One Alpha-hydroxycholecalciferol Improves Growth Performance, Tibia Quality, and Meat Color of Broilers Fed Calcium- and Phosphorus-Deficient Diets
Article information
Abstract
An experiment was conducted to evaluate the effects of one alpha-hydroxycholecalciferol (1α-OH D3) on growth performance, tibia quality, and skin and meat color in broilers fed calcium (Ca)- and phosphorus (P)-deficient corn-soybean meal diets. A total of 288 male Ross broilers, at 21 days of age, were randomly assigned to three treatments with eight cages per treatment. Three levels of 1α-OH D3 (0, 5, and 10 μg/kg) were added to a basal diet (0.50% Ca, 0.13% non-phytate phosphorus (NPP), and 0.35% total phosphorus (tP)) without vitamin D3. As a result of this study, the addition of 1α-OH D3 increased body weight gain (p<0.001), feed intake (p = 0.007), feed efficiency (p<0.001), tibia weight (p = 0.002), length (p<0.001), breaking-strength (p = 0.012), ash (p<0.001), Ca (p<0.001), and P content (p = 0.004). Dietary 1α-OH D3 enhanced breast meat yellowness (p = 0.015) and the length and weight of the small intestine of the broilers. Moreover, 1α-OH D3 decreased serum Ca concentration (p = 0.074) and breast meat redness (p = 0.010). These results indicate that the 1α-OH D3 improves growth, tibia quality, and meat color in broilers fed Ca- and P-deficient corn-soybean meal diets.
INTRODUCTION
As an analog of cholecalciferol (vitamin D3), the one alpha-hydroxycholecalciferol (1α-OH D3) is slightly lower in efficacy than 1,25-dihydroxycholecalciferol (1,25-(OH)2 D3) in broilers (Edwards, 2002). Biehl and Baker (1997b) and Snow et al. (2004) reported that 1α-OH D3 improves growth performance, tibia quality, and phosphorus (P) utilization in broilers. However, most studies focus only on starter broilers, and to date, limited information is available on growing-finishing phase birds.
Previous studies on 1α-OH D3 have been based on adequate dietary vitamin D3 (Biehl and Baker, 1997a, 1997b; Snow et al., 2004; Driver et al., 2005). Edwards (2002) found that an interaction between vitamin D3 and 1,25-(OH)2 D3 exists in tibia ash. To avoid possible interactions between vitamin D3 and 1α-OH D3, we investigate the effect of 1α-OH D3 in a basal diet without vitamin D3. Our unpublished data showed that 1α-OH D3 efficacy responds negatively to increasing dietary calcium (Ca) levels, and 1α-OH D3 yields the highest activities at low concentrations of dietary Ca (Han et al., 2012). Therefore, in the present experiment, a low-Ca diet was designed.
Broiler skin and meat color is important sensory characteristics by which consumers judge meat quality (Petracci and Fletcher, 2002). In our published work, Han et al. (2009) found that 1α-OH D3 enhances the lightness and yellowness of breast and thigh meat of 1- to 21-d-old broilers. Previous studies on swine showed that the lightness of pork decreases and the redness increases or is not affected, by the addition of vitamin D3 (Wiegand et al., 2002; Wilborn et al., 2004). However, the effects of 1α-OH D3 on skin color in broilers have not been examined. Petracci and Fletcher (2002) reported that broiler skin and meat color dramatically changes during the first 6 h postmortem. Thus, broiler skin and meat color were determined immediately after the broilers were slaughtered.
The effect of 1α-OH D3 on weight and length of small intestine remains unclear. Therefore, the objective of the current experiment is to evaluate the effects of 1α-OH D3 on growth performance, tibia quality, skin and meat color, as well as small intestine parameters in broilers fed Ca- and P-deficient diets.
MATERIALS AND METHODS
Birds, diets, and management
All procedures in the present experiment were approved by the Animal Care Committee of Shangqiu Normal University.
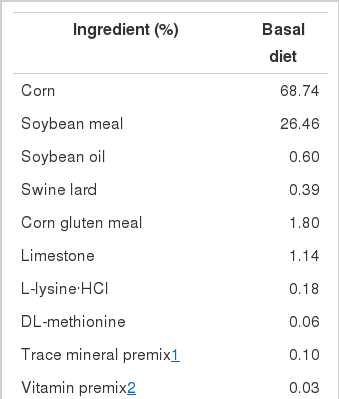
On the day of hatch, 288 male Ross broiler chicks were randomly and equally assigned to 24 cages (70×70×30 cm), with 12 chicks per cage. All chicks were fed a starter mash feed (apparent metabolizable energy (AME), 2,940 kcal/kg; crude protein (CP), 20%; Ca, 1%; total phosphorus (tP), 0.68%; non-phytate phosphorus (NPP), 0.45%) until they were three weeks old. On d 21, the broilers were individually weighed, transferred to growing-finishing cages (200×50×35 cm), and then assigned to one of the three dietary treatments with eight replicates per treatment. Three levels of 1α-OH D3 (0, 5, and 10 μg/kg) were added to a basal diet (0.50% Ca, 0.13% NPP, 0.35% tP) without vitamin D3 (Table 1). The crystalline 1α-OH D3, supplied by Taizhou Healtech Chemical Co., Ltd. (Taizhou, China), was dissolved in ethanol, and then brought to a final concentration of 10 mg/L of 1α-OH D3 in a solution of 5% ethanol and 95% propylene glycol (Biehl and Baker, 1997b).
