Effects of dietary onion (Allium cepa) extract supplementation on performance, apparent total tract retention of nutrients, blood profile and meat quality of broiler chicks
Article information
Abstract
Objective
This study was conducted to investigate the effect of dietary onion (Allium cepa) extract (OE) supplementation on growth performance, apparent total tract retention (ATTR), blood profile, carcass characteristics and meat quality in broilers.
Methods
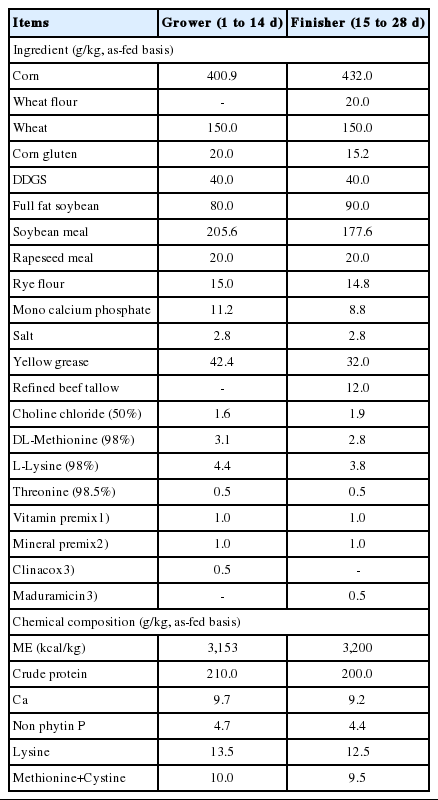
Four hundred male broiler chicks (Ross 308, 3-d old) were randomly allocated to four dietary treatments for 28 d feeding trial. Each treatment has five replications with 20 birds each. Four dietary treatments were designated according to the OE supplementation levels (0 as control, 5, 7.5, and 10 g of OE per kg of basal diet respectively). On d 28, a total of 20 birds from each treatment were subjected for ATTR, serum biochemical assay, carcass characteristic and organ weight measurement.
Results
Overall weight gain of OE 7.5 g/kg group was higher (p = 0.04) than control group. The ATTRs of dietary energy (p<0.01) and ether extract (p = 0.04) linearly increased with increasing levels of dietary OE. However, no difference in ATTR of dry matter and crude protein was evident. Furthermore, serum IgG concentration increased linearly (p<0.01) and quadratically (p = 0.03) with increasing OE supplementation. No differences in carcass dressing weight and amount of abdominal fat by treatments were observed. Also, the weight of organ including immune organ was not different among the treatments. The TBARS values of 10 d stored breast meat decreased linearly (p<0.01) and quadratically (p<0.01) with increasing dietary OE levels. The meat color was also affected, with lower (p<0.01) redness score in meats from OE supplemented groups. This study showed that dietary OE improved broiler weight gain presumably by increasing feed intake and ATTR of both energy and ether extract. The dietary OE increased serum IgG level and meat anti-oxidation capacity.
Conclusion
This study implies that the recommended level of dietary OE supplementation could be beneficial for improving broiler performance and meat quality.
INTRODUCTION
Antioxidant has been used to prevent oxidation in broiler meat, due to the ability to scavenge free radical [1]. Although synthetic antioxidants such as Butylated hydroxyl anisole (BHA), Tertiary-butyl hydroquinone (TBHQ), and Butylated hydroxyl toluene (BHT) are in use, concerns against synthetic substances have not been dissipated. Synthetic antioxidants are suspected to cause mutagenic and toxic effects on the body [2]. As a substitute for the synthetic antioxidants, many natural antioxidants have been evaluated as a feed supplement [3,4].
Onion (Allium cepa) contains several flavonoids including flavonols, flavones, quercetin and catechin [5]. Among those flavonoids, quercetin is the major flavonoid, whereby its content in yellow onion was reported as high as 347 mg/kg [5]. In addition, the quercetin is known to exert the strongest antioxidant potential especially for retarding the meat oxidation [6]. Also, it has been confirmed that dietary supplementation of quercetin decreased TBARS values of beef loin [7] and pork patties [8].
