Protective effects of Bacillus subtilis against Salmonella infection in the microbiome of Hy-Line Brown layers
Article information
Abstract
Objective
This study investigated the effects of Bacillus subtilis CSL2 (B. subtilis CSL2) administration before Salmonella challenge on the fecal microbiota and microbial functionality of Hy-line Brown (HLB) laying hens.
Methods
Fecal samples were collected from control (CON), Salmonella-infected (SAL) and Salmonella-infected, probiotic-treated (PRO) groups before and after Salmonella challenge for microbiome analysis using 16S rRNA gene pyrosequencing.
Results
Infection with Salmonella led to decreased microbial diversity in hen feces; diversity was recovered with Bacillus administration. In addition, Salmonella infection triggered significant alterations in the composition of the fecal microbiota. The abundance of the phylum Firmicutes decreased while that of Proteobacteria, which includes a wide variety of pathogens, increased significantly. Bacillus administration resulted in normal levels of abundance of Firmicutes and Proteobacteria. Analysis of bacterial genera showed that Salmonella challenge decreased the population of Lactobacillus, the most abundant genus, and increased populations of Pseudomonas and Flavobacterium genera by a factor of 3 to 5. On the other hand, Bacillus administration caused the abundance of the Lactobacillus genus to recover to control levels and decreased the population of Pseudomonas significantly. Further analysis of operational taxonomic units revealed a high abundance of genes associated with two-component systems and secretion systems in the SAL group, whereas the PRO group had more genes associated with ribosomes.
Conclusion
The results of this study indicate that B. subtilis CSL2 administration can modulate the microbiota in HLB laying hens, potentially acting as a probiotic to protect against Salmonella Gallinarum infection.
INTRODUCTION
Salmonella enterica (S. enterica) is a pathogen that causes salmonellosis, an acute inflammation of the intestines, in poultry, including broilers and laying hens [1]. In humans Salmonella infection is highly contagious via fecal-oral transmission, consumption of undercooked chicken meat, or mishandling of raw, contaminated poultry-derived products [1]. Poultry-derived products can be infected with many serovars of S. enterica, including S. Typhimurium, S. Typhi, S. Pullorum, and Salmonella Gallinarum (S. Gallinarum) [1]. Most of these serovars are transferable to humans and other livestock animals, resulting in the development of food-borne diseases and diarrhea. The serovar S. Gallinarum has a restricted host range and is usually only associated with poultry animals, but can cause significant losses in profit and low production yields.
In-feed antibiotics are commonly used to control Salmonella infection in chickens; however, the development of antibiotic resistance from chicken meat consumption poses a serious risk to consumers. Thus, antibiotic replacements and in-feed supplements, such as prebiotic oligo-saccharides (i.e., inulin, lactulose) and probiotic bacteria (i.e., Bacillus, Lactobacillus, Bifidobacterium) are being explored as alternative means to control Salmonella infection, and many have shown promising results in nutrient retention, growth performance, and meat quality in poultry [2,3]. In addition, probiotic mixtures have been shown to improve mucosal immunity and inhibit pathogen growth [4]. Feed maldigestion and loss of appetite in animals are initial symptoms of gastrointestinal (GI) diseases [5,6], suggesting that the GI tract plays a significant role in the development of salmonellosis. It is likely that the GI tract acts as the main interaction hub of the host animal’s microbiota and metabolic activities, thus contributing to overall gut immunity. Furthermore, the GI tract provides the first line of defense against intestinal pathogens. However, the effects of in-feed additives like Bacillus subtilis (B. subtilis) on S. Gallinarum infection, and its impacts on gut microecology and biological activities in laying hens, still remain unclear.
New sequencing techniques have recently enabled in-depth investigation of gut microecology. Using 16S rRNA gene pyrosequencing analysis, both culturable and unculturable bacteria can be quantified and identified with great efficiency and accuracy. This technology has been applied in microbiota studies in humans [7], pigs [8,9], and broiler [10]. However, no study has yet sought to characterize the microbiota of Hy-Line Brown (HLB) laying hens under the influence of S. Gallinarum infection or probiotic treatment.
In the present study, the fecal microbiota of HLB laying hens was investigated using 16S rRNA gene pyrosequencing. We compared the diversity and composition of the gut microbiota of HLB laying hens infected with S. Gallinarum, with and without the administration of a probiotic supplement, B. subtilis CSL2. Biological activities and functions in the gut microecology of HLB laying hens, with or without probiotics and S. Gallinarum infection, were determined using PICRUSt analysis.
