Effect of bamboo grass (Tiliacora triandra, Diels) pellet supplementation on rumen fermentation characteristics and methane production in Thai native beef cattle
Article information
Abstract
Objective
The objective of this study was to investigate the effect of bamboo grass (Tiliacora triandra, Diels) pellet (Bamboo-Cass) supplementation on feed intake, nutrient digestibility, rumen microbial population and methane production in Thai native beef cattle.
Methods
Four Thai native beef cattle bulls (190±2 kg) were randomly allotted to four respective dietary treatments in a 4×4 Latin square design. Treatments were the varying levels of Bamboo-Cass supplementation at 0, 50, 100, and 150 g/head/d, respectively. Rice straw was fed ad libitum and the concentrate offered at 0.5% of body weight.
Results
Under this experiment, the findings revealed that acetate and butyrate production were decreased (p<0.05), propionate increased (p<0.05), whilst ruminal NH3-N concentration was decreased (p<0.05) by supplementation of Bamboo-Cass at 150 g/head/d. Moreover, rice straw intake, and microbial population were linearly increased (p<0.05), while methane production was decreased (p<0.05).
Conclusion
The results from the present study indicate that supplementation of Bamboo-Cass at 150 g/head/d significantly enhanced feed intake, decreased protozoa and increased bacterial population, rumen fermentation efficiency while decreased methane production. Therefore, Bamboo-Cass as a supplement is promising as a rumen enhancer in beef cattle fed on rice straw.
INTRODUCTION
Greenhouse gases including CH4, CO2, and N2O are emitted from rumen fermentation and results in a loss of dietary energy intake accounting up to 12%. There are many possible CH4 mitigation strategies but one of the most promising implementations could be the dietary manipulating option which has been reported in the previous studies [1,2]. There have been numerous reports on the potential use of plant secondary compounds, particularly the use of condensed tannins and crude saponins on rumen microorganisms and the consequent fermentation end-products in mitigating rumen CH4 production [3,4].
Bamboo grass (Tiliacora triandra, Diels) is a tropical climbing plant with abundant green leaves and yellow flower, belonging to Menispermaceae family. It has been known as a local resource plant in sub-tropical and tropical regions, used for medicine, cosmetics, and household products [5,6]. Bamboo grass is rich in phenolic compounds which are useful as a source of bioactive compounds [7]. Previous studies were conducted using this plant to investigate their antioxidant activities, and bioactive role as a medical plant [6,8,9]. Bamboo grass contains plant secondary compounds of condensed tannins (2.3% of dry matter [DM]) and crude saponins (1.3% of DM), and is potential feed resource for use as a strategic supplement in ruminant production [10,11]. However, information on the potential use of this plant in ruminant feeding is not yet available. Therefore, the objective of this study was to determine the effect of bamboo grass pellet (Bamboo-Cass) on feed intake, nutrient digestibility, rumen microbial population, and methane production in Thai beef cattle fed on rice straw.
MATERIALS AND METHODS
The experimental area and animals care
This experiment was done at the Ruminant Nutrition and Metabolism Research Center, Tropical Feed Resource Research and Development Center (TROFREC), Department of Animal Science, Faculty of Agriculture, Khon Kaen University (KKU), Khon Kaen, Thailand. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed and allowed by the Khon Kaen University Animal Ethics Committee, ancillary to the Ethic of animal Experimentation of National Research Council of Thailand.
Animals, feeds and experimental design
All experimental beef cattle were treated with vitamin AD3E injection (20 mL/head) and dewormed before imposing the respective treatments. They were raised in individual pens of 3×4 m size where the water and mineral block were always available. Four Thai native beef cattle bulls (190±2 kg) were randomly assigned to receive four dietary treatments in a 4×4 Latin square design and were raised in animal pens with permanent roof. Animals were fed ad libitum with rice straw and water supply. The 14% crude protein (CP) of concentrate was formulated and provided to all animals at 0.5% body weight (BW). All animals received four varying levels of Bamboo-Cass supplementation at 0, 50, 100, and 150 g/head/d, respectively. Each of the periods lasted for 21 days, the first 14 days was for adaptation and measurement of intake, while the last 7 days was for sample collections when the experimental animals were moved to the metabolism crates for total collections.
Bamboo grass (Tiliacora triandra, Diels) was harvested from fresh leaves from the climbing tree and sun-dried for use in the experiment. Bamboo grass pellets were made by combining bamboo grass (90%), cassava chip (9%), and molasses (1%) (Bamboo-Cass) then sun-dried to achieve at least 85% DM before storage for later feeding to the experimental animals [12].
