 |
 |
| Anim Biosci > Volume 34(5); 2021 > Article |
|
Abstract
Objective
Methods
Results
ACKNOWLEDGMENTS
Figure 1
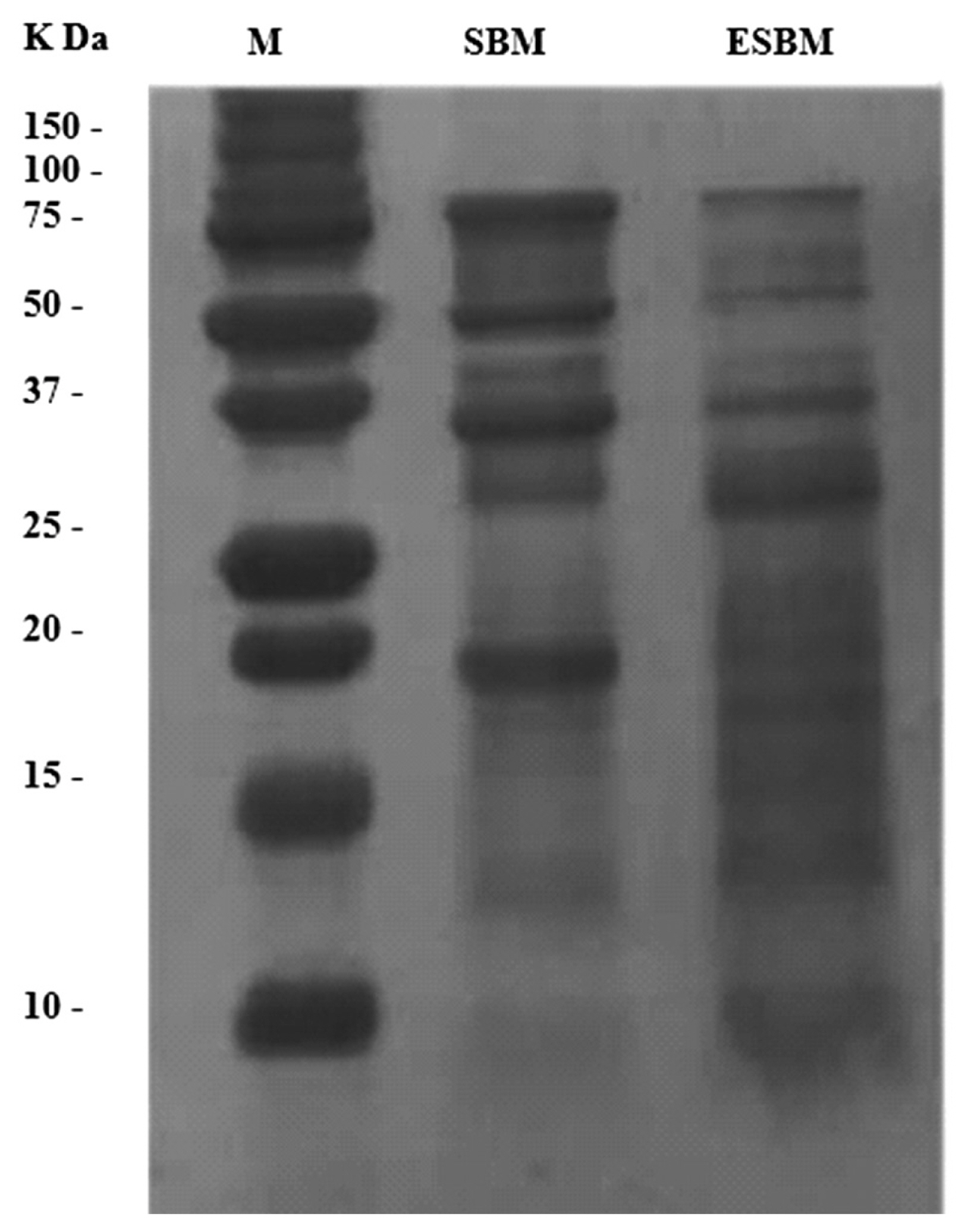
Figure 2
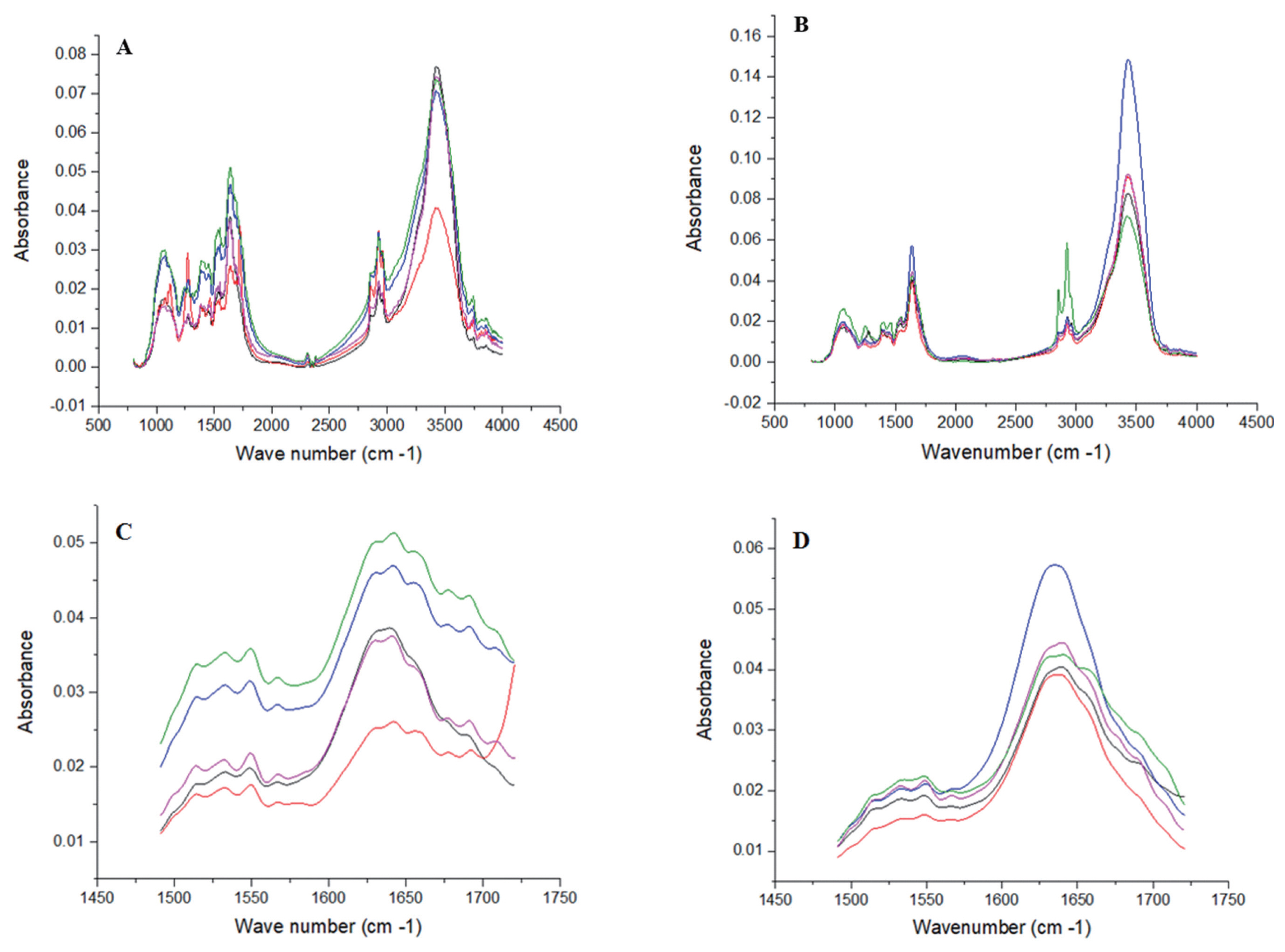
Figure 3

Figure 4

Table 1
| Items | Diets1) | ||||
|---|---|---|---|---|---|
|
|
|||||
| SBM | ESBM25 | ESBM50 | ESBM75 | ESBM100 | |
| Corn grain (fine ground) | 42.2 | 42.2 | 42.2 | 42.2 | 42.2 |
| Soybean meal | 32.4 | 24.2 | 16.2 | 8.1 | 0.0 |
| Extruded SBM | 0.0 | 8.2 | 16.3 | 24.3 | 32.4 |
| Barley | 14.6 | 14.6 | 14.6 | 14.6 | 14.6 |
| Wheat bran | 4.2 | 4.2 | 4.2 | 4.2 | 4.2 |
| Chopped alfalfa hay | 2.4 | 2.4 | 2.4 | 2.4 | 2.4 |
| Mineral supplement2) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Vitamin supplement3) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| DCP | 0.6 | 0.6 | 0.6 | 0.6 | 0.6 |
| Salt | 0.6 | 0.6 | 0.6 | 0.6 | 0.6 |
| Na- bicarbonate | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Chemical composition (% DM) | |||||
| CP | 23.3 | 23.1 | 23.0 | 22.8 | 22.6 |
| ADF | 7.4 | 7.3 | 7.3 | 7.3 | 7.2 |
| aNDF | 15.5 | 15.7 | 16.0 | 16.3 | 16.6 |
| CA | 8.8 | 8.9 | 8.9 | 9.0 | 9.0 |
| EE | 2.7 | 2.8 | 2.9 | 3.0 | 3.1 |
| NFC4) | 49.7 | 49.5 | 49.3 | 49.0 | 48.8 |