MATERIALS AND METHODS
Animals and experimental setup
The Dankook University Animal Care Committee approved all animal protocols used in this study. A total of 24 HLB layers were randomly assigned to cages in the Dankook University Animal Care Facility. Hens were kept under artificial light for 16 hours per day and supplied with clean water ad libitum. The temperature was maintained at 32°C for the first week, and then reduced by 3°C weekly to 26°C. Hens were not given any antibiotics or other feed additives (aside from those specified in the experimental protocol) during the entire period of the experiment. No additional chickens were introduced during the course of the study. Sampling was done before infection, at the end of week 34, and 72 hours after infection. The scope and design of the experiment are outlined in Figure 1.

Experimental design. After rearing for 31 weeks, 24 hens were fed diets with or without the probiotic Bacillus subtilis CSL2 (107 CFU/g, mixed with feed) for 4 weeks. Fecal samples were taken before and after challenge with Salmonella Gallinarum KVCC-BA0700722 (108 CFU/mL, oral delivery). CFU, colony forming units.
This study examined changes in the fecal microbiome over the course of Salmonella infection in laying hens with and without the probiotic supplement B. subtilis CSL2. HLB laying hens aged 31 weeks were divided into three groups of eight birds each: control (CON), Salmonella-infected without probiotic administration (SAL), and Bacillus-fed Salmonella-infected (PRO) groups. All 24 laying hens received the standard basal diet (Supplementary Table S1) from birth to week 34. From week 31 to week 34, the PRO group was also given B. subtilis CSL2 (GenBank accession number: KX281166) at a final concentration of 1.0×107 colony forming units (CFU)/g of feed (Figure 1). B. subtilis CSL2 was provided in the form of a commercial probiotic supplement manufactured by Abson Biochem, Inc. (Ansan, Korea). A spore suspension of B. subtilis CSL2 was mixed into the feed. Feces were sampled from all groups beginning at the end of the week 31. At the end of week 34, both the SAL and PRO groups were orally challenged once with S. enterica subsp. Gallinarum KVCC-BA 0700722 at a final concentration of 1.5×108 CFU/mL. Fresh fecal samples were collected from each bird 72 hours post-infection (week 34). Fecal contents were stored in sterile tubes at 4°C until DNA isolation protocols were performed.
DNA isolation
Genomic DNA was isolated from freshly collected chicken feces according to the previously established protocol [10]. Fecal samples were transported directly in a sealed box to the laboratory for DNA extraction and were maintained at less than 4°C during transport. The Power Fecal DNA isolation kit (Mo Bio Laboratories, Inc., Hercules, CA, USA) was utilized for DNA extraction, according to the manufacturer’s instructions. The quality of the purified DNA was checked through agarose gel electrophoresis and a UV/Vis Spectrophotometer (Mecasys Co., Ltd., Daejeon, Korea).
Sequencing and processing of data
Initially, specific targets from the isolated DNA were amplified using polymerase chain reaction as described previously [11]. The amplified V1–V3 regions of 16S rRNA gene were used for pyrosequencing. Forward primers were tagged with 10-bp unique barcode labels at the 5′ end along with the adaptor sequence to permit numerous samples to be run in a single sequencing plate. The 16S rRNA amplicons were measured and pooled for the sequencing reaction. Sequence collections of 16S rRNA were generated using 454 GS FLX Titanium chemistry (Roche, Basel, Switzerland).
Data were processed through the pipeline established in quan titative insights into microbial ecology (QIIME; ver. 1.9.1; QIIME development team, CO, USA) [12]. Quality processing was done by first removing short (<200 bp) and long (>600 bp) sequences, low quality sequences, sequences containing incorrect primers, and/or sequences that contained more than one ambiguous base, using the split_libraries.py script. Next, chimeric sequences were identified and filtered from the data using the identify_chimeric_seqs.py and filter_fasta.py script. Operational taxonomic units (OTUs) were selected using the pick_open_reference.py script against the Greengenes reference database (13_8) with an identity threshold of 97%. The values for alpha-diversity indices (Shannon and Simpson) were generated using the alpha_diversity.py script. Sequences were rarefied according to lowest sequencing depth. The bacterial composition of the three sampling groups at phylum-to-genus level classification was generated using the summarize_taxa_through_plots.py script. Furthermore, beta-diversity plots were generated using the beta_diversity_through_plots.py in the QIIME software. All reads were pooled for each group of laying hens.