Data collection and sampling procedures
All procedures and collection details of the feeds, urine, rumen fluid, blood samples, and rumen methane calculation were done as described by [13]. Animals were fed twice daily at 08:00 hours in the morning and 16:00 hours in the afternoon. Feed offered and refusal feeds were recorded throughout the experimental period for calculating feed intake. Feed and fecal samples were collected during the 7 days feeding period when the animals were in the metabolism crates for analyzing DM, for each period then stored at −20°C for later chemical analysis. The samples were divided into two parts, the first part was analyzed for DM, and the second part kept for analysis of ash, CP, and acid detergent fiber (ADF) [14], neutral detergent fiber (NDF) [15]. The content of condensed tannins of bamboo grass and Bamboo-Cass were analyzed by using the modified vanillin-HCL method [16]. Crude saponins were analyzed by using methanol extraction [17].
Rumen fluid was taken from the middle part of rumen at 0 and 4 hour post morning feeding on the last day of each period prior to morning feeding using a tube connected with the vacuum pump. Rumen fluid was measured immediately for pH (HANNA Instruments HI 8424 microcomputer, Singapore) and strained through four layers cheesecloth. Fluid sample was divided into three parts for continuing analysis. The first 45 mL of rumen fluid sample was collected and kept in plastic bottle to which 5 mL of 1 M H2SO4 were added for stopping the process of microbial fermentation and then centrifuged with 3,000×g for 10 minutes for volatile fatty acids (VFAs) analysis in the laboratory using high-performance liquid chromatography instruments by water and Novapak model 600E; water mode 1484UV detector; column Novapak C18; column size 3.9×300 mm; mobile phase 10 mM H2PO4 (pH 2.5) [18] and NH3-N analysis [14]. The second part (1 mL of rumen fluid) was quickly fixed with 10% of formalin solution (1:9 v/v, rumen fluid:10% formalin) in plastic bottles for measurement of the microbial population. The total direction counts, method of microbial populations were calculated based on the use of a haemacytometer (Boeco, Singapore) [19]. The third part (10 mL of rumen fluid) was used for variable bacteria counts (cellulolytic, proteolytic, and amylolytic) using the roll-tube technique [20].
Blood samples were collected (about 10 mL) from the jugular vein of all experimental animals and then transferred to tubes containing ethylenediaminetetraacetic acid at the same time as the collection of rumen fluid at 0 and 4 hour post morning feeding. Blood samples were quickly put in the refrigerator for 1 hour and then centrifuged for 20 min to separate plasma from the whole blood. Plasma was stored at −20°C for later analysis of blood urea nitrogen (BUN) [21].
Calculation of ruminal CH4 production was estimated by using VFA proportions by CH4 production = 0.45(acetate, C2) −0.275(propionate, C3)+0.4(butyrate, C4) [22].
Statistical analysis
All data were statistically analyzed in a 4×4 Latin square design using general linear model procedure [23] according to the model: Yijk = μ+Mi+Aj+Pk+ɛijk; where Yijk = observation from animal j, receiving diet i, in the period k; μ = the overall mean; Mi = effect of treatment; Aj = effect of animal; Pk = effect of period, and ɛijk = residual effect. Mean separations with a significantly different value (p<0.05) and difference between treatment means determined by Duncan’s new multiple range test [24]. Trend of Bamboo-Cass level responses was performed by using the orthogonal polynomial.
RESULTS
Chemical composition in feed ingredients
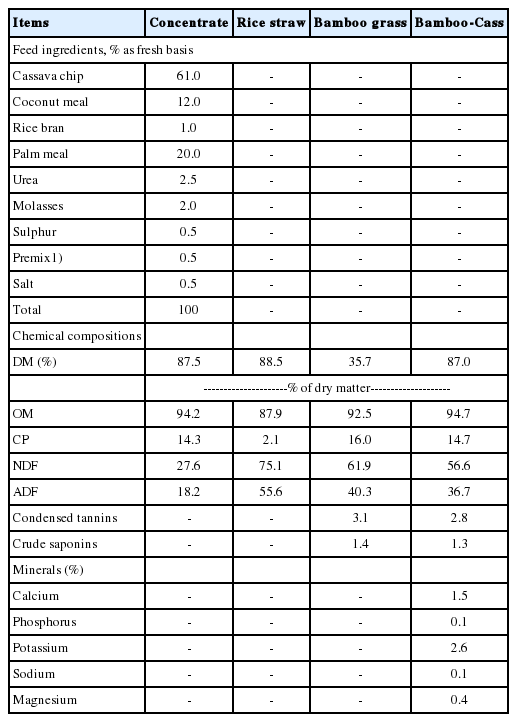
Table 1 presents the data of concentrate ingredients and chemical composition of the experimental feeds. The concentrate was formulated using high level of cassava chip (61%) as the main energy source, oil palm meal (20%) and urea (2.5%) as protein and non-protein nitrogen source, respectively. Important concentrations of nutrients such as CP, NDF for concentrate, rice straw, Bamboo grass and Bamboo-Cass were 14.3%, 27.6%; 2.1%, 75.1%; 16.0%, 61.9%; 14.7%, 56.6%, respectively. Plant secondary compounds namely condensed tannins and crude saponins for bamboo grass and Bamboo-Cass were 3.1%, 1.4%; 2.8%, 1.3%, respectively.
