Contribution of Urine and Dung Patches from Grazing Sheep to Methane and Carbon Dioxide Fluxes in an Inner Mongolian Desert Grassland
Article information
Abstract
The effects of sheep urine and dung patches on methane (CH4) and carbon dioxide (CO2) fluxes were investigated during the summer-autumn in 2010, to evaluate their contribution to climate change in a desert grassland in Inner Mongolia, China. Results indicate that the cumulative CH4 emissions for dung patches, urine patches and control plots were − −0.076, −0.084, and −0.114 g/m2 and these were net CH4 sinks during the measured period. The level of CH4 intake from urine and dung plots decreased 25.7%, and 33.3%, respectively, compared with a control plot. CO2 fluxes differed (p<0.01) in urine plots, with an average of 569.20 mg/m2/h compared with control plots (357.62 mg/m2/h) across all sampling days. Dung patches have cumulative CO2 emissions that were 15.9% higher compared with the control during the 55-d period. Overall, sheep excrement weakened CH4 intake and increased CO2 emissions.
INTRODUCTION
Greenhouse gas (GHG) exchange in the soil-atmosphere of terrestrial ecosystems has an important effect on global climate change. China has the world’s second largest grassland area, with grasslands covering 400 million hectares or 42% of total land area. Of this area, 87 million hectares is within the province of Inner Mongolia. Grazing by sheep (Ovis aries), beef cattle (Bos taurus), and goat (Capra hircus) is the main agricultural activity in the Inner Mongolia grassland. Livestock can be an important source of methane (CH4) through direct enteric emissions and decomposition of excreta deposited on grassland (Kulling et al., 2003; Muhammad et al., 2008). Annual CH4 emission is approximately 535 Tg (410–660 Tg) (1 Tg = 1012 g) worldwide with 85 Tg (65–100 Tg) from livestock (IPCC, 2001). In the grazed grassland ecosystems, nutrient cycles are strongly affected by grazing livestock, and much consumed nutrients are returned in the form of dung and urine, which alters soil conditions for CH4 fluxes (Haynes and Williams, 1993).
Estimates of CH4 emissions have been obtained for temperate grasslands (Wang et al., 2009) and alpine meadows of the Qinghai-Tibetan Plateau (Pei et al., 2003) but animal excreta effects were not considered. Feces and urine voided by grazing livestock can give rise to ‘hotspots’ of GHG production. These ‘hotspots’ represent high local additions of readily available carbon (C) resulting in surface emissions of carbon dioxide (CO2) and CH4 (Clemens and Ahlgrimm, 2001; Roland et al., 2004; Saggar et al., 2004; Cardenas et al., 2007). The focus of much research on the effect of excreta on GHG emissions has largely been on cattle urine or dung (Haynes and Williams, 1992, 1993; Flessa et al., 1996; Lovell and Jarvis, 1996; Flessa and Beese, 2000). Sheep and cattle excreta patches are different in nature by the area covered, nutrient concentration in soil, and the height of fall of excreta (Williams and Haynes, 1994). Reports of GHG emissions from sheep dung and urine are few, but with some notable exceptions (Shand et al., 2002; Saggar et al., 2004). In China, Ma et al. (2006) reported emissions from sheep excreta from typical steppe grassland in Inner Mongolia. In addition to typical steppe, meadow steppe and desert steppe comprise the dominant grassland types in Inner Mongolia. GHG emissions from livestock excreta have not been examined in the latter two grassland types.
The objective of this study was to determine the short-term effects of dung and urine patches from sheep on CH4 and CO2 emissions in a desert steppe. In addition, the relation of composition of excreta to emissions was determined. Finally, the results are discussed in relation to the potential contribution of GHG emissions from livestock excreta in the Inner Mongolian desert grassland.
