0.05) as well. The order Methanococcales and the order Methanosarcinales were not detected by using PCR in any incubated mixtures. The ciliate-associated methanogens population decreased from 25% to 49% in the plant extacts as compared to control. We speculate that the supplementation of juice from pine needles and ginkgo leaves extract (70% ethanol extract) decreased the protozoa population resulting in a reduction of methane emission in the rumen and thus inhibiting methanogenesis. The order Methanobacteriales community was affected by addition of all plant extracts and decreased to less than the control, while the order Methanomicrobiales population showed an increase to more than that of the control. The F. succinogenes, the major fibrolytic microorganism, population in all added plant extracts was increased to greater than that of the control. In conclusion, pine needles and ginkgo leaves extracts appear to have properties that decrease methanogenesis by inhibiting protozoa species and may have a potential for use as additives for ruminants.
INTRODUCTION
Global warming due to increased atmospheric concentration of greenhouse gases including methane is a major interest in recent years. Methane emission from livestock industries, in particular from ruminants, is a significant contributor to this problem. Methane is a 23 times more potent green house gas than carbon dioxide (IPPC, 2007). Methane emission related to enteric fermentation from ruminants is mostly due to the optimal anaerobic conditions for facultative the growth of bacteria, protozoa and fungi. Methane emission in the rumen is considered an energetically wasteful process as it causes a substantial loss of 12% of the dietary energy intake (Johnson et al., 1995). Many chemical dietary additives have been examined for their potential to reduce methane emissions in the rumen, but were either toxic to host animals or caused side effects on beneficial ruminal microbes (Martin and Macy, 1985; Van Nevel and Demeyer, 1988; Moss, 1993; Van Nevel and Demeyer, 1996). The selection of bio-active compounds for the elimination of protozoa has been an approach studied with the aim of reducing methane emission from ruminants. However, the risk of using chemicals in animals destined for human consumption is of growing public concern. In the previous study, Kim et al. (2012) showed that plant extracts were good alternative candidates to manipulate rumen microbial diversity with the goal of reducing methane emission. This approach has been set as one of the most important goals for animal nutritionists (Teferedegne, 2000).
The objective of this study was to evaluate the in vitro effect of pine needle and ginkgo leaves extracts on ruminal fermentation, rumen methanogenesis and methane-related microbes.
MATERIALS AND METHODS
In vitro incubation
Animal:
A fistulated Holstein cow of 500 kg body weight was used as a donor of rumen fluid. Timothy and commercial concentrate (TDN; 73.5%, crude protein; 19%, crude fat; 3%, crude fiber; 12%, crude ash; 10%, Ca; 0.8%, P; 1.2%, AMINOTEK, CJ feeds) in the ratio of 60:40 were fed at 2% of body weight twice a day. Water and mineral-vitamin block were allowed ad libitum.
Rumen fluid:
The rumen fluid was collected from the fistulated Holstein cow before morning feeding. Rumen samples were collected in a bottle, previously kept warm and filled with O2 free-CO2 gas, carried to the laboratory, and filtered through four layers of cheesecloth before mixing with buffer maintained at 39°C and under O2-free N2 gas.
Preparation of extract:
Pine (Pinus densiflora) needles and ginkgo (Ginkgo biloba L.) leaves extracts were prepared in 70% ethanol at 100 g/300 ml of solvent. The flasks of 70% ethanol were stoppered and incubated at 39°C on a rotary shaker (HBS-201SL, HANBAEK, Korea) for 24 h. Juice from pine needles and ginkgo leaves were prepared by extraction from crushed leaves. The extracts were then filtered through filter paper (Whatman No. 1). The filtrates were then collected and the solvent, 70% ethanol, removed using a rotary evaporator, and stored at 4°C for further use.
