Relationships between Single Nucleotide Polymorphism Markers and Meat Quality Traits of Duroc Breeding Stocks in Korea
Article information
Abstract
This study was conducted to determine the relationships of five intragenic single nucleotide polymorphism (SNP) markers (protein kinase adenosine monophosphate-activated γ3 subunit [PRKAG3], fatty acid synthase [FASN], calpastatin [CAST], high mobility group AT-hook 1 [HMGA1], and melanocortin-4 receptor [MC4R]) and meat quality traits of Duroc breeding stocks in Korea. A total of 200 purebred Duroc gilts from 8 sires and 40 dams at 4 pig breeding farms from 2010 to 2011 reaching market weight (110 kg) were slaughtered and their carcasses were chilled overnight. Longissimus dorsi muscles were removed from the carcass after 24 h of slaughter and used to determine pork properties including carcass weight, backfat thickness, moisture, intramuscular fat, pH24h, shear force, redness, texture, and fatty acid composition. The PRKAG3, FASN, CAST, and MC4R gene SNPs were significantly associated with the meat quality traits (p<0.003). The meats of PRKAG3 (A 0.024/G 0.976) AA genotype had higher pH, redness and texture than those from PRKAG3 GG genotype. Meats of FASN (C 0.301/A 0.699) AA genotype had higher backfat thickness, texture, stearic acid, oleic acid and polyunsaturated fatty acid than FASN CC genotype. While the carcasses of CAST (A 0.373/G 0.627) AA genotype had thicker backfat, and lower shear force, palmitoleic acid and oleic acid content, they had higher stearic acid content than those from the CAST GG genotype. The MC4R (G 0.208/A 0.792) AA genotype were involved in increasing backfat thickness, carcass weight, moisture and saturated fatty acid content, and decreasing unsaturated fatty acid content in Duroc meat. These results indicated that the five SNP markers tested can be a help to select Duroc breed to improve carcass and meat quality properties in crossbred pigs.
INTRODUCTION
Duroc boar is used as a terminal sire at commercial pig production. This breed has an excellent growth rate and higher intramuscular fat content than other breeds (Suzuki et al., 2003). Pig breeders are at the top of the pyramid structure of swine industry and play a vital role in the introgression of superior genes to the commercial pigs. Thus, the ability of breeding stock related to economic traits is the most important factor that influences the success or failure of swine industry (Ibáñez-Escriche et al., 2014).
Due to the advances in genetic technology, new livestock sectors such as breeder management, traceability systems, transgenic animal technology, and livestock disease control, which helped improving the meat quality, have been evolved (Dalvit et al., 2007; Jang et al., 2012). Over the past several decades, breeding strategies have been directed to improve carcass traits, meat quality traits, growth rate, and reproductive performance of commercial animals. Of the pork properties, meat quality traits such as color, tenderness, marbling, and juiciness mostly influence the purchase decision of consumers in the market. So, extensive researches were conducted to improve meat quality. Furthermore, many researchers have studied to identify the genes associated with various economic traits in livestock (Mignon-Grasteau et al., 2005).
The protein kinase adenosine monophosphate-activated γ3 subunit (PRKAG3), an RN gene, has a significant impact on processed yield and meat quality as the gene regulates glycogen content in muscle (Milan et al., 2000). Ciobanu et al. (2001) found the PRKAG3 allele in several pig breeds, including Landrace, Large White, Berkshire, Duroc, and Duroc synthetics.
The fatty acid synthase (FASN) gene is located on pig chromosome 12, and has significant effects on the fatty acid composition of pork (Clop et al., 2003). The pig FASN gene which is associated with fatty acid composition of backfat is located in the quantitative trait locus (QTL) region 0 to 40 cM, and is reported to affect the gadoleic acid (C20:1, a mono-unsaturated fatty acid) content in backfat (Muñoz et al., 2007).