MATERIALS AND METHODS
Site description
This study was conducted from August through October 2010, at an experimental site at the Inner Mongolia Academy of Agriculture and Animal Husbandry Research Station (111° 53′ 46″E, 41° 47′ 17″ N). The site has an elevation of 1,450 m and is in a temperate continental climate, characterized by a short growing season and long cold winter with a frost-free period of 175 d. January is the coldest month with an average temperature of −15.1°C while July is the warmest month with an average temperature of 19.6°C. The average annual precipitation is approximately 280 mm, of which nearly 75% falls during June through September. Monthly rainfall was 188, 184 and 59 mm for August, September and October 2010, respectively (Figure 1). Mean monthly temperature was 18, 13 and 5°C for August, September and October 2010, respectively (Figure 1).
The grassland is dominated by Stipa breviflora Griseb, Artemisia frigida Willd, Cleistogenes songorica (Roshev) Ohwi, and accompanied by Convolvulus ammannii Desr, Heteropappus altaicus (Willd) Novopokr, Neopallasia petinata (Pall) Poljak, Bassia prostrate (L.) A. J. Scott, Caragana stenophylla Pojark, Leymus chinensis (Trin) Tzvelev. Mean vegetation properties from 50×50 cm quadrats (n = 5) at the site were: plant cover of 19%, average height of 5.6 cm, and a density of 13 plant clusters/m2. Above ground biomass averaged 95 g/m2. Mean soil properties for the 0 to 15 cm depth from the quadrats was pH (H2O:Siol = 5:1) 8.3, soil organic matter 41.8 g/kg, total organic nitrogen 0.92 g/kg, NO3--N 8.0 mg/kg, and 2 M KCl - NH4+-N 6.1 mg/kg.
Experimental design
The experiment was conducted from 20 August to 14 October 2010. A complete randomized block design was used with three treatments of urine, dung and no addition (control) with three replicate plots. The dimension of each plot was 50×50 cm.
Sheep dung and urine were collected from 70 animals on 20 August 2010 in metabolism cages and were separately mixed and added to the plots. Prior to placement in the metabolism cages, the animals had grazed the vegetation at the site. The dung had a dry matter content of 85%, ash content of 30%, acid detergent fiber content of 35%, neutral detergent fiber 50.3% and crude fat 6.21%. The urine had a total N content of 10 g/L and urea 4.7 g/L. The freshly collected urine and dung were applied at rates of 550 ml and 380 g, respectively, to each plot (0.25 m2) based on deposition rates for each excretion product (Ma et al., 2006). Control plots received only 550 ml of deionized water. A watering can was used to apply the urine from a height of 50 cm above the ground to simulate sheep urination, and the dung was applied onto the plot surface by hand spreading.
Measurements
Fluxes of CH4 and CO2 were measured using the closed static chamber method (Wang and Wang, 2003). The chamber had a dimension of 50×50×50 cm made of stainless steel. The chamber was placed on a steel base frame driven 10 cm into each plot one day prior to the start of the experiment. The base frame had a channel in which the chamber was inserted and the channel was filled with water to seal the chamber atmosphere. A 9 VDC fan was fixed in to the top wall of each chamber to mix the chamber atmosphere. The temperature of the atmosphere in the chamber and soil at 5 cm were determined using thermocouples and a hand-held reader (HH-25TC, OMEGA Engineering Inc., Stamford, USA). The chamber was covered with a shroud made of camel hair, aluminum foil and white canvas to limit heating of the chamber atmosphere during sampling.
During gas flux determination, a disposable syringe (100 ml) with a 3-way valve was used to collect 200 ml of chamber atmosphere into a sample gas bag (Dalian Hede Technologies Co., Ltd., Dalian, China) at a 10 min interval over a 30 min period. The gas samples were taken three times during the first four days at 10:00, 16:00, and 23:00 h, and then once a day between 10:00 to 13:00 h. The concentrations of CO2 and CH4 in the gas samples were analyzed using a cavity ring-down spectrophotometer (Picarro G1301, Santa Clara, USA). Gas fluxes were calculated using the following equation:
where F is the flux (mg/m2/h) of CO2 or CH4; ρ is the density of 1 mole CO2 or CH4 gas (kg/m3); Δc Δt−1 is the rate of change in gas concentration h−1; V and A are the volume (m3) and the chamber base area (m2), respectively.