In vitro incubation:
Four extracts were prepared for their effects on ruminal methanogenesis and fermentation. Treatment designations were non-additives (Control), T1 (5% of juice from pine needles), T2 (5% of 70% ethanol extract from pine needles), T3 (5% of juice from ginkgo leaves) and T4 (5% of 70% ethanol extract from ginkgo leaves). Rumen liquor was filtered through four layers of cheesecloth before mixing with buffer maintained at 39°C. The 30 ml of rumen fluid-buffer mixture, comprising McDougall buffer and rumen liquor in the ratio of 4 to 1, was dispensed anaerobically into 120 ml serum bottles, filled with O2-free N2 gas, containing 0.3 g of timothy substrate and plant extracts (5% of total volume of rumen fluid-buffer mixture (v/v), respectively), and then capped with a rubber stopper. The serum bottles were held in a shaking incubator (HBS-201SL, HANBAEK, Korea) at 39°C for 24 h. The in vitro experiment were evaluated in triplicate run for analysis of gas proflies and VFAs.
Analyses
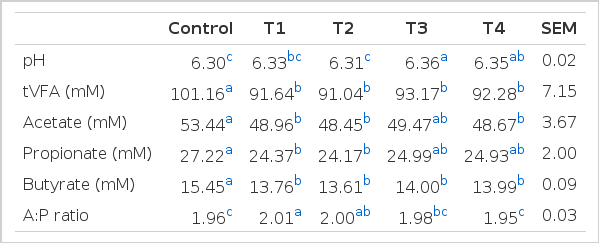
Total gas production was measured by the assay of Theodorou et al. (1994). A detachable pressure transducer and a digital readout voltmeter (Laurel Electronics, Inc., CA USA) were used to measure the headspace gas pressure of fermenting cultures. The transducer was modified to link to the inlet of a disposable Luer-lock three-way stopcock in other to measure total gas production in the system (Theodorou, 1994). Gas pressure in the headspace was read from the display unit after insertion of a hypodermic syringe needle through the butyl rubber stopper above the culture medium. The headspace gas in the serum bottle was collected for analysing the composition of gases, focusing on methane, carbon dioxide and hydrogen. These three gases were analysed using gas chromatography (GC-2010, Shimadzu, Japan) equipped with column (Shincarbon ST. 50/80, Shimadzu, Japan). The incubated inoculum was subsampled for the analysis of pH (Mettle-Toledo, CH/MP220), volatile fatty acid (VFA) concentration and gDNA extraction. VFA analysis was performed with a gas chromatography (GC-2010, Shimadzu, Japan) as described by Erwin et al. (1961).
DNA extraction, PCR primers and Real-time PCR
DNA extraction:
TissueLyser (Retsch; QIAGEN, Valencia, CA), a high-speed reciprocal shaker which retains samples in screw-capped tubes containing silica beads, was used for DNA extraction. Total nucleic acid was extracted from the incubated rumen samples by using the modified bead-beating protocol with the QIAamp DNA mini kit (250) (QIAGEN, USA). For samples a 1.0 ml aliquot was taken from the 30 ml incubated mixture using a wide bore pipette so as to ensure a homogenous sample containing fluid and digesta. Nucleic acid concentrations were measured by using a NanoDrop Spectrophotometer (ND-1000, USA).
PCR primers:
The PCR primer sets used in this study for amplification of general bacteria, Fibrobacter succinogenes, archaea, methanogenic archaea, ciliate-associated methanogen and four different groups of methanogens (the order Methanobacteriales, the order Methanomicrobiales, the order Methanosarcinales and the order Methanococcales) were the same as referenced by Denman and McSweeney (2006), Tajima et al. (2001), Ohene-Adjei et al. (2008), Luton at al. (2002), Medlin et al. (1998) and Yu et al. (2005), respectively as shown in Table 1.
Real-time PCR:
PCR assays for the enumeration of ciliate-associated methanogens, four different groups of methanogens and cellulolytic bacterial species were performed according to the methods described by Denman and McSweeney (2006) and Denman et al. (2007) on a Rotor-Gene Real-Time PCR Machine (Crobett life science, Australia) using the iQ SYBR Green Supermix (Bio-Rad Inc. USA). The values of cycle threshold (Ct) after real-time PCR were used to determine fold change (number of fold difference) of different microbial population relative to control without plant extract and plant extracts treatments. Abundance of these microbes was expressed by the equation: relative quantification = 2−ΔCt(Target)- ΔCt(Control), where Ct represents threshold cycle. All quantative (q) PCR reaction mixture (final volume of 25 μl) contained forward and reverse primers, the iQ SYBR Green Supermix and DNA template ranging from 10 ng to 100 ng. A negative control without the template DNA was used in every qPCR assay for each primer. The PCR conditions for the amplification of target DNA, including the annealing and the extension temperature, were tested before application as shown in following references (Table 1).