| Starch | 42.1 | 42.0 | 41.8 | 41.6 | 41.5 |
| Predicted energy contents (kcal/kg DM) | |||||
| ME | 2,790 | 2,790 | 2,800 | 2,800 | 2,810 |
| NEg | 1,220 | 1,230 | 1,230 | 1,230 | 1,240 |
DM, dry matter; DCP, di-calcium phosphate; CP, crude protein; ADF, acid detergent fiber; aNDF, neutral detergent fiber analyzed with heat stable α-amylase; CA, crude ash; EE, ether extract; NFC, non-fiber carbohydrates; ME, metabolizable energy; NEg, net energy for gain.
1) SBM, diet based on soybean meal; ESBM25, 25% of soybean meal replaced by extruded soybean meal; ESBM50, 50% of soybean meal replaced by extruded soybean meal; ESBM75, 75% of soybean meal replaced by extruded soybean meal; ESBM100, 100% of soybean meal replaced by extruded soybean meal.
2) Contained per kilogram of supplement: 250,000 IU of vitamin A, 50,000 IU of vitamin D, 1,500 IU of vitamin E.
Table 2
| Items | SBM | ESBM | SEM | p-value |
|---|---|---|---|---|
| Chemical composition (% DM) | ||||
| CP | 55.8 | 53.4 | 0.65 | 0.07 |
| EE | 0.8 | 2.1 | 0.26 | 0.03 |
| ADF | 9.5 | 9.0 | 0.45 | 0.39 |
| CA | 7.8 | 8.3 | 1.30 | 0.72 |
| aNDF | 16.6 | 20.5 | 1.31 | 0.11 |
| Crude protein fractions1) (% CP) | ||||
| A | 13.5 | 7.5 | 0.56 | <0.01 |