Statistical analysis
The fluxes of CH4 and CO2 were analyzed using a repeated measures mixed model with treatment, sampling occasion and treatment×occasion as fixed effects, plot as a random and repeating effect with sample occasion using the MIXED procedure of the Statistical Package for Social Science (SPSS 13.0 for Windows, 2003). The model providing the best-fit covariance structure included compound symmetry. The statistical model used was as follows:
where yijk is the response on sample occasion k for plot j (j = 1–3) in treatment group i (i = 1–3); μ is the overall mean; Ti is the fixed effect of treatment i; Rij the random effect of plot j in treatment i; Pk is the fixed effect of sampling occasion k; (T×P)ik is the fixed interaction effect of treatment i with sample occasion k; eijk is the random error for sample occasion k for plot j in treatment group i. Significant differences in treatment means were determined using the least significant difference (LSD) statistic (p = 0.05).
RESULTS
Methane and respiration
Methane flux from urine and dung plots was greater (p<0.01) than from the control plot during the whole study period (Table 1). On the first day of application (Period 1) dung plots emitted significantly different (p<0.05) amounts of CH4 from other treatments (the peak rate was 161.44 μg/m2/h on the first day of dung application). After five hours, dung patches became a weak net CH4 sink and had a weak release on d 4 and d 55 (Figure 1). From d 2 to d 18 (Period 2) and d 22 to d 55 (Period 3), there were similar patterns of variation in all treatments and CH4 fluxes were not significantly different.
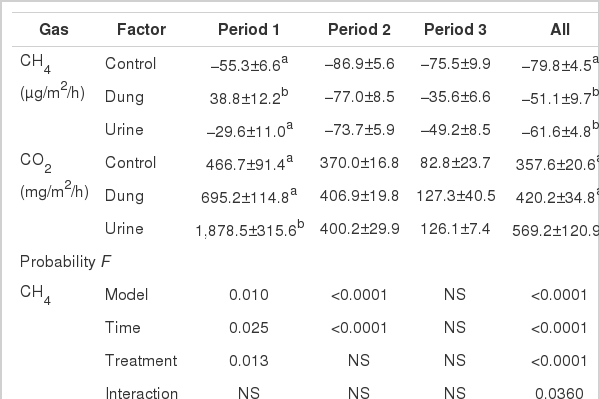
Mean CH4 and CO2 flux (mean±SE) for control, dung and urine treatments for three periods and the whole study period (All)
CO2 emission rates were affected (p<0.01) by treatment, sampling day and their interactions during the whole study period (Table 1). During period 1, CO2 emission rates were affected (p<0.05) by treatment, sampling day and their interactions. After application, CO2 emission increased in <1 h compared with other times in all of the treatments. The second CO2 emissions peak (591.49, 810.09 and 413.51 mg/m2/h in dung, urine and control plots, respectively) was found at h 47, which were 1.4 and 2 times greater than that of the control treatment, respectively (Figure 2).