In addition, the benefits of onion feeding on animal performance have been reported. Broiler chicks fed 30 g/kg onion juice increased body weight gain, increased the serum high-density lipoprotein (HDL)-cholesterol and decreased triglyceride levels [9]. However, little has been reported concerning the benefit of dietary onion supplementation and the appropriate supplementation level to animal diet. It was reported [10] that the supplementation of onion as low as 100 mg/kg diet increased broiler performance. By far, most practical feeding studies have used freshly minced onions, which are difficult to precisely weigh and mix. Therefore, we propose that a liquid onion extract (OE) would be more convenient for supplementing functional substances onto the animal diet. The extraction process typically involves a certain degree of cooking, and that cooking was presumed to change the organoleptic property of the OE as compared to the fresh juice. Hence, this study was carried out to find the appropriate supplementation level of the OE and its effect on broiler growth performance, broiler meat anti-oxidation and serum profile.
MATERIALS AND METHODS
The experimental protocol was approved by Animal Research and Ethics Committee of Kangwon National University, Chuncheon, Republic of Korea.
Preparation of onion extract to the diets
Fresh yellow onions (Allium cepa) purchased from local market were extracted according to the reported procedure [11]. In brief, fresh onion bulbs were cleaned, peeled and cut into small pieces. The pieces of onion were mixed with salt (3 g/kg) and then heated at 60°C for 30 min. Hereafter, the onions were minced using electric blender then extracted through coffee filter to get liquid portion as the onion extract (OE), subsequently the OE was stored at 4°C. The proximate composition of OE was analyzed according to the AOAC [12] method. The total polyphenol and quercetin contents of OE were analyzed using Folin-Ciocalteau assay [13]. Furthermore, prior to experiment, graded levels of OE were evenly mixed to the diets for 10 min using feed mixer, then stored to the feedbag until the experiment started.
Experimental birds and treatments
A total of 400, male broilers (Ross 308, 3-d-old, average 75 g initial body weight) were housed in floor barn bedded with rice husk. Birds were randomly allocated to four dietary treatments for 28 days feeding trial. Each treatment had five replicate pens (2 m×1 m) with 20 birds per each pen. The different levels (0 as control, 5, 7.5, and 10 g of OE) were respectively supplemented to kg of basal diet to make four experimental diets. Two basal diets (Table 1) were used for starter phase (d 1 to d 14) and grower phase (d 15 to d 28). The basal diets were formulated by referring the Korean Feeding Standard [14] for broilers to meet or exceed nutrients levels of (KFS, d 7 to d 21) for the grower and (KFS, d 22 to market) for the finisher phase diets respectively. Feed and water were supplied ad libitum. During the feeding trial, feed intake and body weight (BW) were measured weekly. The room temperature was adjusted at 33°C for the d 1 and then gradually reduced to 22°C±1°C, then maintained the temperature until the end of feeding trial.
Blood collection and analysis
At the end of 28 days feeding trial, 10 chicks with BW similar to average from each treatment were selected for blood biochemical analyses. Blood was taken from jugular veins and placed into vacutainer tube (Becton Dickinson, USA) and stored at 4°C. Then the refrigerated blood samples were centrifuged at 3,000×g for 15 min. Serum was separated and stored at −80°C for further analyses of total cholesterol, triglyceride (TG), glucose, HDL cholesterol and IgG (Immunoglobulin G). The serum biochemicals were determined using kits (Asan Pharm. Co., Seoul, Korea) and spectrophotometer (Optizen 3220 UV, Mecasys Co., Daejeon, Korea). IgG was analyzed using kit (Bethyl Lab Inc., Montgomery, TX, USA) and microplate spectrophotometer (Powerwave XS, BioTek Ins., Winooski, VT, USA).