| B1 | 1.2 | 2.4 | 0.07 | <0.01 |
| B2 | 61.0 | 60.7 | 5.09 | 0.94 |
| B3 | 14.2 | 18.7 | 4.23 | 0.40 |
| C | 10.1 | 10.8 | 1.21 | 0.60 |
| FTIR protein molecular structures2) | ||||
| Amide I | 0.0397 | 0.0447 | 0.0039 | 0.38 |
| Amide II | 0.0257 | 0.0200 | 0.0025 | 0.14 |
| Amide I:amide II | 1.5706 | 2.2499 | 0.1364 | <0.01 |
| Alpha helix | 0.0655 | 0.0498 | 0.0336 | 0.75 |
| Beta sheets | 0.4315 | 0.3366 | 0.1544 | 0.67 |
| Alpha helix:beta sheets | 0.2035 | 1.1224 | 0.7391 | 0.40 |
| Others | 0.5031 | 0.6136 | 0.1663 | 0.65 |
FTIR, fourier transform infrared spectroscopy; SBM, soybean meal; ESBM, extruded soybean meal; SEM, standard error of means; CP, crude protein; EE, ether extract; ADF, acid detergent fiber; CA, crude ash; aNDF, neutral detergent fiber analyzed with heat stable α-amylase.
1) Protein fractions according to CNCPS include PA, fraction of CP that is instantaneously solubilized at time zero and determined as NPN; PB1, soluble true protein calculated as buffer soluble CP minus non-protein nitrogen; PB2, intermediate degradable true protein calculated as CP – (PA + PB1 + PB3 + PC); PB3, slowly degradable true protein calculated as NDICP – ADICP; PC, undegradable CP determined as ADICP.