The calpastatin (CAST) which has been found in all the tissues that contain calpains, is a specific inhibitor of calpain enzymes (Lonergan et al., 2010). Ciobanu et al. (2004) found that two associated CAST genes (Arg249Lys and Ser638Arg) located on the swine chromosome 2 region were related to the tenderness of longissimus dorsi (LD) muscle in pigs. The endogenous calpain system plays a major role in postmortem muscle proteolysis and meat tenderization processes (Lonergan et al., 2010). The high mobility group AT-hook 1 (HMGA1) gene which is related to the obesity of humans has been mapped at the peak of the fat deposition QTL on pig chromosome 7 (Kim et al., 2004). The HMGA1 polymorphisms are consistently associated with fat deposition traits across several pig populations, except for a pure Duroc in which the distribution of a specific HMGA1 genotypic class was relatively small (Kim et al., 2004).
Growth performance and carcass characteristics are very important in the pig industry, and the melanocortin-4 receptor (MC4R), a Rhodopsin-like G protein-coupled receptor, is primarily expressed in nerve systems and plays an important role in the regulation of food intake, energy balance, and body weight in mammals (Adan et al., 2006).
Therefore, the objective of this study was to determine the relationships between the five single nucleotide polymorphism (SNP) markers (PRKAG3, FASN, CAST, HMGA1, and MC4R) and carcass traits as well as the quality traits of Duroc meat.
MATERIALS AND METHODS
Animals, carcass grading, and carcass measurements
A total of 200 purebred Duroc gilts were used in this study. Duroc pigs were a part of the pig improvement network program in Korea (Korea Animal Improvement Association, KAIA), and raised in four farms from 2010 to 2011. In the each farm, 50 gilts of progenies from 2 sires and 10 dams were allotted to five pens as 10 pigs. Pigs raised on feeds were based on the Korean Feeding Standard for Swine (National Institute of Animal Science, 2012). After pigs per pen reached the market weight of 110 kg on average, they were slaughtered following the guidelines of Livestock Products Sanitary Control Act (Ministry of Food and Drug Safety in Korea, 2010), and then chilled overnight in a 4°C cold room. At 24 h postmortem, carcass traits including carcass grading, backfat thickness, and carcass length were measured by the personnel from the Animal Products Grading Services, Korea. Then, the LD muscle from the left side of carcass between 5th and 13th rib was collected and analyzed in the respective laboratories for meat quality and genomic DNA information at Chungbuk National University, Korea.
Genomic DNA analysis
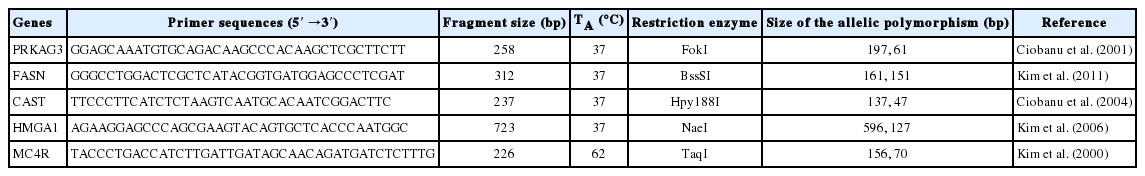
Genomic DNA was extracted from 300 g of tissue samples according to a standard protocol (DNeasy Blood & Tissue Kit, Qiagen, Hilden, Germany). A total of five candidate gene (PRKAG3, FASN, CAST, HMGA1, and MC4R) polymorphisms were previously reported and detailed information about these SNPs and their respective polymerase chain reaction (PCR)-restriction fragment length polymorphism (RFLP) genotyping approaches is illustrated in Table 1. The PCRs were performed in a 10-μL PCR buffer (10 mM Tris-HCl, 50 mM KCl, and 0.1% TritonX-100) containing 12 ng of genomic DNA, 1.5 mM MgCl2, 10 pmol of each primer, 200 μM of each dNTP, and 2.5 units of Taq DNA polymerase (Solgent, Daejeon, Korea). The thermocycling reaction was performed in a PTC-200 thermocycler (MJ Research, Watertown, MA, USA) with a 10 min initial denaturation at 95°C. A total of 40 cycles of PCR reactions were carried under the following thermocycling conditions: 95°C for 30 s, 45°C to 65°C for 30 s and 72°C for 40 s. The final extension was done at 72°C for 5 min. The result of the PCR reaction was identified by 2% agarose gel electrophoresis at 100 mV for 20 min. The information for each primer sequence, annealing temperature, and fragment size is given in Table 1. All restriction enzymes were purchased from New England BioLabs (Ipswich, MA, USA), and restriction digests were performed according to the manufacturer’s recommendations. Digested PCR products were analyzed on a 2.5% to 4% agarose gel, and each allele was scored manually. The restriction enzymes and the polymorphic fragments used for SNP genotyping are given in Table 1.