Immediately after blood collection, the birds were slaughtered and then the weight of carcass (without feet, neck, and head) was measured using a scale. Also, organs including abdominal fat, liver, heart, spleen, bursa of Fabricius and thymus were weighed. After the weighing, breast meat was collected and packaged then kept at −4°C until time course analysis of thio barbituric acid reactive substances (TBARS) and meat color scores at 0, 5, and 10 days of storage. Before measurement of TBARS and color scores, breast meat samples were thawed at room temperature for an hour. Then, the TBARS was analyzed according to procedure of [19] and meat color was measured using color difference meter (Yasuda Seiko Co., Osaka, Japan).
The ATTR was evaluated by total tract excreta collection method using broilers selected after feeding trial. Forty birds (ten birds per treatment and two birds in a replicate pen) were placed in metabolic cage (0.3 m×0.3 m×0.4 m) for 4 d. After acclimatization for 4 d, total excreta from each cage were collected for 72 h. The collected excreta was dried in a dry oven at 65°C for 4 d and stored for further chemical analysis. Dry matter, crude ash, crude protein, and ether extract (EE) of feed and excreta were analyzed according to the AOAC [12] methods and the gross energy was analyzed using bomb calorimeter (Parr 1261, Parr Ins., Moline, IL, USA).
Statistical analysis
All the data collected were statistically analyzed using a one-way analysis of variance. The means were subjected to the test of significance by Duncan Multiple Range Test. Pen was considered as the experimental unit for all statistical analyses. Both linear and quadratic effects were analyzed by specifying each OE levels into the polynomial contrast of SPSS software [16]. Significant differences among mean values were presented as p-values and superscripts.
RESULTS
The proximate composition, total polyphenol and quercetin concentration of OE are presented (Table 2). Since the fibrous portion was eliminated from fresh onion, the moisture level of OE increased up to 93%. Furthermore, amount of total polyphenol concentration was 0.39 g/kg of OE, whereby quercetin was the major component representing 0.36 g/kg of OE.
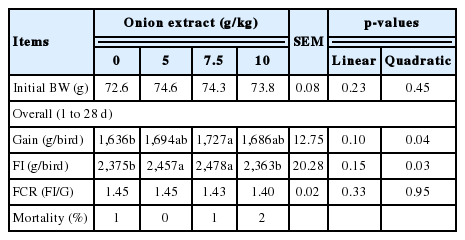
Overall (1 to 28 days) period, the body weight gain of broilers fed 7.5 g/kg OE group was higher than the control group. Increase of broiler body weight gain by dietary OE supplementation showed quadratic response, but slight decrease occurred with the highest (10 g/kg) OE supplementation. Also, similar quadratic response (p = 0.03) appeared on feed intake according to the levels of dietary OE supplementation (Table 3). No differences in feed conversion ratio and mortality among the dietary OE treatments were observed.
The ATTRs of energy (p<0.01) and ether extract (p = 0.04) linearly increased with increasing levels of dietary OE. Furthermore, the energy ATTR of 10 g/kg OE group was higher than 5 g/kg OE group. The EE ATTR of 7.5 and 10 g/kg OE groups was higher than control group. However, the ATTRs of crude protein and dry matter were not different among the experimental diets (Table 4).
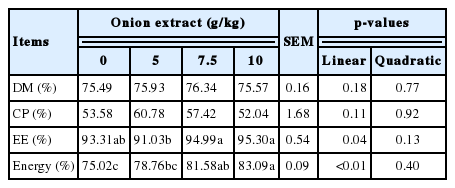
Effect of dietary onion extract supplementation on apparent total tract retention of nutrients in experimental diet
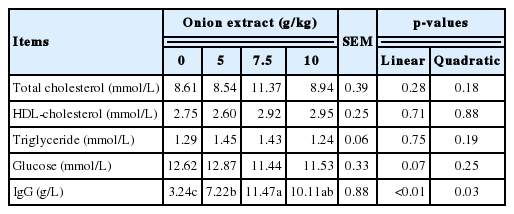
Furthermore, the serum biochemical assay did not exert any significant differences on serum TG, glucose, and HDL-cholesterol (Table 5), although the serum glucose level showed a tendency (p = 0.07) of linear increase with increasing OE levels. However, the serum IgG level of OE groups was higher than control group with both linear (p<0.01) and quadratic (p = 0.03) response by dietary OE levels.
