Effect of KiFAY on Performance, Insulin-like Growth Factor-1, and Thyroid Hormones in Broilers
Article information
Abstract
A comparative study was performed to investigate the efficacy of KiFAY as a feed additive on performance parameters, thyroid, and pancreatic hormone levels in broilers. Ninety birds (Vencobb 400) were randomly divided into three groups viz., Control (no DL-methionine supplementation), Treatment1 (containing added DL-methionine) and Treatment 2 (containing KiFAY and without DL-methionine supplementation). The performance parameters (weekly body weight, body weight gain, feed intake, and feed consumption ratio) were recorded and calculated during the whole study of 4 weeks. Analyses of insulin and insulin-like growth factor (IGF 1), triiodothyronine (T3), thyroxine (T4) and thyroid stimulating hormone (TSH) were performed at the end of the study. The results show that birds on supplementation of KiFAY performed significantly (p<0.001) better than other treatments. The weekly body weight, body weight gain, feed in-take and feed consumption ratio improved in KiFAY treated birds. The study found an increase in insulin and IGF1 levels (p<0.001) in KiFAY compared with the other treatments. Serum T3, T4, and TSH levels in the Treatment 2 were higher than other treatments (p<0.001). The KiFAY supplementation was able to improve performance with associated responses at a hormonal level in broilers.
INTRODUCTION
Amino acid supplementation has been the backbone of poultry nutrition. This well-known fact is demonstrated with methionine being the first limiting amino acid for broilers raised on a corn-soy based diet (Burley, 2012). The utilization of synthetic methionine and improving its bioavailability in feed has played a key role in constantly improving poultry performance. There are two main synthetic methionine varieties produced chemically to be used in poultry viz., DL-methionine powder form (DLM, 99%), and 2-hydroxy-4-(methylthio) butyric acid liquid form (HMB, 88%). Recently a third variety derived from biomass has been introduced. The relative bio-efficacy of DLM and HMB differs as shown in laying hens. A study by Danner and Bessei (2002) demonstrated the efficacy of HMB relative to DLM, which found to be 69% (feed conversion ratio, FCR) and 67% (daily egg mass) lower than DLM. Another study by Meirelles et al. (2003) compared DL-methionine hydroxy analog-free acid (MHA-FA) to DLM supplementation and demonstrated the latter to be a better performing feed additive for methionine supplementation. Recently, a direct relation in the relative bioavailability of MHA-FA, a poly herbal ingredient (PHI) with DLM was proposed as a feed additive The PHI claims to contain a blend of dipeptides and oligopeptides of methionine as well as certain intermediates of the same amino acid. The study states a 58% and 4.5% relative bioavailability for MHA-FA and PHI to that of DLM on a product basis respectively (Sangali et al., 2014).
Poultry nutritionists formulate dietary levels as per ileal amino acid digestibility of poultry feed ingredients. The digestibility rates among ingredients show intra-ingredient interactions to alter digestibility of the complete feed formulation. Digestibility of protein relates to particle size and digestive rates of ingredients (Pacheho et al., 2013). Slowly digestible starches (SDS) have positive effects on feed conversion rates in low protein diets (Weurding et al., 2003). Another study suggests that the interaction between protein (glutamine and casein) and SDS benefits FCR in broilers due to protein-sparing effects (Enting et al., 2005) and therefore enhances the growth efficiency of broiler chickens. It also suggests that starch digestion rate might alter the metabolic responses of insulin. The addition of synthetic amino acids at high levels also stimulates insulin secretion from pancreas (Murray et al., 1998).
The effects of amino acid availability on the avian endocrine system have been least studied. Reports indicate that plasma level of triiodothyronine (T3) were elevated in broilers with methionine, arginine, lysine and isoleucine deficient dietary levels compared to pair-fed controls (50/50 mixture of broiler feed and purified amino acid diet)(Carew et al., 1997). Later Carew et al. (2003) further reported Met deficiency alters the normal thyroid hormone metabolism along with depressed levels of insulin-like growth factor 2 (IGF-2), but the effect is dependent on the degree of deficiency. In the same experiment, IGF-1 did not show any variation even in a Met deficient diet. However, decrease in plasma IGF-1 levels were exhibited in the broilers with a low protein diet, which was brought to normal by feeding dietary protein (Rosebrough and McMurtry, 1993).
Onion (Allium cepa) and Garlic (Allium sativum) belong to the family Liliaceae (Ebesunun et al., 2007). These phytogenic derivatives show the presence of bioactive component, dialkylpolysulfides, which is a sulfur-containing organic compound. A study led by Aji et al. (2011) used onion and garlic to improve the productive performance of broiler chickens. Garlic and onion consumption also have shown insulin like effect on the body (Keusgen, 2002). Arginine also shows properties of being a metabolic regulator, stimulating protein synthesis and reducing protein stress induced catabolism by stimulating the secretion of insulin, growth hormone and glucagon (Frank et al., 2007).