Physico-chemical analysis of carcass
pH of meat
The pH was measured in triplicate using a digital pH meter (8603, Metrohm, Herisau, Swiss). About 10 g of the sample was cut into small pieces to which 90 mL of distilled water was added, and a slurry was made using a homogenizer (T25B, IKA Sdn, Bhd., Rawang, Malaysia). The pH meter was calibrated daily with standard buffers of pH 4.0 (9863 pH buffer solution, Mettler Toledo, Schwerzenbach, Swiss) and 7.0 (9865 pH buffer solution, Mettler Toledo, Swiss) at 25°C.
Moisture and lipid
Moisture and lipid were assayed according to the AOAC methods (1995).
Shear force
Slice of LD muscle, weighing approximately 100±5 g, was placed was placed into polypropylene bag and then cooked for 40 min at 70°C in the water-bath, and subsequently cooled for 30 min. Samples were cut into 1×2×1 cm (width×length×height) pieces. The maximum weight were measured by a shearing test using a Rheo meter (Model Compac-100, SUN SCIENTIFIC Co., Tokyo, Japan) under the following operational conditions: Table speed of 110 mm/min, Graph interval of 20 msec and Load cell (max) of 10 kg using the RDS (Rheology Data System, Tokyo, Japan) Ver 2.01.
Color of meat
The Commission Internationale de l’Eclairage redness (a*) of sausage was measured using a Minolta colorimeter (CR-400, Konica Minolta, Tokyo, Japan). The instrument was standardized using a whiteplate (Y = 93.5, x = 0.3132, y = 0.3198) and D65 illuminant source before the measurements. The color variables were measured at five points on the central part of the cut surface of the slices of the samples. The thickness of samples were between 12 and 15 mm so that they did not absorb the light reflected from the bottom.
Texture
A total of five well trained in-house tasting panelist evaluated subjective characteristics of texture using a 5-point scale, score 1 through 5 (extremely undesirable to highly desirable). The sample were evaluated independently by the panelists in three different times.
Fatty acids
Total lipids of samples were extracted by using chloroform-methanol (2:1, v/v) according to the procedure of Folch et al. (1957). An aliquot of the total lipid extract was methylated as described by Morrison and Smith (1964). Fatty acid methyl esters were analyzed using a gas chromatograph (Varian 3800, Agilent Technology, Palo Alto, CA, USA) fitted with a fused silica capillary column, Omegawax 205 (30 m×0.32 mm I.D., 0.25-μm film thickness). The injection port was at 250°C and the detector was maintained at 300°C. Results were expressed as percentages based on the total peak area.
Statistical analysis
The genotypes of the five candidate genes (PRKAG3, FASN, CAST, HMGA1, and MC4R) obtained from the testing materials were classified, and their frequencies were calculated. To determine possible effects of SNP genotypes on traits, the chi-square statistic was used for significant different test among means. Genotypic effects of variations in the five candidate genes were estimated with mixed-model analysis using the PROC MIXED of SAS 9.3 software package (SAS, 2003). The model used in the statistical analysis are as follow:
where Yijklm is the observed value of phenotype traits, μ is the mean of the samples, Gi is the fixed effect of genotype, Fj is the covariate effect of farms, Dk is the covariate effect of slaughter date, Pl is the polygenic effect of Duroc lines, Am is the covariate effect of age and eijklm is the random error.