Statistical analysis
Data were analyzed using the general linear model (GLM) procedure of the Statistical Analysis System Institute, Inc. (SAS, 2002). Differences among means were tested for significance using Duncan’s multiple range test of SAS (2002).
RESULTS AND DISCUSSION
Ruminal fermentation characteristics
The in vitro total gas production, methane emission and carbon dioxide production are shown in Table 2. Pine needles and ginkgo leaves extracts affected total gas production as well as methane emission and carbon dioxide production in the rumen. Total gas production in samples with added pine needles and ginkgo leaves extracts were lower than that of the control (Table 2). The in vitro ruminal fermentation might be affected by the dosage of plant extracts as well as the extract solvents (Patra et al., 2010). Pfister et al. (1992) also suggested that 15% to 30% pine needles in cattle diets appears to be the threshold level for toxic effects on ruminal fermentation. However, adverse effects of plant extracts on digestive performance in cattle should be considered for further studies. The methane emission in samples with pine needles juice and ginkgo leaves extracts added (70% ethanol extract and juice) were significantly lower (p<0.05, 27.1, 28.1 and 28.1 vs 34.0 ml/g DM) than that of the control. The pH in addition of ginkgo leaves extracts was significantly affected (p<0.05, Table 3). Total VFAs in addition of all plant extracts were significantly lower than that of the control (p<0.05, Table 3). Although H2 was analyzed by gas chromatography (GC-2010, Shimadzu, Japan), it was not detected. This possibly indicates that H2 products might have been utilised as a substrate for transfer of CH4 by methanogens in the rumen. Thus, H2 could be of critical concern to the microbial ecosystem in ruminants. Also, the formation of propionate from succinate would result in a lower availability of H2 for methanogenesis. H2 produced during enteric fermentation is the precursor of methane in ruminants and the regulation of H2 rather than methane is the key to the control ruminant methane emission. This study showed that plant extracts, in particular ginkgo leaves extract can potentially influence methanogenesis, resulting in a reduction in methane emission from the rumen.
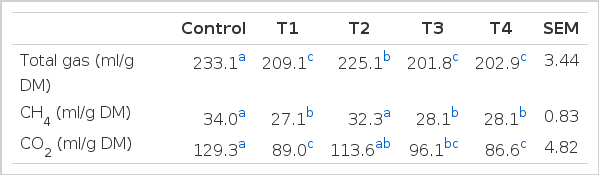
The effect of pine needles and ginkgo leaves extracts on in-vitro total gas, CH4 and CO2 productions after 24 h incubation