Table 3
| Items | Treatments1) | SEM | Level of significant (p-value) | Response curves | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||||
| SBM | ESBM25 | ESBM50 | ESBM75 | ESBM100 | Treat | Week | Treat×week | Linear | Quadratic | Qubic | ||
| Pre-weaning (kg) | ||||||||||||
| iBW | 39.8 | 39.6 | 39.0 | 39.8 | 42.8 | 1.41 | 0.16 | - | - | 0.68 | 0.72 | 0.78 |
| WW | 78.6 | 77.7 | 79.4 | 80.9 | 83.7 | 1.9861 | 0.27 | - | - | 0.04 | 0.33 | 0.84 |
| wBW | 58.2bc | 57.1c | 58.2bc | 59.0b | 60.7a | 0.48 | <0.01 | <0.01 | 0.99 | <0.01 | <0.01 | 0.18 |
| SI (g/d) | 379.2 | 371.5 | 364.6 | 321.8 | 407.6 | 28.57 | 0.054 | <0.01 | 0.85 | 0.91 | 0.05 | 0.05 |
| tDMI (g/d) | 981.9 | 974.3 | 967.4 | 924.5 | 1,010.3 | 28.57 | 0.054 | <0.01 | 0.85 | 0.91 | 0.05 | 0.05 |
| ADG (g/d) | 526.9 | 506.2 | 540.0 | 546.6 | 568.7 | 29.85 | 0.30 | <0.01 | 0.82 | 0.06 | 0.46 | 0.55 |
| FCR | 2.4 | 2.4 | 2.8 | 2.4 | 2.7 | 0.55 | 0.92 | 0.06 | 0.95 | 0.74 | 0.84 | 0.82 |
| Post weaning | ||||||||||||
| BW (kg) | 87.0b | 85.7b | 90.7ab | 91.2ab | 96.3a | 2.18 | <0.01 | <0.01 | <0.01 | <0.01 | 0.15 | 0.71 |
| DMI (g/d) | 1,885.5 | 1,790.0 | 2,074.0 | 1,998.1 | 2,241.0 | 158.16 | 0.06 | <0.01 | 0.95 | <0.05 | 0.45 | 0.86 |
| ADG (g/d) | 694.3ab | 645.5b | 820.6ab | 781.4ab | 925.9a | 70.37 | 0.05 | 0.14 | 0.84 | <0.01 | 0.51 | 0.85 |
| FCR | 3.4 | 3.7 | 3.8 | 3.2 | 3.2 | 1.11 | 0.10 | 0.02 | 0.95 | 0.25 | 0.28 | 0.05 |
| Entire period | ||||||||||||
| wBW (kg) | 64.4bc | 63.2c | 65.1b | 65.9b | 68.3a | 0.62 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | 0.21 |
| DMI (g/d) | 1,175.5ab | 1,149.1b | 1204.5ab | 1,154.6ab | 1,274.1a | 41.56 | 0.02 | <0.01 | 0.85 | 0.03 | 0.09 | 0.34 |
| ADG (g/d) | 564.0ab | 536.9b | 599.4ab | 596.8ab | 643.2a | 30.92 | 0.01 | <0.01 | 0.85 | <0.01 | 0.32 | 0.55 |
| FCR | 2.6 | 3.3 | 3.0 | 2.6 | 2.8 | 0.54 | 0.62 | <0.01 | 0.85 | 0.69 | 0.53 | 0.18 |
| fBW (kg) | 90.8 | 89.7 | 93.5 | 95.3 | 100.0 | 3.03 | 0.16 | - | - | 0.02 | 0.48 | 0.72 |
SEM, standard error of means; iBW, initial body weight; SI, starter intake; tDMI, total dry matter intake, FCR, feed conversion ratio; ADG, average daily gain; BW, body weight; fBW, final body weight.
