Evaluation of Optimum Dietary Threonine Requirement by Plasma Free Threonine and Ammonia Concentrations in Surgically Modified Rainbow Trout, Oncorhynchus mykiss
Article information
Abstract
This study was carried out to evaluate the dietary threonine requirement by measuring the plasma free threonine and ammonia concentrations in rainbow trout, Oncorhynchus mykiss after dorsal aorta cannulation. A total of 70 fish (average initial weight 506±8.2 g) were randomly distributed into each of the 14 net cages (5 fish/cage). After 48 hours (h) of feed deprivation, each group was intubated at 1% body weight with one of the seven L-amino acid based diets containing graded levels of threonine (0.42%, 0.72%, 0.92%, 1.12%, 1.32%, 1.52%, or 1.82% of diet, dry matter basis). Blood samples were taken at 0, 5, and 24 h after intubation. Post-prandial plasma free threonine concentrations (PPthr) of fish 5 h after intubation with diets containing 1.32% or more threonine were significantly higher than those of fish intubated with diets containing 1.12% or less threonine (p<0.05). Post-absorptive free threonine concentrations (PAthr) after 24 h of intubation of the fish with diets containing 0.92% or more threonine were significantly higher than those of fish intubated with diets containing 0.72% or less threonine. Post-prandial plasma ammonia concentrations (PPA, 5 h after intubation) were not significantly different among fish intubated with diets containing 1.12% or less threonine, except the PPA of fish intubated with diet containing 0.42% threonine. Broken-line model analyses of PPthr, PAthr, and PPA indicated that the dietary threonine requirement of rainbow trout should be between 0.95% (2.71) and 1.07% (3.06) of diet (% of dietary protein on a dry matter basis).
INTRODUCTION
Essential amino acids are required by all fish species as well as mammals for normal growth and metabolic functions. Knowledge of the dietary essential amino acids requirement of a fish species is of great importance in formulating cost-effective feeds. Threonine is the third limiting amino acid after methionine and lysine and it must be provided as threonine or threonine containing proteins in the diet. Even for some fish species, threonine could be potentially marginal or limiting in feed formulations especially when plant proteins are used as a substitute of fish meal. Threonine deficiency resulted in retarded growth and reduced feed intake in some species such as Japanese flounder, Paralichthys olivaceus (Alam et al., 2003) and rainbow trout, Oncorhynchus mykiss (Rodehutscord et al., 1995). Therefore, it is important to determine the threonine requirements of cultured fish species for normal growth and feed utilization. Dietary threonine requirements have been estimated for commonly cultured species of fish, and wide variations have been reported (0.6% to 2.0% of diet), even among salmonids (0.6% to 1.23%) (Wilson, 2002).
The threonine requirements of different species of fish were determined by feeding trials which are expensive and last for several weeks to months. On the other hand, determination of amino acid requirements by measuring plasma free amino acid (PFAA) and ammonia concentrations can provide useful information on the amino acid requirements within weeks. This method has been employed in several species and results are similar to those obtained by feeding trials (Bae et al., 2011). Furthermore, PFAA concentrations had been used to investigate amino acids metabolism and to evaluate the quality of dietary protein in rainbow trout (Schuhmacher et al., 1997); common carp (Dabrowski, 1982) and Atlantic salmon, Salmosalar (Sunde et al., 2003).
Evaluation of PFAA concentrations has led to the discovery of genetic defects of amino acid metabolism as a result of primary renal or liver disease and the effects of amino acid deficiencies, imbalances and toxicities on amino acid metabolism (Zicker and Rogers, 1990). The relationships between the concentration of PFAA and intake of dietary amino acids have been investigated by several researchers (Bai et al., 2003). Plasma free amino acid concentrations of fish fed with graded levels of the amino acid had been used in several experiments in an attempt to confirm requirement values determined by growth trials. Although the effects of dietary protein or amino acid mixtures on PFAAs concentrations in rainbow trout (Schuhmacher et al., 1997) and sea bass, Dicentrarchus labrax (Thebault, 1985) had been reported, the complete dose response relationships for amino acids have not been investigated. In our previous studies, requirements for dietary essential amino acids such as arginine, methionine, lysine and valine were obtained in rainbow trout using PFAA in surgically modified rainbow trout (Bae et al., 2011). As a part of our consecutive studies on the essential amino acid requirements of rainbow trout, the purpose of the present investigation was to estimate the dietary threonine requirement based on the plasma free threonine and ammonia concentrations in dorsal aorta cannulated rainbow trout.