Microbial diversity related methane emission in the rumen
Pine needles and ginkgo leaves extracts similar to those used in the present experiment are known to have anthelmintic, antibacterial and antioxidant activities, and have been used as medicinal materials (Choi et al., 1997; Kim and Shin, 2005; Lee et al., 2006). The secondary compounds from pine needles and ginkgo leaves, such as flavonoids, might manipulate ruminal fermentation and inhibit microbes, especially protozoa, in the rumen (Figure 2). Pine needles and ginkgo leaves have been shown to possess strong antibiotic characteristics against various bacteria (Choi et al., 1997; Lee et al., 2006), in particular, ginkgo leaves extract is well known for having a natural insecticidal effect and perhaps have anthelmintic and antibacterial properties. Antioxidative activities of pine (Pinus denstifora) needle extracts have been tested in vitro experimental models by Yoo et al. (2004). The concentration of total polyphenolic compound of water extracts from pine needles was 1.61%. This finding is in agreement with other studies where the concentration of total polyphenolic compounds are 1.80% (Kang et al., 1995), and 1.32% (Kim et al., 2000), respectively. The microbial population related to methanogens determined with PCR is shown in Figure 1. The order Methanococcales and the order Methanosarcinales were not detected in this study in any of the incubated mixtures. Behlke (2007) also could not detect these two orders (the order Methanococcales and the order Methanosarcinales) and reported that either these two orders were not in the rumen or they were less than the detection limit. The ciliate-associated methanogens population in samples containing any of the plant extracts was affected (Figure 2). The Ciliate-associated methanogens population decreased from 25% to 49% in the plant extracts compared to the control. As several rumen ciliates and methanogens are known to be symbionts (Vogels et al., 1980; Stumm et al., 1982), the result of this study also showed that the supplementation of pine needles juice and ginkgo leaves extract (70% ethanol extract) decreased the protozoa population resulting in reduction of methane emission in the rumen (Figure 2; Table 2) and thus inhibiting methanogenesis. In a previous study, Broudiscou et al. (2000) screened plant extracts, selected for their high flavonoid