Table 4
| Items | Treatments1) | SEM | Level of significance | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|||||||||
| SBM | ESBM25 | ESBM50 | ESBM75 | ESBM100 | Treat | Linear | Quadratic | Cubic | ||
| Week 4 (%) | ||||||||||
| DMD | 57.4 | 66.4 | 61.5 | 57.6 | 66.2 | 5.04 | 0.23 | 0.44 | 0.98 | 0.03 |
| OMD | 59.3 | 68.0 | 60.1 | 58.5 | 67.0 | 5.02 | 0.23 | 0.61 | 0.73 | 0.03 |
| CPD | 74.2b | 87.2a | 74.1b | 79.6ab | 83.8a | 3.32 | 0.04 | 0.28 | 0.93 | 0.02 |
| Week 10 (%) | ||||||||||
| DMD | 63.4 | 61.5 | 68.3 | 67.4 | 62.8 | 3.05 | 0.13 | 0.47 | 0.11 | 0.08 |
| OMD | 65.0 | 63.5 | 70.0 | 68.9 | 63.9 | 2.32 | 0.20 | 0.67 | 0.11 | 0.12 |
| CPD | 71.0 | 70.2 | 75.9 | 78.2 | 72.3 | 2.53 | 0.16 | 0.19 | 0.16 | 0.08 |
| Week 13 (%) | ||||||||||
| DMD | 56.2 | 57.9 | 56.0 | 62.3 | 64.1 | 2.92 | 0.21 | 0.04 | 0.45 | 0.92 |
| OMD | 58.0 | 59.9 | 57.1 | 63.9 | 65.7 | 3.03 | 0.23 | 0.06 | 0.41 | 0.97 |
| CPD | 77.2b | 78.7ab | 72.6b | 81.0ab | 83.7a | 2.34 | 0.03 | 0.05 | 0.06 | 0.45 |
SEM, standard error of means; DMD, dry matter digestibility; OMD, organic matter digestibility; CPD, crude protein digestibility.
Table 5
| Items | Treatments1) | SEM | Level of significant (p-value) | Response curves | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||||
| SBM | ESBM25 | ESBM50 | ESBM75 | ESBM100 | Treat | Week | Treat×week | Linear | Quadratic | Qubic | ||
| Pre-weaning | ||||||||||||
| IHeight2) (cm) | 79.1 | 78.6 | 78.1 | 78.8 | 79.7 | 0.94 | 0.80 | - | - | 0.63 | 0.25 | 0.94 |
| iHeart girth2) (cm) | 77.9 | 78.3 | 77.6 | 78.7 | 79.5 | 1.00 | 0.70 | - | - | 0.25 | 0.48 | 0.70 |
| Height (cm) | 86.2ab | 85.4b | 86.3ab | 86.4ab | 87.2a | 0.53 | 0.03 | <0.01 | 0.95 | 0.01 | 0.13 | 0.42 |
| Heart girth (cm) | 88.5b | 87.7bc | 87.5c | 89.1ab | 89.7a | 0.31 | <0.01 | <0.01 | 0.85 | <0.01 | <0.01 | 0.02 |
| Fecal score | 3.0 | 3.1 | 3.1 | 3.1 | 3.1 | 0.069 | 0.94 | <0.01 | 0.75 | 0.47 | 0.64 | 0.96 |
| Post-weaning | ||||||||||||
| Height (cm) | 95.9b | 95.8b | 96.0ab | 96.6ab | 97.8a | 0.65 | 0.02 | <0.01 | 0.92 | <0.01 | 0.08 | 0.45 |
| Heart girth (cm) | 100.3ab | 99.1b | 100.3ab | 101.0ab | 102.3a | 0.74 | <0.01 | <0.01 | 0.84 | <0.01 | 0.02 | 0.24 |
| Fecal score | 3.4 | 3.3 | 3.3 | 3.4 | 3.4 | 0.13 | 0.51 | 0.10 | 0.69 | 0.65 | 0.13 | 0.37 |
| Entire period | ||||||||||||
| Height (cm) | 88.3ab | 87.6b | 88.4ab | 88.6ab | 89.5a | 0.31 | <0.01 | <0.01 | 0.88 | <0.01 | 0.04 | 0.49 |
| Heart girth (cm) | 91.0bc | 90.1c | 90.2c | 91.6ab | 92.4a | 0.21 | <0.01 | <0.01 | 0.75 | <0.01 | <0.01 | <0.01 |
| Fecal score | 3.2 | 3.2 | 3.1 | 3.2 | 3.1 | 0.06 | 0.93 | <0.01 | 0.87 | 0.41 | 0.92 | 0.78 |
1) SBM, diet based on soybean meal; ESBM25, 25 percent of soybean meal replaced by extruded soybean meal; ESBM50, 50 percent of soybean meal replaced by extruded soybean meal; ESBM75, 75 percent of soybean meal replaced by extruded soybean meal; ESBM100, 100 percent of soybean meal replaced by extruded soybean meal.