Data analysis
R software (ver. 3.1.0; R Development Core Team, Auckland, New Zealand) was utilized for statistical and computational analysis of group bacterial and functional profiles. Differences in the proportions of bacterial taxa (phylum- and genus-level classification) were calculated among the three groups before and after inoculation with S. Gallinarum. The visual outputs of the principal component analysis (PCA) plot were created from QIIME beta diversity and 2D plot scripts. Furthermore, PICRUSt was used to identify differences between the functional potential of the bacterial communities among different HLB laying hen groups [10]. Using the Kyoto encyclopedia of genes and genomes (KEGG) prediction for level 3 ortholog functions, differences between the CON, SAL, and PRO groups were observed and a loading plot was created to identify the most distinguishing functions among groups after 72 hours. Tukey’s honestly significant difference was employed to compare functional differences between groups, with a significance threshold of p<0.05.
RESULTS
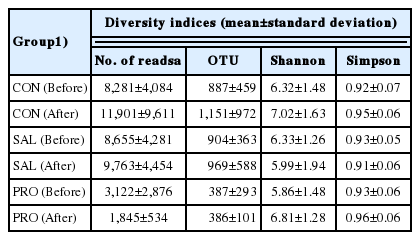
Diversity values in probiotic-fed and infected laying hens
The effects of S. Gallinarum infection and B. subtilis CSL2 administration on the alpha diversity of the fecal microbiota of laying hens were determined (Table 1). Alpha diversity values were measured as Shannon and Simpson diversity indices in the CON, SAL, and PRO groups. While the Shannon and Simpson values were similar across all groups before S. Gallinarum infection, after infection the diversity values of the SAL group decreased relative to those of the CON and PRO groups. Previous studies in pigs and broiler chickens have shown that pathogen infection lowers the alpha diversity of the microbiota, while probiotic or prebiotic administration may increase bacterial diversity [8,10].
Changes in the fecal microbiota of laying hens after infection
The most abundant bacterial phylum in the fecal microbiota of laying hens was Firmicutes, accounting for 53.0% to 72.0% of the bacterial population, followed by Bacteroidetes or Proteobacteria (Figure 2A). Previous studies have also demonstrated that these bacterial phyla are the main inhabitants of livestock and avian guts [13].
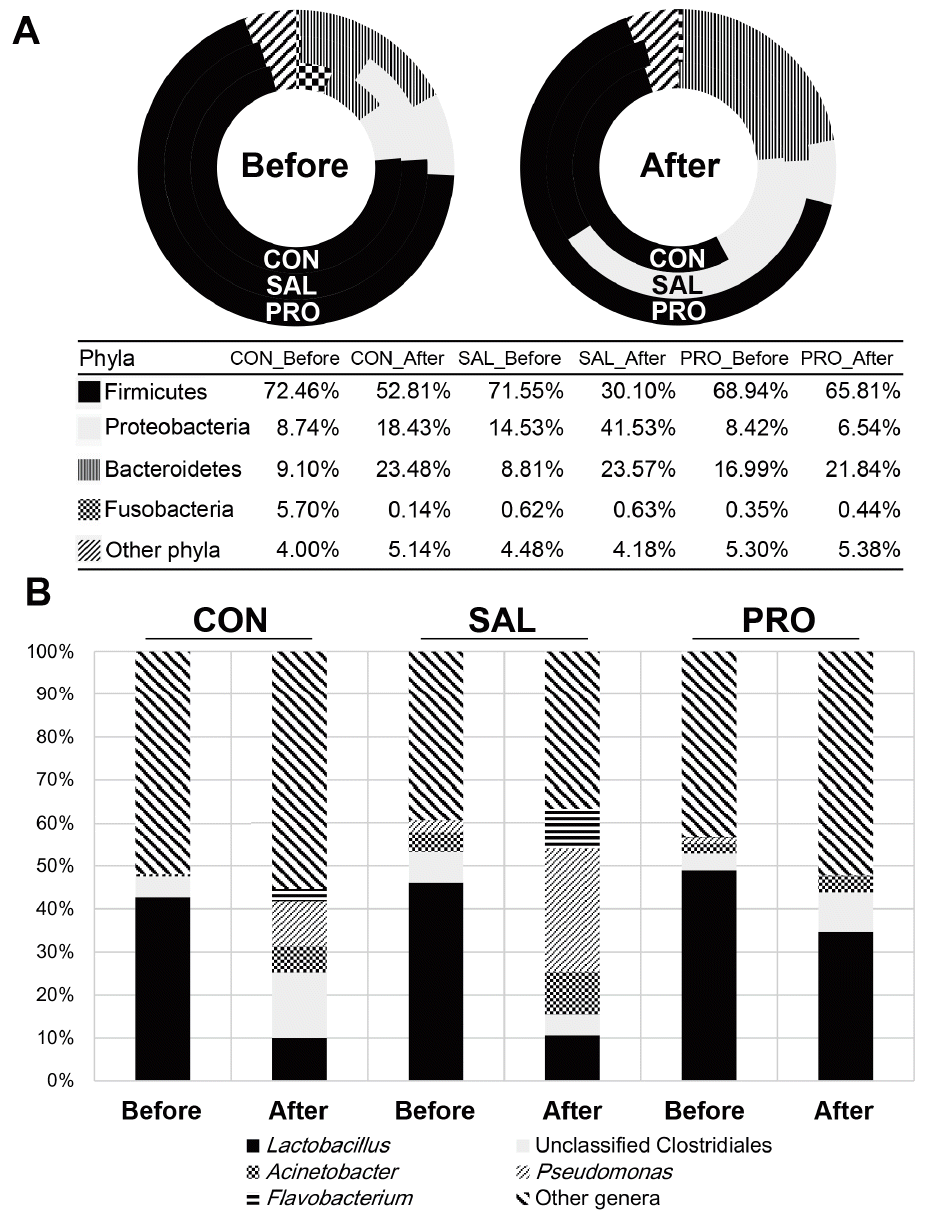
Compositional changes in the fecal microbiota of laying hens at the phylum (A) and genus (B) levels. Mean relative abundance level of bacterial taxa in experimental groups before and after Salmonella challenge. All laying hens (n = 24) were separated into three groups (8 animals/group): CON, control/basal diet; SAL, basal diet challenged with Salmonella Gallinarum; and PRO, basal diet supplemented with Bacillus subtilis CSL2 and challenged with Salmonella Gallinarum.