The birds were given access to mash feed and water ad libitum during the 21-d experiment, with an 18-h lighting schedule.
Collection and analysis of samples
To collect blood and tibias, two chicks were randomly selected from each replicate and weighed (Edwards, 2002). Blood samples (5 ml) were collected into 5-ml anticoagulated syringes (Shanghai K&G International Co. Ltd., Shanghai, China) through the wing vein, and then centrifuged for 10 min at 3,000 g at 20°C. The birds were sacrificed by exsanguination. Serum Ca and inorganic phosphate (Pi) were determined with a Shimadzu CL-8000 Analyzer (Shimadzu Corp., Kyoto, Japan) following the manufacturer’s instructions.
The left and right tibias of the individual birds were excised. According to the method by Hall et al. (2003), the left tibias were boiled for 5 min to loosen the muscle tissues. The meat, connective tissue, and the fibula bone were then removed completely using scissors and forceps. After the tibias were cleaned, they were put into a container with ethanol for 48 h (removing water and polar lipids). The bones were further extracted in anhydrous ether for 24 h (removing non-polar lipids). The tibias were dried at 105°C for 24 h, and then weighed. The tibia diameter was determined at the medial point, and the tibia ash content was determined by ashing the bone in a muffle furnace for 18 h at 600°C (Han et al., 2009). Skin color values of lightness (L*), redness (a*), and yellowness (b*) were determined on the area between the pectoral and sternal feather tracts and the meat colors measured on the raw muscles (Petracci and Fletcher, 2002). Color values of skin and meat were measured by a Chroma Meter WSC-S (Shanghai Precision and Scientific Instrument Co., Ltd., Shanghai, China).
The right tibia was used to analyze the breaking-strength. Tibia breaking-strength was determined using an all-digital electronic universal testing machine (Shenzhen Hengen Instrument Co. Ltd., Shenzhen, China). The tibias were cradled on two support points 4 cm apart. By using a 50-kg load cell and a crosshead speed of 10 mm/min, force was applied to the midpoint of the same facial of each tibia (Jendral et al., 2008).
The whole small intestine was isolated immediately from the gastrointestinal tract and cut into 3 pieces: duodenum, jejunum, and ileum. The weight and length of the three intestinal pieces were measured (Wang and Peng, 2008).
Statistical analysis
The replicate means served as the experimental unit for statistical analysis. All data were analyzed using the ANOVA procedure of SAS software (SAS Institute, 2001) as factorial experiments, with all statements of significance p≤0.05 unless indicated otherwise.
RESULTS AND DISCUSSION
Growth performance and tibia quality
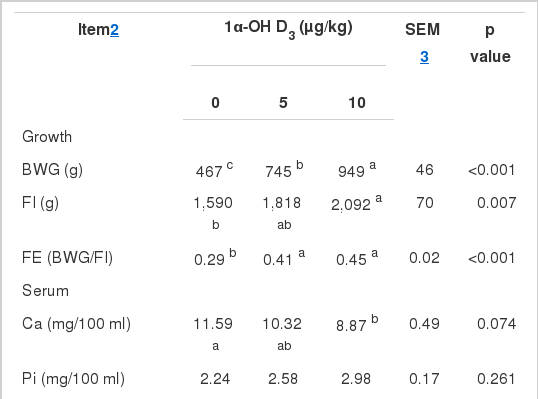
The diet consisting of 0.50% Ca and 0.13% NPP resulted in severe deficiencies of Ca and P in the broilers. The body weight gain (BWG) in birds fed basal diets was only 467 g (Table 2). The addition of 5 and 10 μg/kg of 1α-OH D3 corrected the Ca and P deficiency and increased the BWG to 745 and 949 g, respectively. Dietary 1α-OH D3 also improved feed intake (FI, p = 0.007), feed efficiency (FE, p<0.001), tibia weight (p = 0.002), length (p<0.001), breaking-strength (p = 0.012), ash (p<0.001), Ca (p< 0.001), and P content (p = 0.004). However, 1α-OH D3 did not significantly increase serum Pi (p = 0.261) or decreased the serum Ca concentration (p = 0.074).
Research has shown that sodium-dependent phosphate absorption by intestinal brush-border membrane vesicles (BBMV) decreases with age in rats; NaPi-IIb gene expression also decreases proportionally with age (Xu et al., 2002). The compound 1α-OH D3 is metabolized to 1,25-(OH)2 D3 in the intestines of chicks (Edelstein et al., 1978). Dietary 1,25-(OH)2 D3 increases phosphate absorption in suckling and adult rats and enhances NaPi-IIb mRNA abundance in suckling rats (Xu et al., 2002). However, 1,25-(OH)2 D3 has no effect on NaPi-IIb mRNA in adults (Xu et al., 2002). These results indicate that the efficacy of vitamin D analogue on P metabolism is influenced by animal age.