Effect of Bamboo-Cass supplementation on feed intake and nutrients digestibility
Concerning feed intakes (Table 2), as clearly found, rice straw intakes was linearly enhanced (p<0.05) by the supplementation, which raised (p<0.05) the total intake presented as in terms of %BW and g/kg BW0.75. Regarding nutrients digestibility of DM, organic matter, CP, NDF, and ADF were increased (p<0.05) when fed the higher level of Bamboo-Cass supplementation.
Effect of Bamboo-Cass supplementation on rumen fermentation
Rumen parameters are shown in Table 3. The rumen pH (6.7 to 6.8) and temperature (39.2°C to 39.5°C) were in normal range. There were no influences from the dietary treatments on total VFAs (100.4 to 106.8 mmol/L) and C4 (10.4% to 10.8%), while the C2 and C3 concentration, and C2:C3 ratio were different among treatments (p<0.05). Level of Bamboo-Cass supplementation decreased C2, while increased C3, as well as the ratio of C2:C3 (p<0.05). CH4 productions (mmol/L) were different by Bamboo-Cass supplementation (p<0.05). Furthermore, ruminal NH3-N (mg/dL) concentrations were influenced (p<0.05), while BUN (mg/dL) was similar among treatments.
Effect of Bamboo-Cass supplementation on microorganism population
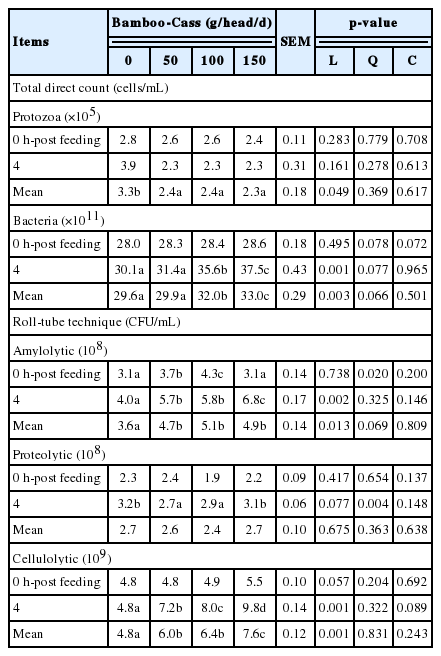
Table 4 presents the data of rumen protozoal and bacterial population as influenced by the treatments. Bacterial populations were significantly increased by increasing Bamboo-Cass supplementation, while protozoa populations were linearly reduced (p<0.05). Using roll-tube technique, the rumen amylolytic bacteria (p<0.05) and cellulolytic bacterial populations (p<0.01) were enhanced, while proteolytic count remained unchanged.
DISCUSSION
Chemical composition in feed ingredients
The protein content of bamboo grass was 16% of DM, which was within the range of the previous values [9], but higher than the value in literature [7], and lower than the result of previous work [10]. However, condensed tannins and crude saponins contained in bamboo grass was 3.1% and 1.4% of DM which were higher than those in literature [10,11]. The differences occurred could be due to the stage of growth, drying process, harvesting method etc.
Effect of Bamboo-Cass supplementation on feed intake and nutrients digestibility
The linearly increase of DM intake could be explained with the effect of Bamboo-Cass supplementation that contained additional protein and plant secondary compounds as condensed tannins and crude saponins when fed higher level of Bamboo-Cass. These findings agree with the previous studies [25,26], who reported that supplementation of higher protein with suitable level of fibrous fraction would improve rumen ecology in fermentation and feed degradation, while the cellulolytic bacteria was increased. The digestibility of nutrients would be improved due to ruminal pH, whereas microbial degradation of fiber was inhibited at pH = 6.0 or lower [27]. It enhanced ruminal pH (optimal 6.7 to 6.8) which would increase fiber degradability [28,29]. Furthermore, apparent nutrients digestibilities were affected by Bamboo-Cass supplementation, mainly CP, NDF, and ADF. The decrease of apparent nutrient digestibility of CP was correlated with low NH3-N concentration in rumen which could be explained by the effect of condensed tannins in Bamboo-Cass being bound with dietary protein as the protein-tannins complexes which would not degrade in the rumen [30]. However, NDF and ADF were also decreased by supplementation higher level. These could be explained as the effect of tannins could result lower digestibility [31]. The increase of feed intake, feed digestibility, and microbial activity was improved when degradable protein was added [32].