S. Gallinarum infection triggered significant alterations in the composition of the fecal microbiota. The proportion of Proteobacteria increased dramatically, from 14.5% to 41.5%, whereas that of Firmicutes decreased from 71.6% to 30.1% (Figure 2A). On the other hand, the administration of B. subtilis CSL2 (PRO group) controlled the Proteobacteria population and even caused it to diminish to below the level of CON group. This phylum includes a wide variety of pathogens, such as Escherichia, Salmonella, Vibrio, Helicobacter, Yersinia, etc. In previous studies, animals infected with strains of Salmonella demonstrated a significant increase in Proteobacteria [10,12]. Probiotic administration in pigs and broilers correlated with increased abundance of Firmicutes, and decreased abundance of Proteobacteria [10,14].
Further analysis of bacterial genera offers a clearer picture of the microbial changes induced by Salmonella infection. A total of 173 differentially abundant genera were identified in the feces of laying hens before Salmonella challenge (Supplementary Table S2). The most abundant genus in all groups before Salmonella infection was Lactobacillus, ranging from 42.7% to 49.0% of the population (Figure 2B). Other abundant genera included Pseudomonas, Flavobacterium, and Acinetobacter. Sequencing also identified unclassified Clostridiales, Ruminococcaceae, Enterobacteriaceae, and Lachnospiraceae bacteria (Supplementary Table S2). Earlier studies found that these bacterial genera and groups are natural residents of broiler hens and other livestock animals [8,12].
After Salmonella challenge in the SAL group, populations of Pseudomonas and Flavobacterium increased by a factor of 3 to 5, whereas populations of unclassified Clostridiales decreased significantly (Figure 2B). On the other hand, in the PRO group, the abundance of Lactobacillus recovered to the level of the CON group, and the population of Pseudomonas decreased significantly. Oh et al [10] reported that pathogenic infection decreases Lactobacillus populations in the microbiome. However, animals given feed supplemented with a probiotic harbored increased Lactobacillus populations [10]. A previous report showed that Pseudomonas are highly prevalent in patients infected with bacterial pathogens compared with uninfected subjects [15].