RESULTS AND DISCUSSION
Analysis of SNP marker genotypes
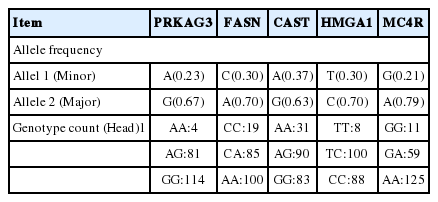
Table 2 shows genotype and allele frequency analyses of five polymorphisms in the candidate genes of the Duroc breeding stock. The genotype counts of the PRKAG3 were 4, 81, and 114 for AA, AG, GG genotypes, respectively. The frequency of the A allele was 0.224 and G allele was 0.776. In the FASN gene, the genotype counts were 19, 85, and 100 for CC, CA, AA genotypes, respectively. The frequency of the C allele was 0.301 and A allele was 0.669. All the observed counts of the genotype at the CAST gene were 31, 90, and 83 for AA, AG, GG genotypes, respectively. The frequency of the A allele was 0.373 and G allele was 0.627. The genotype frequencies of the HMGA1 gene were 8, 100, and 88 for TT, TC, CC genotypes, respectively. The frequency of the T allele was 0.296 and C allele was 0.704. Finally, the genotype counts of the MC4R gene were 125, 59, and 11 for AA, AG, GG genotypes, respectively. The frequency of the G allele was 0.208 and A allele was 0.792. Recently, many studies on gene markers associated with economic traits have been reported. Choi et al. (2012) studied a total of 81 SNP markers in the US commercial Duroc populations and found that 11 out of the 81 SNP markers were significantly associated with the carcass and meat quality traits of Duroc meat. Also, Rohrer et al. (2012) reported that PRKAG3, IGF2, CAST, and MC4R genes in commercial pigs have the greatest effects on the meat quality of pigs among the 45 candidate genes they have tested. On the other hand, according to the precedent studies, the frequency of PRKAG3 and MC4R genes from US Duroc pigs was similar with those of this study (Ciobanu et al., 2001; Davoli et al., 2012), However, we could not discover same FASN, CAST, and HMGA1 genes with this study in other study.
Relationship between five SNP genotypes and the carcass characteristics
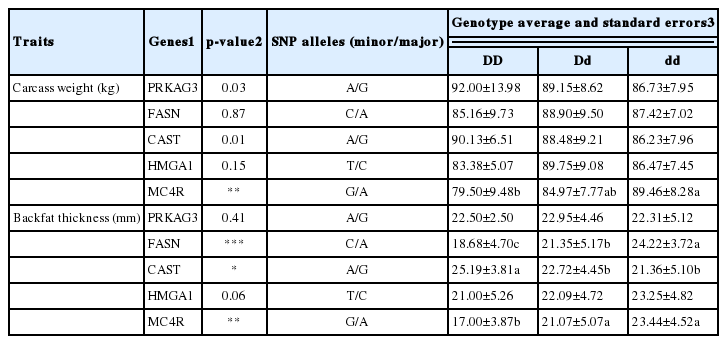
The associations of five SNP genotypes with the carcass characteristics of Duroc breeding stock are presented in Table 3. The carcass weight significantly was associated with MC4R gene (p<0.0001). The pigs with MC4R AA genotype had heavier carcass weight than those carrying AG and GG genotypes. Whereas, the PRKAG3, FASN, CAST, and HMGA1 genes did not show significant association with carcass weight. On the other hand, the pigs with AA genotype of FASN gene significantly were higher in backfat thickness than CC and AC genotype pigs. Also, the AA genotype of the CAST gene significantly increased the backfat thickness of pigs compared with AG and GG genotypes. The pigs with GG genotype of MC4R gene were lower in the backfat thickness than GA and AA genotype pigs significantly (p<0.0001). However, there were no significant associations between the PRKAG3 and HMGA1 genes and backfat thickness.