MATERIALS AND METHODS
Experimental fish
The experimental rainbow trout (Oncorhynchus mykiss) were obtained from the Ewhajung trout farm, Sangju City, South Korea. Fish were fed with a commercial rainbow trout diet (Woo-sung Feed Co. Ltd., Daejon, Korea) for 72 h until they recovered from the operation of dorsal aorta cannulation. At the beginning of the feeding trial a total of 70 rainbow trout (average initial weight 506±8.2 g) were carefully selected from the stock tanks and randomly distributed into each of the 14 net cages (5 fish/cage). Each cage (1.3×1.3×1.3 m) was then randomly assigned to one of two replicates of seven dietary treatments and the cages were placed in a flow-through concrete raceway with a continuous water flow of 60 L/min. Supplemental aeration was also provided to maintain the dissolved oxygen at 6.5±0.5 mg/L, and water temperature was maintained at 16±0.4°C
Preparation of experimental diets
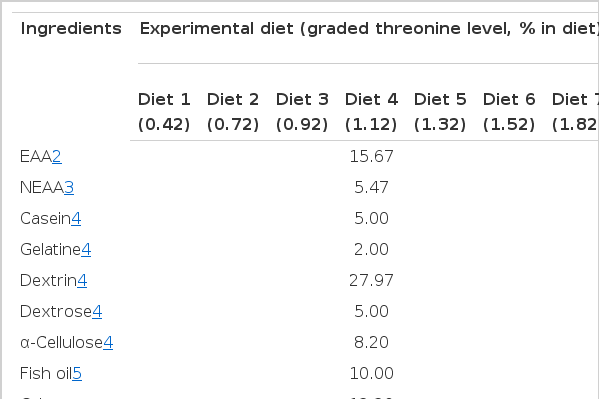
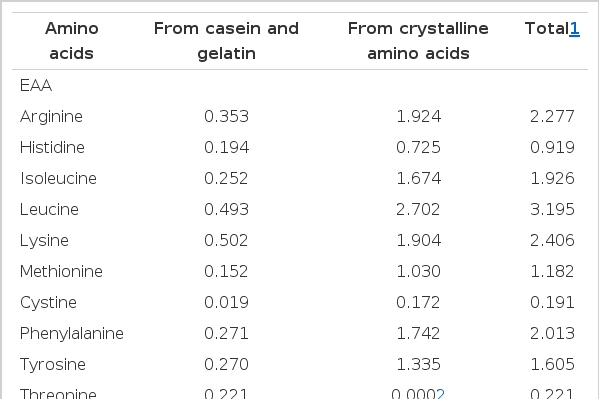
A basal diet containing 36.6% crude protein (29.6% crystalline amino acids mixture, 5% casein and 2% gelatin) was formulated by the modification of Kim (1997). Seven experimental diets were formulated to contain graded levels of threonine (0.42%, 0.72%, 0.92%, 1.12%, 1.32%, 1.52%, or 1.82% of diet, on a dry matter basis) based on the basal diet. Equal amounts of aspartic acid and glutamic acid by weight were substituted for the graded amounts of threonine in the basal diet to maintain the isonitrogenous of the seven experimental diets. Formulation and amino acid composition of the experimental diet are shown in Table 1 and 2, respectively. The ingredient mixtures without oil were stored at −80°C until used and the diets were prepared by adding fish oil (10% of diet) and water (dry ingredients plus 0.4 parts of distilled water per diet) before intubation.
Dorsal aorta cannulation and intubation
Rainbow trout were anaesthetized with 200 mg/L 3-aminobenzoic acid ethyl ester methanesulfonate (MS-222, Sigma Chemical Company, St. Louis, MO, USA) for 3 to 5 minutes(m) by placing the fish on a V-shaped table while their gills were irrigated continuously with 16°C water containing 100 mg/L MS-222 during the operation. A 50 cm long PE 50 tubing (Clay Adams PE 50 tubing, Parsippany, NJ, USA) with a bubble of about 5 to 6 cm on one end was used for the dorsal aorta cannula after it was flushed with the heparinized Cortland saline solution (Houston, 1990). A 13-gauge needle was used to pierce a hole on the right nostrum of fish (ventral side up) as an exit for the cannula and a 19-gauge needle was used to bore a small hole in the roof of the mouth of fish at the mid-line behind the third gill arc at a 30° angle, and a piano wire was inserted into the PE 50 tubing as a guide. The proper insertion was verified by the observation of a slow blood flow into the PE 50 after the wire was withdrawn from the cannula.