Previous studies on 1α-OH D3 have been based on starter broilers, in which 1α-OH D3 improves growth performance, tibia ash, and P retention (Biehl and Baker, 1997b; Edwards, 2002). In the present study, the addition of 1α-OH D3 increased BWG, FI, and FE in Ca- and P-deficient diets of 21- to 42-d-old broilers, which agreed with the results of 1- to 21-d-old broilers reported by Snow et al. (2004). Tibia quality was also enhanced by the addition of 1α-OH D3. The tibia weight and breaking-strength improvement by 1α-OH D3 came from the increase in tibia Ca and P. Dietary 1α-OH D3 decreased serum Ca concentration (p = 0.074), which agreed with the previous research on starters by Edwards (2002) and Han et al. (2009). The addition of 1α-OH D3 showed a trend of increasing serum Pi concentration, but the difference was not significant.
Skin and meat color
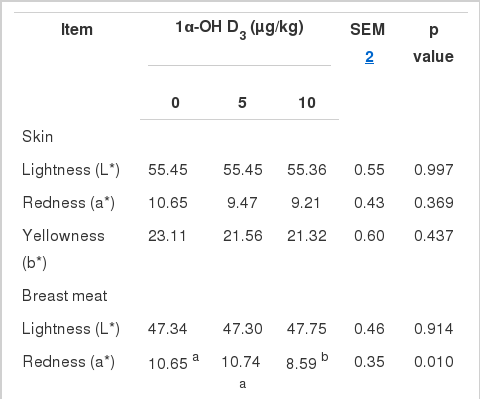
The addition of 1α-OH D3 did not influence the skin color of 21- to 42-d-old broilers (Table 3), but increased breast meat yellowness (b value, p = 0.015) and decreased its redness (a value, p = 0.010). The thigh meat redness in 10 μg/kg of the 1α-OH D3 group was lower than that of the 5 μg/kg of 1α-OH D3 group (p = 0.027).
Previous studies have shown that vitamin D3 improves the lightness and redness of pork (Wiegand et al., 2002; Wilborn et al., 2004), while 1α-OH D3 increases the lightness and yellowness of the breast and thigh meat of starter broilers (Han et al., 2009). In the current study, 1α-OH D3 enhanced breast meat yellowness and decreased its redness. Meat color is affected by pH and myoglobin. The pH value of breast meat or thigh meat is related positively to redness, but negatively to lightness and yellowness (Allen et al., 1998; Fletcher, 1999; Fletcher et al., 2000; Qiao et al., 2002). A low pH reduces the importance of myoglobin in selectively absorbing green light, resulting in meat that appears less red and more yellow (Castellini et al., 2002).
Small intestine features
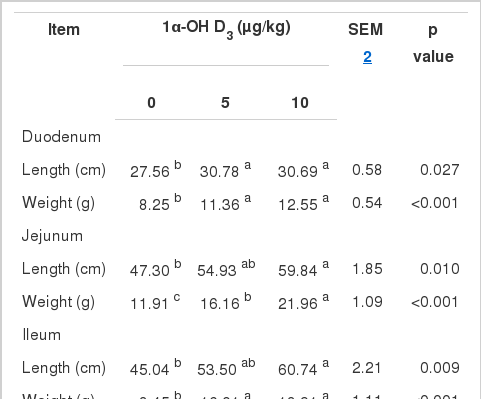
The addition of 1α-OH D3 increased the length (p = 0.027, p = 0.010, p = 0.009, p = 0.001) and weight (p< 0.001, p<0.001, p<0.001, p<0.001) of the duodenum, jejunum, ileum, and the whole small intestine, respectively.
Fassbinder-Orth and Karasov (2006) have found that feed restriction don’t significantly influence the small intestine weight in the Leghorn cockerel chicks. Thus, the small intestine weight and length in boilers fed basal diets were lower significantly than those of birds fed diets with 1α-OH D3, which had nothing to do with feed intake in our experiment. Previous research has shown that the addition of vitamin D2 or D3 results in longer and thinner villi and higher duodenal mucosa weights in rats (Urban and Schedl, 1969; Sampson and Krawitt, 1976). Peebles et al. (2011) reported that percentage of duodenum weight in birds fed 1.5% poultry fat with phytase and 25-OH D3 was greater than that of birds fed 0.75% poultry fat. In the present study, 1α-OH D3 increased the small intestine weight and length of broilers fed with Ca- and P-deficient diets. These data suggest that vitamin D3 and its metabolites can stimulate intestine development.
In conclusion, by increasing feed intake, improving feed efficiency, stimulating small intestine development, and facilitating Ca and P retention in bones, 1α-OH D3 improves growth, tibia quality, and meat color.
ACKNOWLEDGEMENTS
This work was supported by the National Natural Science Foundation of China (No. 31101732), the Henan Province Programs for Science and Technology Development (No. 102102110173), the Natural Science Foundation of Henan Province (No. 2011B230011), and the Youth Foundation of Shangqiu Normal University (No. 2009QN11).