Kim et al. (2005) reported that the MC4R NN genotypes of Duroc breed increased daily gain, feed conversion, backfat thickness and meat yield. Similar results were observed in another study using an Italian Duroc breed (Davoli et al., 2012). Previous studies have reported that the FASN gene was involved in the synthesis of fatty acids (Kim et al., 2011; Maharani et al., 2011). In this study, the AA genotype of the FSAN gene increased the backfat thickness of Duroc breed. The research of Kurył et al. (2003) and Krzęcio et al. (2008) demonstrated the effect of CAST gene on the increase or decrease of backfat thickness in various pig breeds.
Relationship between five SNP genotypes and chemical composition
The associations of five SNP genotypes with the chemical composition of LD muscle in Duroc breeding stock are described in Table 4. In the moisture content, the MC4R gene showed significant impact, whereas the PRKAG3, FASN, CAST, and HMGA1 genes were not associated with moisture content of LD muscle significantly. The GG genotype of MC4R gene increased moisture content, whereas AA genotype lowered moisture content in LD muscle of Duroc population. Of the five genes, the FASN gene only was associated with intramuscular fat of LD muscle significantly. Meats from pigs with the AA genotype of FASN gene had significantly higher intramuscular fat than the CC genotype.
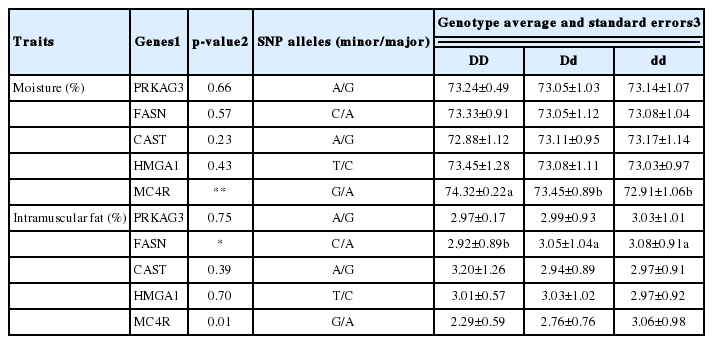
Association of SNP genotypes and their harboring genes with chemical composition of LD muscle in Duroc breeding stock
Ovilo et al. (2006) reported that the MC4R gene was not associated with moisture, protein and intramuscular fat content of LD muscle in commercial pigs. On the other hand, Kim et al. (2006a) indicated that the MC4R genotypes affected lean meat growth in Duroc pigs. The moisture content of meat had a negative correlation with intramuscular fat content (Chin et al., 2012). Thus, an increase in leanness resulted in a decrease in fat content. Kim et al. (2011) reported that intramuscular fat content was associated with the FASN gene from the native Korean pigs crossed with Yorkshire or Landlace breeds. The relationship between the FASN gene and fat content in this study agreed with the tendency of native Korean pigs.
Relationship between five SNP genotypes and meat quality
The associations of five SNP genotypes with meat quality characteristics from LD muscle of Duroc breeding stock are shown in Table 5. The PRKAG3 gene significantly influenced on the pH24h of LD muscle from Duroc pigs (p<0.003). The AA genotype of pig showed a higher pH24h than those of GG genotype pig (p<0.05). In the shear force, the CAST gene had significant effect on the shear force of LD muscle of Duroc population. The pigs with AA genotype of the CAST gene had significantly lower shear force value than those with GG genotype pigs. The PRKAG3 gene also influenced on the redness and texture of LD muscle from Duroc pigs. The AA genotype of PRKAG3 gene significantly increased the redness of LD muscle compared with AG and GG genotypes, as well as the LD muscle containing the AA genotype of PRKAG3 gene showed more desirable texture score than GG genotype LD muscle (p<0.003). In addition, the FASN gene also affected the texture score of LD muscle significantly, and the texture score of muscle containing A allele was significantly higher than those of CC genotype.