A 3 mL syringe with a 23-gauge needle was used to remove air and blood clot and the cannula was flushed with the heparinized saline solution. The cannula was sutured behind the bubble on the roof of the mouth, led out from the right nostrum, plugged with a head pin, and sutured at the dorsal fin (Bai et al., 2003; Bae et al., 2011). Thirty five dorsal aorta cannulated rainbow trout were randomly distributed into seven groups including five fish per group and intubated with one of the seven experimental diets per group. After 24 h feed deprivation, the fish were anesthetized with 200 mg/L MS-222 and fed the experimental diet by the stomach intubation method using a 3 mL syringe.
Sample collection and analysis
Five fish per group were anesthetized with 200 mg/L MS-222 and blood was sampled using a 3 mL syringe from each fish at 0, 5, and 24 h after intubation of the experimental diets. Plasma samples were prepared by centrifugation at 3,000g for 10 min at room temperature. For deproteinization, plasma samples were mixed with a 10% 5-sulphosalicylic acid solution in the ratio of 4:1 (v/v), cooled on ice for 30 m and re-centrifuged. The protein-free supernatant was dissolved in pH 2.2 lithium citrate sample dilution buffer in the ratio of 1:1 (v/v), and the samples were stored at −80°C until analysis. Plasma free amino acids were quantified using a S433 amino acid analyzer (Sykam, Gilching, Germany) using the ninhydrin method. Plasma ammonia concentrations were analyzed using the Berthelot reaction (Sigma, USA).
Statistical analysis
Data were subjected to one way analysis of variance test using Statistix 3.1 (Analytical Software, St Paul, MN, USA). When a significant treatment effect was observed, a Least Significant Difference test was used to compare means. Treatment effects were considered significant at p<0.05. The breakpoints for post-prandial plasma free threonine concentrations (PPthr), post-absorptive free threonine concentrations (PAthr) and post-prandial plasma ammonia concentrations (PPA) were estimated by using the broken line model of Robbins et al. (1979).
RESULTS
Plasma free threonine concentrations
Results of PPthr (5 h after intubation) and PAthr (24 h after intubation) in dorsal aorta cannulated rainbow trout intubated with diets containing seven graded levels of threonine were summarized in Table 3. Post-prandial plasma free threonine concentrations of fish intubated with the diets containing 1.32% or more threonine were significantly higher than those of fish fed diets containing 1.12% or less threonine (p<0.05). There were no significant differences in PPthr among fish intubated with diets containing 1.32% or more threonine.
Broken-line model analysis on the basis of PPthr as shown in Figure 1 indicated that the dietary threonine requirement of trout could be 1.07% (3.06) of diet (% dietary protein on a dry matter basis). Post-absorptive plasma free threonine concentrations of fish intubated with the diets containing 0.92% or more threonine were significantly higher than those of fish intubated with diets containing 0.72% or less threonine (p<0.05). There were no significant differences in PAthr among fish intubated fed with diets containing 0.92% or more threonine. Broken-line model analysis on the basis of PAthr in Figure 2 indicated that the dietary threonine requirement of trout could be 0.97% (2.77) of diet (% dietary protein on a dry matter basis).