Effect of Bamboo-Cass supplementation on rumen fermentation
The rumen pH is a key factor to efficient rumen fermentation by activities of microorganisms. The weak acid pH between 6.7-6.9 has been reported the optimal for activity of cellulolytic bacteria namely Ruminococcus albus, Ruminococcus flavefaciens, Fibrobacter succinogenes. Clearly, roughage and concentrate ratio influences ruminal pH and fermentation end-products with a higher supplementation level of concentrate resulting in a higher pH with a higher roughage intake and more C3 production [13]. The values of ruminal pH (6.7 to 6.8) and temperature (39.3°C to 39.5°C) were similar and rather stable, which were in the normal range of rumen ecology for fermentation by rumen microbes [28,29]. Effect of Bamboo-Cass on VFA revealed that C2 and C4 were decreased while C3 increased at 150 g/head/d supplementation. This result could be explained that when C2 decreased, C3 could be increased while the absolute level of C3 remained unchanged [33,34]. When acetate to propionate ratio decreased, total VFAs would be decreased with supplementation with high level of dietary condensed tannins [35]. The effect of dietary condensed tannins changed propionate and acetate ratio in the in vitro study, by giving the highest propionate and low acetate to propionate ratio [36]. Furthermore, C2 inhibitory effect could decrease the C2 to C3 ratio and inhibiting methanogens in the rumen [37]. This decline of estimated CH4 production (p<0.05), resulted by increasing of C2 to C3 ratio [37], as well as due to decreasing of protozoal population by diets containing condensed tannins [38]. In addition, the formation processes of both propionate and CH4 require H2, so the increase of propionate could be a competitive pathway with CH4 production in the rumen [22]. Supplementation of tannic acid at 6.5, 13.0, and 26.0 g/kg of DM in beef cattle significantly decreased CH4 production from 11.1%, 14.7%, and 33.6%, respectively [36]. Tannins could play the role in inhibiting rumen methanogens’ growth and activity by binding with microbial enzymes and proteins as CH4 production was decreased [39,40]. Moreover, NH3-N concentration was also decreased when supplemented with high level of Bamboo-Cass. This result was similar to the previous study [31] who reported that condensed tannins in the diet were bound by forming protein-tannin complexation, resulting in decreasing protein degradation and NH3-N production. Additionally, it was reported that condensed tannins could bind with protein, resulting in lower NH3-N concentration [36]. This result could be due to lower NH3-N concentration from reducing protein degradability to NH3-N in the rumen, hence enhanced protein flowing to intestine [41]. However, there were no effects of saponins on feed intake or digestibility, but saponins were directly affected protozoa numbers and bacterial population by inhibiting the protozoa population and increasing bacterial population resulting a low of methane production when supplementation was at a high level [42].
Effect of Bamboo-Cass supplementation on microorganism population
Rumen protozoal population decreased, while bacterial population increased, would be due to the effect of condensed tannins and crude saponins in bamboo grass, which has the potential to suppress protozoal growth, as indicated by previous work [43]. Importantly, the increase in bacterial population and the decrease in protozoal population were found when ruminates were fed with the plants containing condensed tannins [40,44]. Moreover, supplementations of Bamboo-Cass, resulted in increasing the amylolytic bacteria, and cellulolytic bacteria while proteolytic bacterial was not affected with 150 g/head/d of Bamboo-Cass supplementation, which could be due to the decrease of protozoal population and hence, the methanogens [45].
CONCLUSION
Supplementation of Bamboo-Cass can be used as a rumen dietary enhancer as it improved rumen fermentation end-products (C3), reduced methane by suppressing protozoal population and increased nutrients digestibility.
ACKNOWLEDGMENTS
The authors would like to express their most sincere thanks to Tropical Feed Resources Research and Development Center (TROFREC), Khon Kaen University, Royal Scholarship under Her Royal Highness Princess Maha Chakri Sirindhorn Education Project to the Kingdom of Cambodia, Thailand Research Fund (TRF) through the International Research Network (IRN) program and The Royal Golden Jubilee (TRF-IRN 57W0002) and TRF-RGJ 598001 for providing scholarship for the first author and provision of research funds and facilities.
Notes
CONFLICT OF INTEREST
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.