Table 6
| Items | Treatments1) | SEM | Level of significant (p-value) | Response curves | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
||||||||||
| SBM | ESBM25 | ESBM50 | ESBM75 | ESBM100 | Treat | Week | Treat×week | Linear | Quadratic | Qubic | ||
| Pre-weaning period | ||||||||||||
| Glucose (mg/dL) | 92.8 | 98.0 | 104.3 | 106.3 | 106.2 | 5.98 | 0.43 | <0.01 | 0.75 | 0.07 | 0.50 | 0.87 |
| Urea (mg/dL) | 21.6ab | 19.0ab | 16.6b | 20.8ab | 22.0a | 1.26 | 0.02 | 0.04 | 0.68 | 0.49 | <0.01 | 0.44 |
| Albumin (g/dL) | 3.1 | 3.0 | 2.9 | 3.1 | 3.0 | 0.05 | 0.11 | 0.32 | 0.85 | 0.95 | 0.28 | 0.26 |
| Total protein (g/dL) | 6.4 | 6.5 | 6.3 | 6.1 | 6.2 | 0.21 | 0.61 | <0.01 | 0.92 | 0.15 | 0.97 | 0.48 |
| Globulin (g/dL) | 3.4 | 3.4 | 4.0 | 3.0 | 3.1 | 0.28 | 0.12 | <0.01 | 0.69 | 0.27 | 0.20 | 0.44 |
| BHBA (mg/dL) | 0.25 | 0.25 | 0.27 | 0.28 | 0.32 | 0.026 | 0.3181 | <0.01 | 0.85 | 0.04 | 0.58 | 0.81 |
| Post-weaning | ||||||||||||
| Glucose (mg/dL) | 65.8 | 60.3 | 64.5 | 65.8 | 71.5 | 6.44 | 0.80 | - | - | 0.41 | 0.43 | 0.81 |
| Urea (mg/dL) | 25.1 | 27.2 | 29.0 | 25.6 | 30.0 | 3.09 | 0.76 | - | - | 0.41 | 0.95 | 0.42 |
| Albumin (g/dL) | 3.1 | 3.1 | 3.1 | 3.2 | 3.0 | 0.07 | 0.51 | - | - | 0.86 | 0.39 | 0.24 |
| Total protein (g/dL) | 6.3 | 6.6 | 6.0 | 6.3 | 6.1 | 0.22 | 0.36 | - | - | 0.26 | 0.93 | 0.60 |
| Globulin (g/dL) | 3.2 | 3.5 | 2.9 | 3.1 | 3.1 | 0.22 | 0.44 | - | - | 0.29 | 0.86 | 0.38 |
| BHBA (mg/dL) | 0.38 | 0.35 | 0.48 | 0.48 | 0.40 | 0.096 | 0.83 | - | - | 0.57 | 0.54 | 0.47 |
| Entire period | ||||||||||||
| Glucose (mg/dL) | 83.4 | 85.5 | 91.2 | 93.0 | 94.6 | 6.49 | 0.13 | <0.01 | 0.77 | <0.01 | 0.76 | 0.84 |
| Urea (mg/dL) | 22.7 | 21.8 | 20.7 | 22.4 | 24.7 | 1.33 | 0.32 | <0.01 | 0.65 | 0.28 | 0.07 | 0.88 |
| Albumin (g/dL) | 3.1 | 3.1 | 3.0 | 3.1 | 3.0 | 0.04 | 0.06 | 0.06 | 0.95 | 0.88 | 0.68 | 0.11 |
| Total protein (g/dL) | 6.4 | 6.5 | 6.2 | 6.1 | 6.1 | 0.16 | 0.36 | <0.01 | 0.82 | 0.07 | 0.97 | 0.40 |
| Globulin (g/dL) | 3.3 | 3.6 | 3.6 | 3.0 | 3.1 | 0.20 | 0.20 | <0.01 | 0.66 | 0.17 | 0.27 | 0.31 |
| BHBA (mg/dL) | 0.28 | 0.28 | 0.32 | 0.33 | 0.34 | 0.028 | 0.38 | <0.01 | 0.49 | 0.05 | 0.96 | 0.72 |
1) SBM, diet based on soybean meal; ESBM25, 25 percent of soybean meal replaced by extruded soybean meal; ESBM50, 50 percent of soybean meal replaced by extruded soybean meal; ESBM75, 75 percent of soybean meal replaced by extruded soybean meal; ESBM100, 100 percent of soybean meal replaced by extruded soybean meal.






 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print