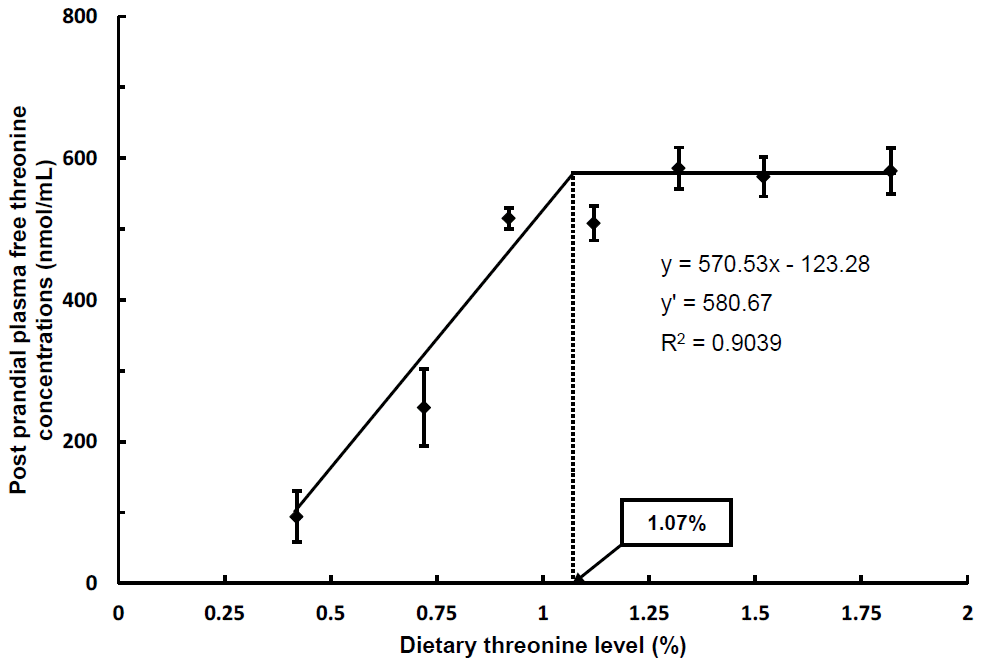
Broken line analysis of post-prandial plasma threonine concentrations (nmol/mL) in rainbow trout fed graded levels of dietary threonine. Values of the X-axis are the threonine levels in the experimental diets. Values are means±standard deviation of 5 replicates. Y = 570.53x–123.28, Y ′ = 580.67, R2 = 0.9039.
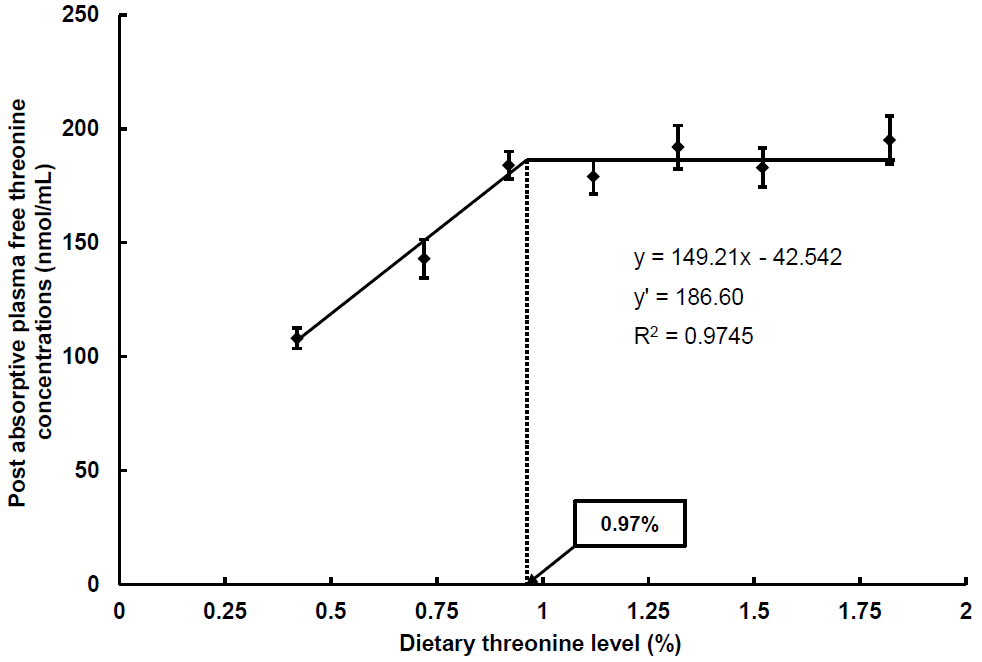
Broken line analysis of post-absorptive plasma threonine concentrations (nmol/mL) in rainbow trout fed graded levels of dietary threonine. Values of the X-axis are the threonine levels in the experimental diets. Values are means±standard deviation of 5 replicates. Y = 149.21x–42.542, Y ′ = 186.6, R2 = 0.9745.