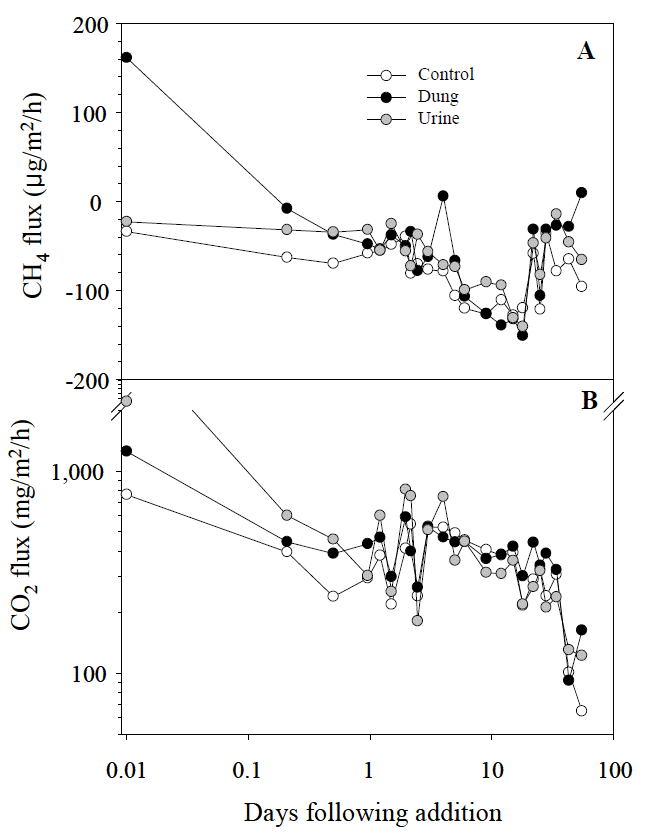
Mean CH4 and CO2 flux from urine, dung, and control plots (n = 3). The negative values of CH4 flux express uptake by soil.
CO2 flux differed (p<0.01) in the urine and control plots during the whole study period and during period 1, but there were no significant differences in period 2 and period 3. The average CO2 flux for urine and control treatments across all sampling days were 569.20 and 357.62 mg/m2/h, respectively (Table 1). Urine patches showed a CO2 emissions peak at 4,575.35 mg/m2/h immediately after application.
Cumulative CO2-C equivalent emission
The cumulative CH4 emission for dung patches, urine patches and control plots were net CH4 sinks (−0.076, −0.084 and −0.114 g/m2, respectively) (Table 2) during the measured period of 55 d following one excreta event. This indicates a decrease of 25.7 and 33.3% for urine, and dung plots compared with the control. Field cumulative CH4 fluxes were −0.053, −0.115 and −1,140 g/ha from urine, dung plots and control plots, respectively. During the 55-d study period, field cumulative CH4 fluxes were −1.758, −4.104 and −33,225.751 g/ha from urine, dung plots and control plots, respectively (Table 2).
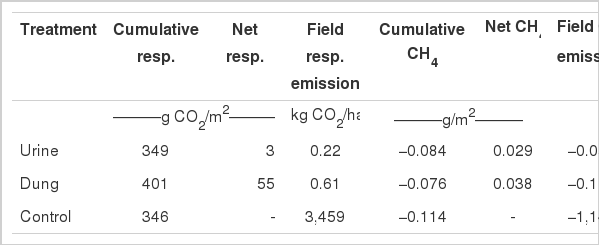
Cumulative respiration and CH4 emission, net emission and cumulative CH4-CO2 equivalent emission potentials over the 55-d study period
The average CO2 and cumulative emissions from dung patches increased 15.9% compared with the control during the 55-d measured period following just one excretion event (Table 2). The dung patches had a greater effect on global warming potentials than urine patches in the Inner Mongolian desert grassland.
DISCUSSION
Methane and respiration
Ma et al. (2006) reported that fresh dung and urine plots decreased significantly (p<0.05) the cumulative CH4 consumption of typical grassland in Inner Mongolia, but their figures were slightly lower than our research results. Plant biomass and stem density might affect CH4 emissions, which correlates with not only clipped plants (Cheng et al., 2007) but also air temperature (Kaharabata et al., 1998; Massé et al., 2003), and manure temperature (Husted, 1994; Park et al., 2006). Simona and Angelo (2005) also found that soil water content and temperature were the main factors driving CH4 fluxes. In the driest season, low water content and higher temperature probably stimulated microbial activity, and even landscape position and biogeochemical disturbances may be primary factors affecting CH4 fluxes (Gulledge and Schimel, 2000). The main cause of the difference between our result and those of others may be the differences in climatic and geographical position between desert grassland and typical grassland.