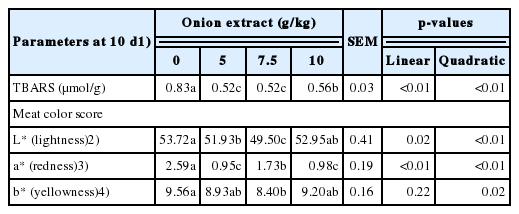
The percentage of both carcass weight and abdominal fat and relative weight of liver, heart, spleen, bursa of Fabricius and thymus were not affected due to dietary OE levels (Table 6). However, the TBARS values of 10 d stored breast meat decreased linearly (p<0.01) and quadratically (p<0.01) with increasing dietary OE levels (Table 7). The meat color lightness (L*) values were varied quadratically (p<0.01) by dietary treatments with lower values in medium OE level groups. The color redness (a*) values of OE groups were lower (p<0.01) than the control group with higher magnitude by increasing OE levels within all OE groups. The meat color yellowness (b*) value was varied quadratically (p = 0.02) with lower value in 7.5 g/kg OE group than both control and 10.0 g/kg OE groups.
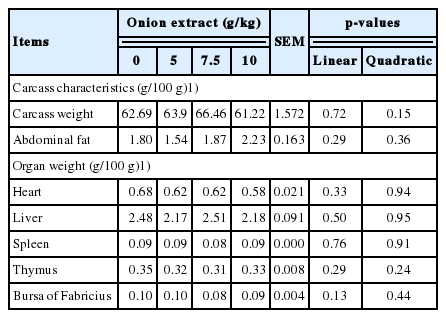
Effect of dietary onion extract supplementation on carcass characteristics and organ weight of broiler chicks
DISCUSSION
Onion has been known as a source of bioactive phenolic compounds. Many of these compounds have drawn attention due to their antioxidant properties. Total polyphenol concentration of the present experimental OE was 0.39 g/kg, which is similar to the value 0.397 g/kg [13], but lower than the value 0.62 g/kg [17]. As it was discussed [13], the total polyphenol content would be varied by types of onion, weather during harvest and storage condition. However, the quercetin concentration was 0.36 g/kg OE, representing 92.3% proportion of total polyphenol in the present study, which is higher than 85% quercetin [18].
Body weight gains were quadratically improved by increasing levels of dietary OE supplementation in the present study. Another broiler study [10] found the same increase on body weight gain although the supplementation level of onion was relatively low as 100 mg/kg. The improvement on body weight gain could be attributed to an increase of feed intake by dietary OE supplementation as confirmed by the present study as well as other studies [9,10]. The increase of feed intake by OE supplementation could be induced by the favorable taste of OE. It is already proved that the sulfur-containing components in onion change by heating, which then induces bispropenyl disulfide with a sweet taste and flavor [19]. In addition, chicks are known to have reliable gustatory impulses [20], although it has not yet been proved whether the chicks have preference for a specific taste. Both feed intake and body weight gain was not increased [21] by dietary OE supplementation. However it should be noted that in this study the OE [21] was a fermented OE and the fermented OE was supplemented until the broiler reached to 800 g body weight. After all, it is still not evident how the onion or its extract affects growth performance and feed intake. However, considering all these results, it can be concluded that the dietary OE is certainly beneficial for improving body weight gain. In addition, the benefit appears to disappear when the OE supplementation level was too high as in 10 g/kg of diet.