Plasma ammonia concentrations
The PPA (5 h after intubation) and post-absorptive plasma ammonia concentrations (PAA, 24 h after intubation) in rainbow trout after intubated with graded levels of dietary threonine were shown in Table 3. Post-prandial plasma ammonia concentrations were not significantly different among fish intubated diets containing 1.12% or less threonine, except for the PPA of fish intubated with diet containing 0.42% threonine (p< 0.05). However, PPA linearly increased with dietary threonine levels from 1.12% to 1.82%. There was no significant difference in PPA of fish intubated with diets containing threonine between 1.52% and 1.82% threonine (p< 0.05). Post-absorptive plasma ammonia concentrations in the present study were not significantly different among the groups. Broken-line model analysis on the basis of PPA (Figure 3) indicated that the dietary threonine requirement of rainbow trout could be 0.95% (2.71) of diet (% dietary protein on a dry matter basis).
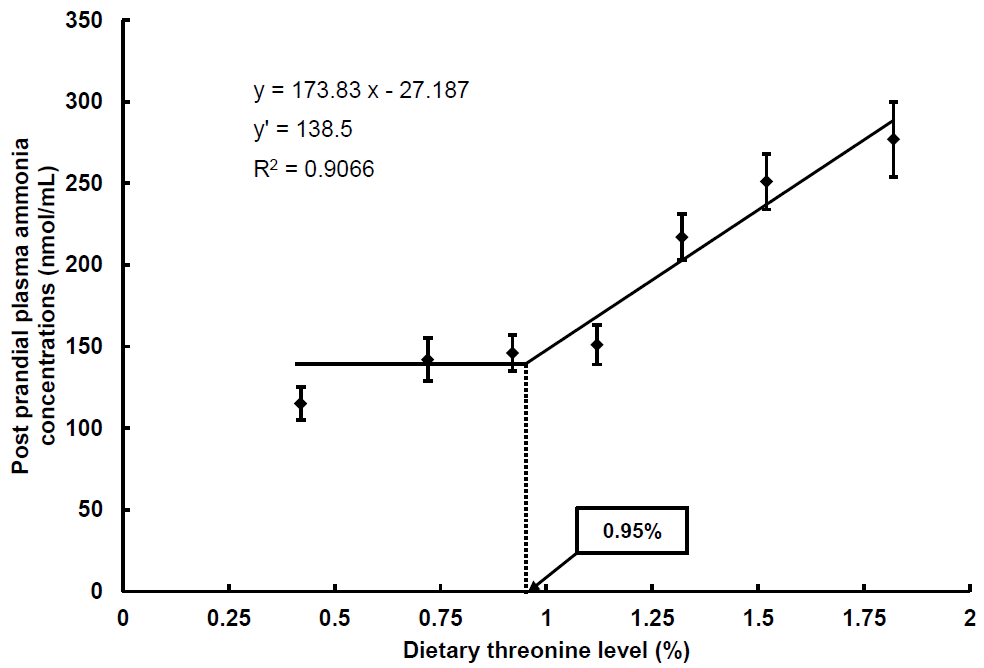
Broken line analysis of post-prandial plasma ammonia concentrations (nmol/mL) in rainbow trout fed graded levels of dietary threonine. Values of the X-axis are the threonine levels in the experimental diets. Values are means±standard deviation of 5 replicates. Y = 173.83x–27.187, Y ′ = 138.5, R2 = 0.9066.
DISCUSSION
In the present study, the dietary threonine requirement for the rainbow trout based on plasma free threonine concentrations (PPthr and PAthr) could be between 0.97% (2.77) and 1.07% (3.06) of diet (% dietary protein basis). In previous studies, the dietary threonine requirements of rainbow trout have been reported to be 1.36% (3.4) of diet (dietary protein) by Ogino (1980), 1.04% to 1.23% (3.1 to 3.7) by Rodehutscord et al. (1995) and 1.06% (2.6) by Bodin et al. (2008) based on the dose response or ideal protein. The highest value of 1.36% of diet reported by Ogino (1980) may have been overestimated because the feed intake value considered in the essential amino acid requirement calculation was underestimated (Kim et al., 1992). Recalculation of the relative threonine requirement from Ogino′s data, on the basis of an estimation of feed intake gives a value of 0.97% of diet which is more similar to the observed value for rainbow trout in the present study (Kim et al., 1992; Bodin et al., 2008).