content, on their action on methanogenesis, fermentation pattern and protozoa numbers. Broudiscou et al. (2000) reported that plant extracts slightly inhibited methanogenesis. The order Methanobacteriales community was affected by addition of all plant extracts (Figure 3). Methanobacteriales community decreased more than that of the control with addition of all plant extracts, while the order Methanomicrobiales population increased to more than that of the control (Figure 4). Plant extracts have been placed under the spotlight as a potentially useful alternative feed additives source, but there is limited data on ruminal fermentation. Compounds of selected plant extracts with known medicinal properties have been reported to interact with micro-organisms in other ecosystems, in a positive as well as in a negative way. The in vivo study by Jo et al. (1997) was conducted to evaluate the value of forages (alfalfa, acacia leaf, orchardgrass and pine needles) on feed intake and digestibility in the Korean native goat. The result of that in vivo study showed that dry matter intake and dry matter digestibility of pine needles were lower than other forages, although the result of this in vitro study did not show an adverse effect on the F. succinogenes population by the addition of plant extracts. The F. succinogenes population in all added plant extracts treatments increased to greater than that of the control (Figure 5).

Relative quantification analysis of ciliate-associated methanogen community in vitro ruminal fermentation following the addition of pine needles and ginkgo leaves extracts after 24 h incubation (Control: no additive, T1: 5% of juice from pines needle, T2: 5% of 70% ethanol extract from pine needles, T3: 5% of juice from ginkgo leaves, T4: 5% of 70% ethanol extract from ginkgo leaves).

The PCR amplification of microbial population - related to methanogens in the rumen (a: Archaea, b: Methanococcales, c: Methanosarcinales, d: Methanobacteriales, e: Methanomicrobiales).
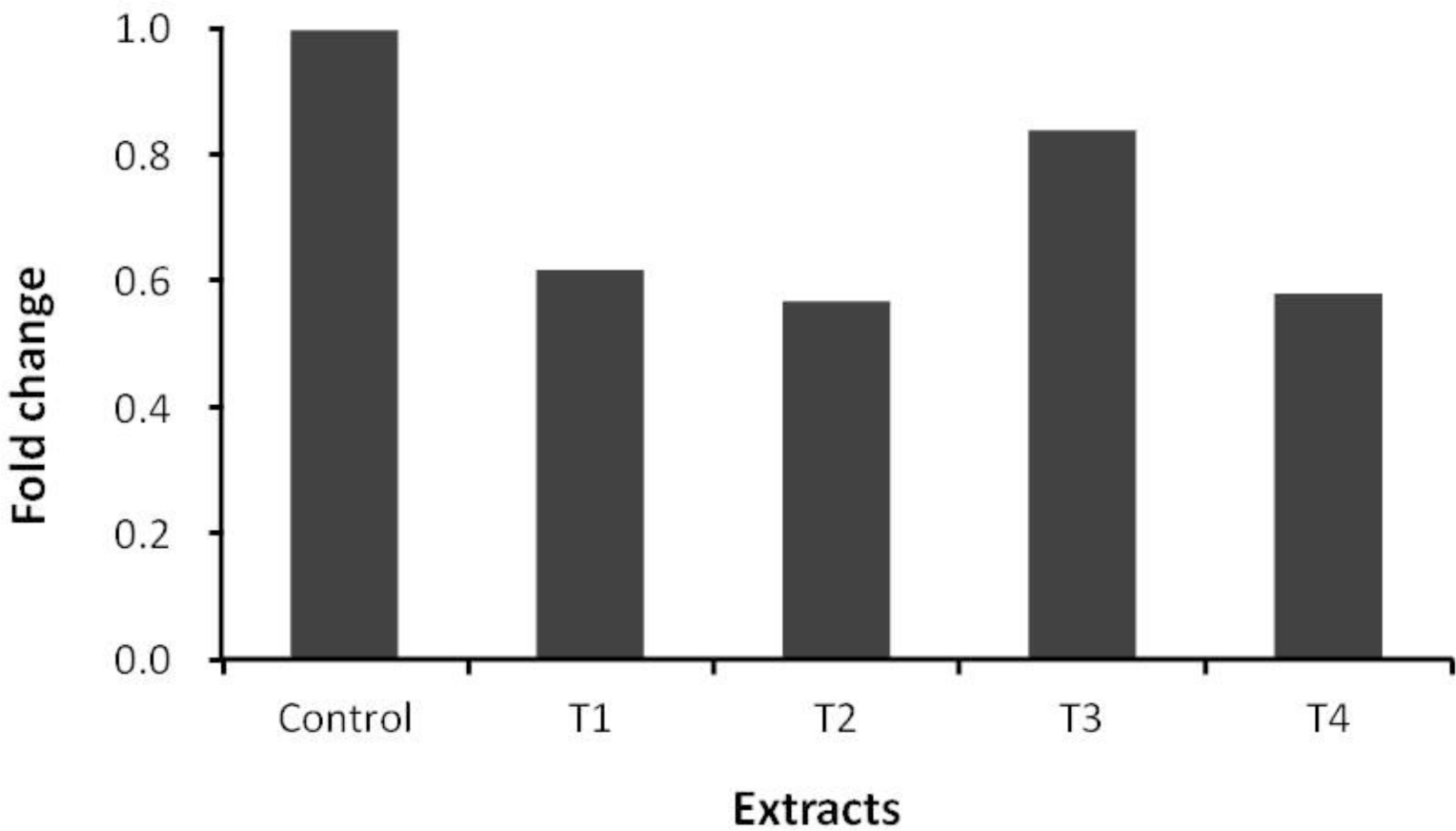
Relative quantification analysis of Methanobacteriales population in vitro ruminal fermentation following the addition of pine needles and ginkgo leaves extracts after 24 h incubation (Control: no additive, T1: 5% of juice from pines needle, T2: 5% of 70% ethanol extract from pine needles, T3: 5% of juice from ginkgo leaves, T4: 5% of 70% ethanol extract from ginkgo leaves).