CH4 consumption in sheep excreta patches was greater in this study than in yak excreta patches in the Qinghai-Tibetan plateau (Lin et al., 2009). CH4 consumption in low-elevation soil increased compared with high-elevation; and mean annual temperature could impact on soil-atmosphere trace gas exchanges, and soil microbial communities (Hart, 2006). However, Lin et al. (2009) reported that dung patches were a strong CH4 source, which was different from our study. Probable reasons are that there are different nutrient transformation characteristics for sheep and yak dung patches which depend on the covered area, nutrient concentration, and differences in shape between sheep and yak feces.
CH4 emissions occurred at d 4 and d 55 in this study. Gulledge and Schimel (2000) reported that nitrogen (N) fertilization inhibited CH4 consumption, a similar result, and Lin et al. (2009) found the combined effect of adding water and N was to increase CH4 production and decrease CH4 consumption in urine patches. Increased CH4 emissions were also correlated with rainfall events, being about 1.2–4 times higher than prior to and following the rainfall event (Kaharabata et al., 1998). Compared to other sites reported in the literature, in desert grasslands, rainfall is less, soil gas permeability is good and CH4 oxidation rates are high. Further research is needed on the relationship between CH4 uptake and hydrothermic factors.
CO2 emissions increased in urine and control plots in our study. In many similar studies, after urine application a jump in CO2 emissions has been found (Bol et al., 2004; Petersen et al., 2004; Lin et al., 2009). The short jump in CO2 might be due to stimulating microbial respiration (Chadwick and Pain, 1997) or urea hydrolysis to CO2 (Saggar et al., 2004). For feces, the CO2 emissions were from heterotrophic respiration, which is an aerobic microbial process (Billings and Ziegler, 2008) regulated by oxygen availability, and usually determined by moisture content (Bauer et al., 2008). Rapid formation of a crust on fresh dung in warm and dry conditions may create anoxic conditions and CO2 production stops. Gulledge and Schimel (2000) reported that only N fertilization had an increasing effect on CO2 fluxes, which was significantly positively correlated with the content of the soil organic C content, the content of soil total N, and the C/N ratio (Geng et al., 2001). However, there was no significant difference between cumulative CO2 emissions from dung patches and control plots in our study. The CO2 emissions peak from fresh dung soils was greater immediately after application than from the control probably because the soil microbial respiration rate was stimulated by additional substrate in the soil (Kelliher et al., 2005).
Urea in animal urine undergoes hydrolysis and is catalysed by the enzyme urease and increased soil solution pH, dissolved organic carbon and total dissolved phosphorus concentrations (Shand et al., 2000), so microbial activity was increased in urine patches, thus stimulating CO2 emissions. Lee et al. (2002) reported that post-rainfall events increased soil CO2 fluxes, but this was not determined in our study.
Cumulative CO2-C equivalent emission
During the 55-d experiment, in sheep excrement plots (1.2%) CH4 flux was 0.02% of non-excrement areas (98.8%). Compared with the control, cumulative CH4 consumption decreased 25.7 and 33.3% from urine and dung plots, respectively. Cumulative CO2 emissions increased 0.9 and 15.9% from urine and dung plots, respectively. In this study, sheep excrement weakened CH4 intake and increased CO2 emissions. Although the other GHG effects (e.g. nitrous oxide) were not considered, livestock excreta has the potential to contribute to global warming in the Inner Mongolia desert grassland.
ACKNOWLEDGEMENTS
This research was supported by the Chinese National Natural Science Foundation (30960072, 31160109), National Commonweal Project (200903060, 201003019), and Inner Mongolia Palmary Youth Project (2011JQ04). The authors gratefully acknowledge the technical assistance of X. Z. Ma and S. P. Wang. We also thank the staff at the research station for their care of the sheep and collection of urine and feces.