Along with the increase in the feed intake, an improvement in ATTR could be another contributor for the increased weight gain. Increased ATTRs of both dietary energy and EE by OE supplementation indicates that the improvement in energy ATTR could be induced from the improved ATTR of EE. No improvement in EE ATTR was noticed along with no improvement in energy ATTR in the low level OE group. Contrary to EE, CP, ATTR was not closely affected by the increased OE levels. Since the increase in feed intake generally inversely affects the energy ATTR, it is still not sufficient enough to explain how the energy ATTR improved although the feed intake increased by dietary OE supplementation. It could be explained by a report [22] that the onion is able to synthesize bile acid and thereby was able to improve the lipid digestion. The same author mentioned that the onion stimulates the activity of pancreatic enzymes, especially lipase and amylase. Another study [23] suggested the essential oil in onion as a digestion enhancer.
Effect of dietary onion on serum cholesterol could be varied. Similarly to the present study, dietary onion as much as 30 g/kg broiler diet did not exert any change on serum total cholesterol [9]. However, other studies reported significant decrease of serum total cholesterol by dietary quercetin [24] and onion extract [21]. Serum IgG value increased in the present study, whereas a mouse study [25] reported no effect on IgG value by dietary onion feeding. In addition, dietary onions increased immune capability of rats by increasing CD (cluster of differentiation) 4 and white blood cells [26].
Feeding onion is presumed not to affect carcass percentage and organ weights. No effect of dietary onion on carcass percentage was found [10], which is similar to the present study. However, the anti-oxidative capability of meat during storage was affected by dietary OE. The decrease on the TBARS values of breast meat in this study is in agreement with a report [28] that found the retardation of meat lipid oxidation by dietary quercetin. The direct onion addition to beef patty [27] also reduced TBARS value of both non-irradiated and irradiated beef after 7 d storage. It is therefore evident that the onion itself or its extractable substances are, more or less, acting as an antioxidant during meat storage. However, it is not fully known how and what anti-oxidative substances in onion would impart the anti-oxidative action. Total phenolic content in broiler breast meat increased proportionally with increasing levels of dietary quercetin and the phenolic content is also proportional to the meat diphenyl-picrylhydrazyl (DPPH) scavenging activity [28]. The above result proved that the dietary quercetin can be absorbed and accumulated into broiler meat by dose dependent manner. In addition, the phenolic accumulation in the meat has contributed for the meat anti-oxidation.
The meat color is known to be associated with the degree of meat oxidation [28]. In this study, meat color L* values statistically differed by dietary OE, although the difference did not show any meaningful trend. Feeding onion-garlic supplemented diet resulted the increase of L* value at 7 d stored meat of broiler [29]. Contrary to the above onion-garlic supplementation, the OE supplementation decreased the L* value in the present study. The meat color a* values were lowered by dietary OE, although there was no dose dependent manner. In a pig feeding study [30], dietary quercetin decreased a* value of pork. However, there are other studies [8,21] that reported either no or inconsistent effect by dietary onion or quercetin on meat color. Therefore, it could be summarized that the dietary OE was evidently beneficial for preventing meat oxidation. However, the degree of anti-oxidation is not consistently represented as the respective change of meat color. In addition, the OE and its polyphenol could not be a significant contributor for the change of broiler meat color.
In this study, dietary supplementation of OE as high as 7.5 g/kg was able to not only increase feed intake and weight gain, but also decrease TBARS value of stored breast meat. Therefore, the OE could be recommended as an alternative natural growth promoting and meat anti-oxidative feed additive for broiler production, where the 7.5 g OE/kg diet could be the recommendation level. However, further studies are needed to explain how the OE increased the serum IgG level.
ACKNOWLEDGMENTS
This study was supported by 2013 research grant from Kangwon National University (No.120131797).
Notes
CONFLICT OF INTEREST
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.