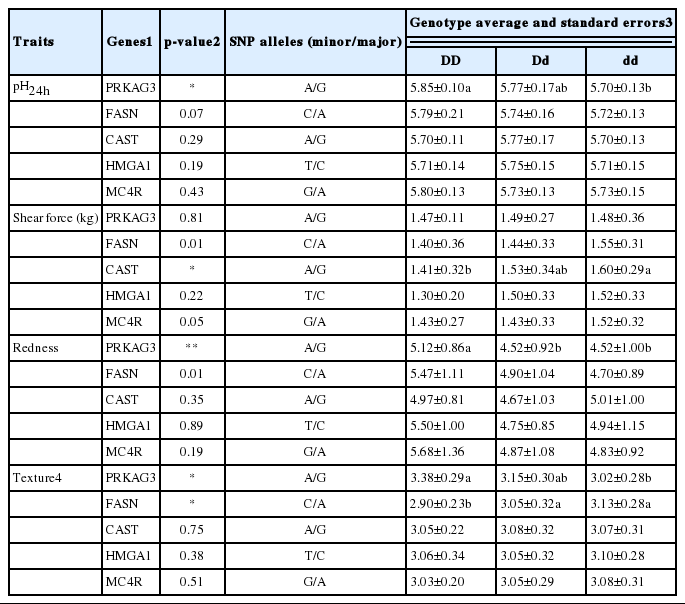
Association of SNP genotypes and their harboring genes with meat quality traits of LD muscle in Duroc breeding stock
The associations between meat quality and the PRKAG3 gene that regulates the glycogen content of intramuscular tissue are well established (Ciobanu et al., 2001). Furthermore, meat quality was influenced considerably by PRKAG3 genotypes. In post-mortem meat, the intramuscular glycogen content determines the pH of the meat through the glycolysis process. The ultimate pH of a meat is an important indicator to evaluate meat quality (Park et al., 2002). Ciobanu et al. (2004) reported that CAST genotypes significantly affected the shear force, cooking loss, and juiciness values of pork from Berkshire× Yorkshire crossbred. However, in the present study, the CAST gene showed a significant association only with the shear force value of Duroc LD muscles. Significant association between PRKAG3 gene and redness was also revealed in the LD muscle of Duroc population. The meats from pigs with AA genotype showed a higher redness value than those with AG and GG genotypes. However, no significant associations between the meat quality parameters and rest of the genes tested were found.
The color of pork is largely determined by the pH of meat. Since the pH near the isoelectric point of muscle is caused by the accumulation of lactic acid, the moisture content of muscle is not preserved due to the weak ionic strength of myofibril (Park et al., 2002). Meat texture was closely associated with the PRKAG3 and FASN genes. The meat from pigs with AA genotype of the PRKAG3 gene showed a higher texture score than the AG and GG genotypes. The AA genotype of the FASN gene earned a higher score compared to that of the AC and CC genotypes. Texture score is evaluated by the firmness and springiness of meat surface. The PRKAG3 gene is highly correlated with the pH of meat (Milan et al., 2000; Ciobanu et al., 2001). The pH of meat is an indicator for determining normal, dark firm dry (DFD) or pale soft exudative (PSE) meat. Therefore, the PRKAG3 gene is significantly associated with the texture of meat. Furthermore, the FASN gene is known to be involved in the saturated/unsaturated ratio of fatty acids (Clop et al., 2003; Muñoz et al., 2007). Due to the differences in melting points of saturated and unsaturated fatty acids and the chain length of fatty acids, the fatty acid composition affects the texture of meat (Wood et al., 2004).