Multivariate and functional diversity analyses
Multivariate analysis was carried out in R Software using OTUs at 97% identity. The PCA showed that the microbiota of the CON, SAL, and PRO groups were similar before S. Gallinarum infection (Figure 3A), however, due to the dynamic nature of the chicken gut microbiota certain animal-to-animal variation was still observed as [10,13]. After infection, the microbiota of the SAL group became significantly different from those of the CON and PRO groups, suggesting that S. Gallinarum might trigger general changes in the fecal microbiota of laying hens (Figure 3B). In previous studies, administration of probiotic supplements induced shifts in the microbiota of pigs and broiler chickens [10,14].

Clustering analysis of laying hen microbiota before (A) and after (B) Salmonella challenge. Discriminant analysis of principal components revealing distinct clustering of the control (CON, light grey), Salmonella-challenged (SAL, dark grey), and Bacillus-treated (PRO, black) groups using operational taxonomic units defined at a 97% identity level. Significant differences (p<0.05) were calculated using compare_categories.py using the PERMANOVA test.
Further analysis of OTUs using PICRUSt revealed the meta bolic and functional profiles of the microbiota in each group (Figure 4) [16]. About 80 (out of 149 total) KEGG functions were differentially expressed in the CON, SAL, and PRO groups (Supplementary Table S3). Among these, 16 were significantly different between the SAL and PRO groups (Figure 4A). The abundance of genes associated with two-component systems, ribosomes, and secretion systems differed significantly between the PRO and SAL groups; these differences were significantly influential and discriminative, as confirmed by the loading plot (Figure 4B). In addition, many other functions of bacteria within the poultry gut were also influenced by Salmonella infection and recovered by Bacillus administration.

Comparison of microbial functionality in the microbiota. (A) Heat map showing normalized values of 16 differentially abundant functions (>1.0% abundance) between the negative control (CON), Salmonella-challenged (SAL) and Bacillus-treated (PRO) groups after the Salmonella Gallinarum challenge. Tukey’s honestly significant difference showed significant differences between the groups (p<0.05). The normalized abundance values are depicted visually from white to black; black represents the highest abundance, and white the lowest. (B) Canonical loading plot showing the magnitude of the influence of each variable on the separation of individual peaks in the laying hen microbiota (0.05 threshold level).
DISCUSSION
S. Gallinarum causes serious illness that results in morbidity and mortality among poultry and other livestock [1]. S. Gallinarum is a major source of dehydration, loss of appetite, and diarrhea in poultry with inadequate protection and reduced resistance against bacterial infection. Previous studies have reported that the distal gut and fecal isolates of infected poultry may serve as sites for bacterial colonization and growth of S. Gallinarum [17]. Many studies have investigated the mode of infection of S. Gallinarum and its relatives, but to our knowledge no studies have explored the impact of S. Gallinarum infection and probiotic administration on the gut microbiota and functional profiles of laying hens. In the present study, laying hens were given feed with or without the probiotic B. subtilis CSL2. S. Gallinarum was then orally administered to the SAL and PRO groups. The microbiota of HLB laying hens orally challenged with S. Gallinarum and not given B. subtilis CSL2 displayed lower bacterial diversity than that of uninfected laying hens, but the greatest diversity was exhibited in the PRO group. These compositional changes in the microbiota were also observed in downstream analysis.
In hens with dysbiosis of microbial diversity due to S. Gallinarum infection, the abundance of Proteobacteria showed the greatest degree of increase, while the abundance of Firmicutes decreased the most. These microbial shifts promote the proliferation of enteropathogenic bacteria, leading to inhibition of the growth of anaerobic and short chain fatty acid-producing bacteria [13,18]. Administration of B. subtilis CSL2 reversed the effect triggered by S. Gallinarum infection on Firmicutes and Proteobacteria abundance. The pathogenic infection of animals induces growth of other opportunistic pathogens in the microbiota [10]. In this study, S. Gallinarum induced the growth of opportunistic pathogens, such as Pseudomonas and Flavobacterium. In previous studies, increased abundance of Pseudomonas in the gut microbiota of immunocompromised subjects was associated with bacterial infection. An earlier report of bacterial infection by Flavobacterium was reported in chickens [19], causing fatal diseases both in poultry and humans. However, some probiotic and prebiotic compounds are able to protect against pathogenic infection, or control the growth of opportunistic and multidrug-resistant bacteria [20].