When expressed as a percentage of dietary protein, results of the present study was similar to those reported for several other species, such as 2.6% to 3.0% of dietary protein in European sea bass (Tibaldi and Tulli, 1999); 2.7% to 3.20% in Atlantic salmon (Rollin et al., 2003) and 3.04% in chum salmon, Oncorhynchus keta (Akiyama et al., 1985). Somewhat lower threonine requirement values have been reported in other species of fish including 2.0% in coho salmon, O. kisutch (Halver and Shanks, 1960) and 2.28% in red drum, Sciaenopso cellatus (Boren and Gatlin, 1995); while higher levels of threonine in the dietary protein have been reported for white sturgeon, Acipenser transmontanus, 3.30% (Ng and Hung, 1995). These wide variations observed in the threonine requirements among different fish species may be due to the differences in dietary protein sources, the reference protein whose amino acid pattern is being imitated, diet formulation, size and age of fish, genetic differences, feeding practices and rearing conditions (Rodehutscord et al., 1997).
Zhou et al. (2007) reported that the dietary lysine requirements of fish species that have high protein requirements, such as marine fish and freshwater carnivorous fish, are higher than those of species that have low protein requirements including omnivorous and herbivorous fish. However, the differences in the dietary threonine requirement values listed above indicated that there was no apparent distinction in food habits or environmental needs which could help to give a clear classification of dietary threonine requirements for various fish species.
The effects of dietary threonine intake on PPthr and PAthr were dependent upon the relative adequacy of the dietary threonine supply, increasing up to the dietary threonine requirement levels with no further increase above the requirement values. In other words, a breakpoint was observed in the plasma concentration value at 1.07% (PPthr) or 0.97% (PAthr) dietary threonine. Similar results have been reported for juvenile yellowtail, Seriolaquin queradiata (Ruchimat et al., 1997) and rainbow trout, (Bae et al., 2011). The pattern of the post-prandial or absorptive plasma free amino acid concentrations of fish intubated with graded levels of the amino acid had been used in several studies in an attempt to confirm the required values assessed by growth (Thebault et al., 1985; Tibaldi and Tulli, 1999).
In the present study, the dietary threonine requirement of rainbow trout based on the PPA (5 h after intubation) could be 0.95% (2.71) of diet (% dietary protein basis). Post-prandial plasma free ammonia concentration was significantly affected by the dietary threonine levels. However, PAA (24 h after intubation) were not affected by the dietary threonine level. Post-prandial plasma free ammonia concentration remained low and virtually constant, but increased linearly when dietary threonine level was higher than the required level. Similar observations were reported that PPA was affected by the dietary arginine, lysine, methionine and valine levels in rainbow trout (Bae et al., 2011). This observation could be attributed to an increase in net protein synthesis with the increase in dietary amino acids. The ammonia excretion was related to the intake of protein (Koshio et al., 1993) and dietary amino acids balance (Kaushik et al., 1988). Considering protein and amino acid metabolism, results of the present study indicated that the dietary threonine requirement in rainbow trout can be estimated based on PPA. Furthermore, the results showed that estimated threonine requirement value based on PPA was lower than those based on PPthr and PAthr. Future studies would look into the cause of the discrepancy in these values.
The patterns of most plasma free amino acid concentrations from force-fed rainbow trout with and without dorsal aorta cannulation were similar, and the dorsal aorta cannulation did not affect their patterns (Bae et al., 2011). These results suggested that dorsal aorta cannulated rainbow trout is able to recover from the operation after a 48 h recovery period. Furthermore, the dorsal aorta cannulation would allow repeated sampling on the same individual fish to study nutrient metabolism in the blood circulation.
Plasma free amino acid concentrations may be useful in determining the optimum blood sampling time and also in evaluating protein quality and essential amino acid requirements. Furthermore, this technique can be used in pharmaceutical, toxicological and nutritional research in rainbow trout. Results in this and other studies including measurement of plasma free amino acids and ammonia are similar to those obtained by feeding trial. This suggests the dorsal aorta technique can be used to estimate the requirement of other amino acids by rainbow trout.
In conclusion, quantitative estimation of the amino acid requirement using the broken-line model analysis of plasma free amino acid or ammonia concentrations (post-prandial or absorptive) is an acceptable method for the determination of essential amino acid requirements of fish species. Broken-line model analyses on the basis of PPthr, PAthr, and PPA concentrations indicated that the dietary threonine requirement of rainbow trout could be between 0.95% (2.71) and 1.07% (3.06) of diet (dietary protein basis).
ACKNOWLEDGMENTS
This research was supported by the grants from the National Fisheries Research and Development Institute (RP-2013-AQ-180) and Feeds and Foods Nutrition Research Center (FFNRC) at Pukyong National University, Busan, Republic of Korea.