Relationship between five SNP genotypes and fatty acids
The associations of five SNP genotypes with fatty acids of Duroc breeding stock population are reported in Table 6. The FASN, CAST, and MC4R genes showed significant effects on the fatty acids of LD muscle of Duroc population. The FASN and CAST genes had significant effects in increasing or decreasing palmitoleic acid (C16:1n7), stearic acid (C18:0) and oleic acid (C18:1n9) content of meat. Furthermore, the meats from pigs that carry AA genotype of the FASN gene showed significantly lower arachidonic acid (C20:4n6), but higher linoleic acid (C18:2n6), linolenic acid (C18:3n3), and poly-unsaturated fatty acid content than CC genotype. The meats from pigs that carry AA genotype of the CAST gene showed significantly lower content of mono-unsaturated fatty acids than the AG and GG genotypes. Cameron and Enser (1991) reported that flavor quality traits were improved as the concentration of mono-unsaturated fatty acids increased and poly-unsaturated fatty acids decreased. Further, De Smet et al. (2004) indicated that the contents of saturated and mono-unsaturated fatty acids increased faster than those of poly-unsaturated fatty acids as the fatness increased. Although, the fatty acid composition of meat is influenced by genetic factors, their influence was lower than that of the dietary factors. The MC4R gene was significantly associated palmitic acid (C16:0), oleic acid (C18:1n7), linolenic acid (C18:3n6), arachidonic acid (C20:4n6), total saturated fatty acid, and total unsaturated fatty acid contents of LD muscle. Our results agreed with those of Ovilo et al. (2006) who reported that the pigs with MC4R AA genotype showed higher saturated fatty acids and lower unsaturated fatty acids content than the GG genotype pigs. Excessive unsaturated fatty acids (C18:2n6, C18:3n3, and C20:4n6) causes softening of meat and accelerates oxidative changes. Also, the concentration of saturated fatty acids is related to the hardness of meat (Wood et al., 2004; Bertol et al., 2013). However, the intake of saturated fatty acids in meat is recently becoming an issue due to increased health concerns about saturated fatty acids. So, the genes involved in fatty acid compositions of meat need further attentions.
CONCLUSION
All five SNP marker (PRKAG3, FASN, CAST, HMGA1, and MC4R) genes, except for HMGA1 were significantly associated with the meat quality traits of Duroc population. The PRKAG3 AA genotype increased pH24h, redness, and texture values of pork LD muscle. The FASN gene AA genotype increased backfat thickness, texture values, stearic acid, oleic acid, and poly-unsaturated fatty acid content, whereas the CAST AA genotype increased backfat thickness, lowered shear force, palmitoleic acid, oleic acid, and increased stearic acid content in Duroc pork. The MC4R AA genotype increased backfat thickness, carcass weight, moisture, and saturated fatty acid, and decreased unsaturated fatty acid. Therefore, the genetic information from Duroc breeding stocks can be utilized effectively by swine industry to improve pork quality, and to meet the changing consumer demands. The grade evaluation standard of pork in Korea was revised in 2013 to enhance the meat quality by reducing the importance of backfat thickness and carcass weight. For quality grade, grading items for fat deposition, meat color, meat texture, fat color, and fat texture have also been changed (Guidelines of Livestock Products Sanitary Control Act, Ministry of Food and Drug Safety in Korea, 2014). Obviously, this study of genomic information related to carcass characteristics from each pig will be helpful in controlling the backfat thickness and carcass weight of pigs. Thus, the genotypes of gene can be used to determine the phenotypes of each individual pig, and this information can be applied to the breeding industry to improve pork quality in commercial pigs.
ACKNOWLEDGMENTS
This research was supported by Korea Institute of Planning and Evaluation for Technology in Food, Agriculture, Forestry and Fisheries (IPET) through Agri-Bioindustry Technology Development Program funded by Ministry of Agriculture, Food and Rural Affairs (114073-3). This research was also supported by the Priority Research Centers Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (2009-0093813).
Notes
CONFLICT OF INTEREST
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.