Supplementing feed with B. subtilis CSL2 promoted the growth of Lactobacillus in laying hens. Probiotics and synbiotic compounds are also able to increase the growth of potentially probiotic groups, such as Bifidobacterium and Lactobacillus [8,21]. The Lactobacillus group is known to include several strains reported to be beneficial to their hosts [22]. Some of the beneficial effects of probiotic strains include production of antimicrobials, including hydrogen peroxide, lactic acid, and bacteriocin [23], production of enzymes for the breakdown of feed-based polysaccharides [24,25], and immunomodulation [26]. High levels of lactobacilli have been shown to inhibit the growth of Salmonella, Escherichia coli K88, and several other pathogens in broiler chickens [10] and pigs [8], thereby enhancing the performance of livestock animals by protecting against bacterial infection.
In the present study, the overall microbial profiles of laying hens were significantly changed by infection with S. Gallinarum and administration of B. subtilis CSL2, as depicted in the PCA plot (Figure 3). The apparent clustering by group suggests that birds develop distinct microbial signatures based on their infection status and probiotic administration. Laying hens were either uninfected, infected by S. Gallinarum, or Salmonella-infected but protected by B. subtilis CSL2. Microbial shifts can induce protection or dysbiosis depending on the groups of proliferating and/or inhibited bacteria. This phenomenon has also been observed in the microbiota of livestock infected with Salmonella or fed with probiotics [14,27].
The overall compositional changes in HLB microbiota reflect alterations in the biological activities in laying hens’ guts. Further analysis of these microbial changes uncovered differences in microbial functions in response to Salmonella infection or protection by Bacillus. The metabolic and functional profiles of each group of laying hens were compared, which provided deeper insights into the GI processes influenced by S. Gallinarum and B. subtilis CSL2. The gut functions related to two-component systems, ribosomes, and secretion systems showed striking variations in HLB laying hens, particularly between the SAL and PRO groups. Genes associated with ribosomes were highly abundant in the PRO group. Ribosomes play an important role in many basic biological processes, particularly in the synthesis of structural proteins and enzymes [28]. The increase in ribosomal activity as a result of probiotic supplementation suggests that enzymatic reactions, and proteins that provide structural support, are important in defending against S. Gallinarum infection and the proliferation of opportunistic pathogens. In contrast to the PRO group, the SAL group showed a greater abundance of genes associated with two-component and secretion systems. Bacterial two-component systems play a role in sensing and adapting to environmental and cell surface signals [6]. Several studies have demonstrated that two-component systems are also essential to the ability of Salmonella to assemble virulence factors (i.e. biofilm formation and invasion) and cause disease [29]. In addition, the type three secretion system (T3SS) is associated with pathogenicity by delivering effector proteins from Salmonella-containing vacuoles into host cells [6]. Collectively, the abundance of genes related to signaling and secretion suggests that Salmonella-induced dysbiosis is mediated by the release of virulence factors, resulting in successful infection that might be reversed in the presence of the probiotic B. subtilis CSL2. Therefore, a detailed analysis of these key functions might shed light on the probiotic mode of action and mechanisms of S. Gallinarum infection in laying hens.
CONCLUSION
The microbial and functional profiling of HLB laying hens demonstrated the potential protective effects of using an in-feed probiotic supplement, B. subtilis CSL2, against S. Gallinarum infection. Protective effects include improvement of bacterial diversity, enhanced metabolic activity and gut functionality, and reversal of the effects of S. Gallinarum infection, including increased growth of Lactobacillus and inhibition of Pseudomonas and Flavobacterium. The insights of this study will improve preventive measures and strategies against Salmonella infection in laying hens. Moreover, the results of the study provide additional insight into the mode of Salmonella infection and the potential of probiotics as protective agents in poultry production.
Supplementary Data
ACKNOWLEDGMENTS
This work was supported by a grant from the Next-Generation BioGreen 21 Program (PJ01115903), Rural Development Administration, Republic of Korea.
Notes
CONFLICT OF INTEREST
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.