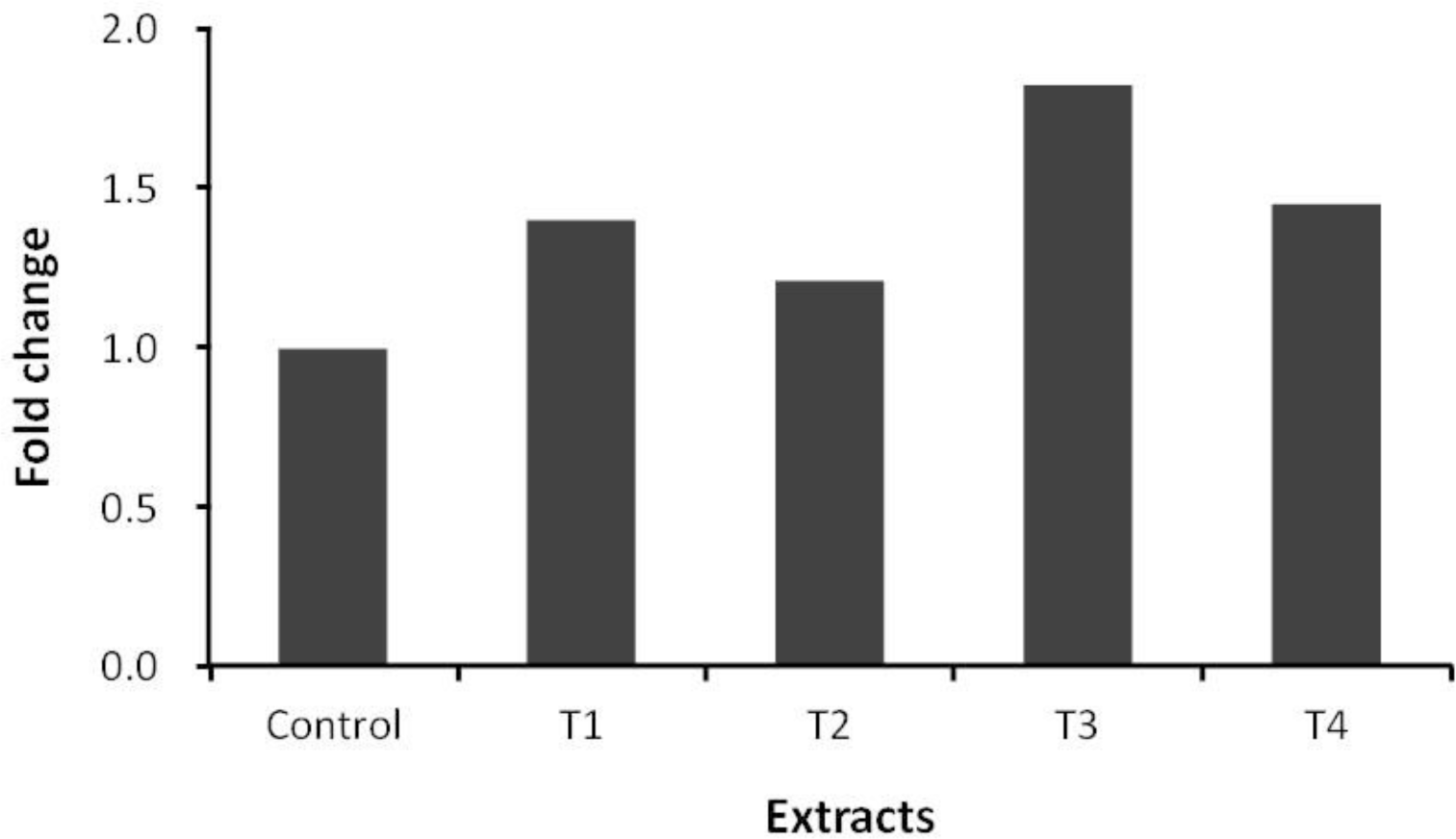
Relative quantification analysis of Methanomicrobiales population in vitro ruminal fermentation by following the addition of pine needles and ginkgo leaves extracts after 24 h incubation (Control: no additive, T1: 5% of juice from pines needle, T2: 5% of 70% ethanol extract from pine needles, T3: 5% of juice from ginkgo leaves, T4: 5% of 70% ethanol extract from ginkgo leaves).
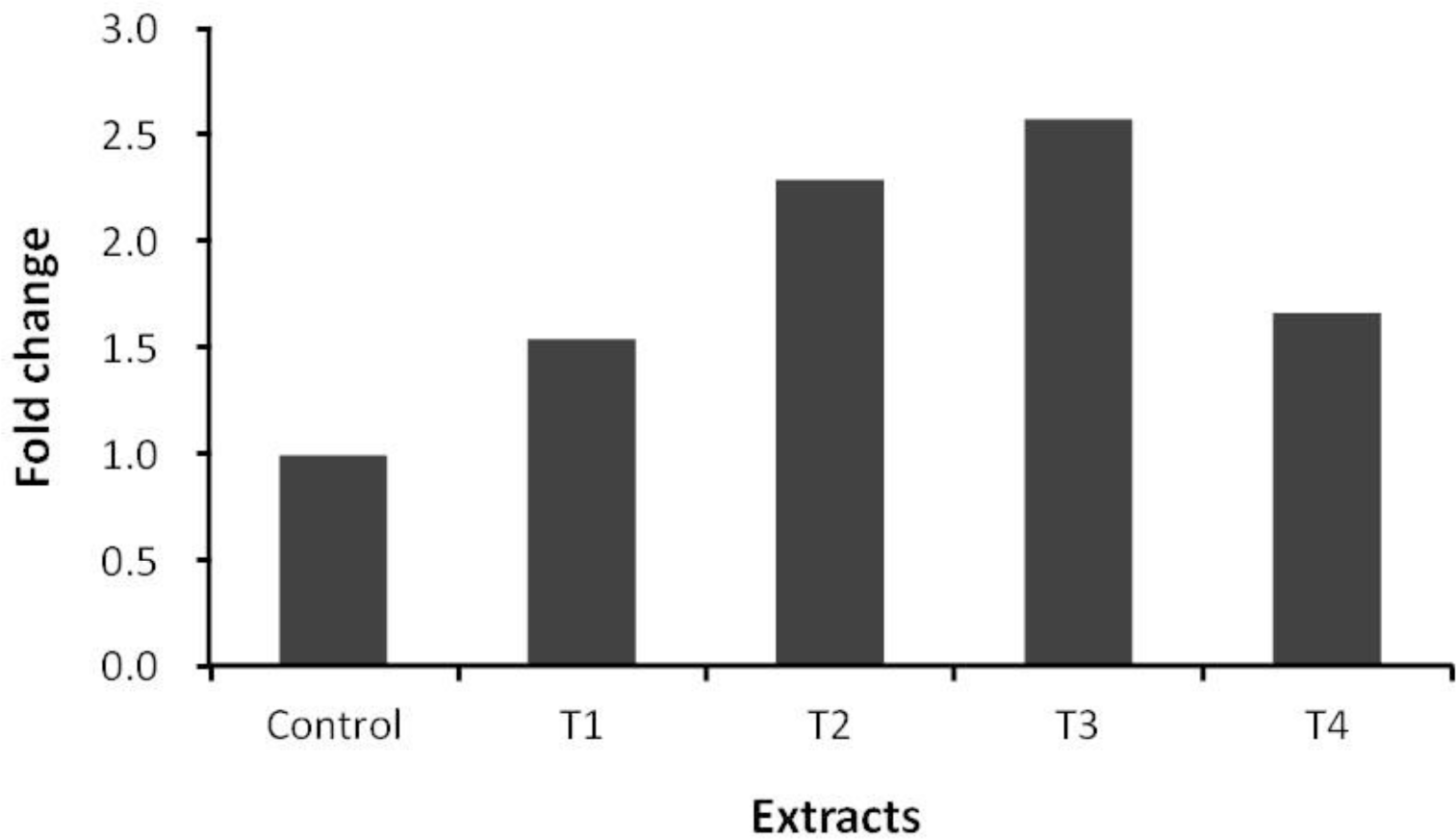
Relative quantification analysis of Fibrobacter succinogenes diversity in vitro ruminal fermentation following the addition of pine needles and ginkgo leaves extracts after 24 h incubation (Control: no additive, T1: 5% of juice from pines needle, T2: 5% of 70% ethanol extract from pine needles, T3: 5% of juice from ginkgo leaves, T4: 5% of 70% ethanol extract from ginkgo leaves).
Plant extracts (pine needle and ginkgo leaves extracts) with known to medicineal properties used in this study were found to have properties that decreased ruminal methanogenesis and consequently, there is a need to consider the dosage of plant extracts and whether there are adverse effects of plant extracts on aspects of the ruminal fermentation in further studies.
Acknowledgements
This research was supported by Bio-industry Technology Development Program, Ministry for Food, Agriculture, Forestry and Fisheries, Korea, and E. T. Kim was supported by a scholarship from the BK21 Program, the Ministry of Education, Science and Technology, Korea.