KiFAY is a tan colored amorphous powder, composed of phytoconstituent fractions obtained from onion, garlic in diatomaceous earth blended with algal cell wall components. It claims to be a natural amino acid optimizer that improves utilization of methionine in the body. Thus, in this study, we investigated the effect of KiFAY in diets deficient in methionine in contrast to the DLM supplemented diet. The comparison is based on commercial productive performance parameters and hormonal correlations.
MATERIALS AND METHODS
Birds and management
The experimental protocol was verified as per Institutional Animal Ethics Committee (IAEC) guidelines. The birds were raised on a deep litter system, using rice husk as bedding. The chicks were housed in environmentally controlled sheds with a stocking density of one bird per square foot approximated for the bird size at the end of 4 weeks. The Feed in a mash form and water were provided ad libitum. The basal feed diet was formulated as per National Research Council (1994) recommendations. The ingredients and chemical compositions of the diets were analyzed and are shown in Table 1. Brooding was carried out for the first 5 days providing 1 watt per bird. The temperature and humidity in the house was maintained in the range of 25°C to 28°C at 70% humidity. The light and dark period was maintained with 23 h light:1 h dark program.
Experimental design
Ninety day-old broiler chicks (Vencobb 400) were divided in three equal groups with equal number of male and females as confirmed by wing sexing. The birds were individually wing banded to determine the flock uniformity during performance analysis. The treatments comprised three replicates of ten birds each. The Treatments composed of control (C), basal Diet deficient in methionine; Treatment 1 (T1), basal Diet with DL-methionine (starter phase-2.7 g/kg, grower phase-2.35 g/kg, and finisher phase-2.06 g/kg) supplementation and Treatment 2 (T2), basal Diet with KiFAY (starter phase-2.7 g/kg, grower-2.35 g/kg, and finisher-2.06 g/kg) supplementation. At the end of each week, body weight gain and feed intake were recorded. Weekly FCR was calculated and Mortality was recorded on a daily basis.
Blood collection from sample space
At 28 d, two birds from each replicate were sacrificed by a cervical bleeding method. Blood was collected in a Vacuette tube for hormone estimation.
Estimation of T3, T4, thyroid stimulating hormone, insulin and insulin-like growth factor 1
After the collection of whole blood, the blood was allowed to clot for 1 h. The tubes were centrifuged at 1,200 g to separate the clot from serum, for 20 minutes at 4°C. The serum was collected in a fresh tube and sent for testing at Suburban diagnostics laboratory, Satara, Pune, 411009. The IGF 1 was estimated using a sandwich enzyme-linked immunosorbent assay kit (Elabscience, Wuhan, Hubei, China) as per the manufacturer’s instructions.
Statistical analysis
Statistical analyses were performed by GraphPad Instat version 5.0 from Microsoft Windows 7. The means were compared using one-way analysis of variance with the Tukey-Kramer multiple comparisons test. Less than 0.05 p value was considered statistically significant.
RESULTS AND DISCUSSION
The use of KiFAY in the current study focuses on the evaluation of performance and hormones used as a markers of methionine accretion in broilers. Table 2 summarizes the effect of different dietary supplementation on performance parameters. In the present study, we were able to achieve 10.06% better final weight with 7% better FCR at 28 days. The weight of the chicks on day zero differed non-significantly (p>0.05) among treatments. During the whole study, the feed intake differed significantly every week in each treatment (p<0.001). The highest feed intake was observed with control (p<0.001), whereas, the highest body weight gain was recorded with T2 (p<0.001). The FCR was found to be significantly better in T2 than the other treatments (p<0.001). The feed consumption increased in control and T2. The increase in feed intake with consistent FCR in T2 could be due to the positive effect on palatability attributed to the presence of garlic extracts in the composition of KiFAY (Abouelfetouh and Moussa, 2012). The increase in feed intake was also suspected due to deficiency of vital nutrients in feed. The study led by Berhe and Gous (2008) reveals that broilers treated with low protein feed tend to have an increase feed intake in Cobb strain but decrease in Ross strain. On the other hand, a methionine deficient diet specifically resulted in reduced feed consumption in Hubbard strain (Carew et al., 2003). However, broadening of the FCR due to reduced methionine supplementation evident in the control group of the present study was not seen in KiFAY treated birds. Mortality during whole study was within permissible limit (less than 1%) in all groups.
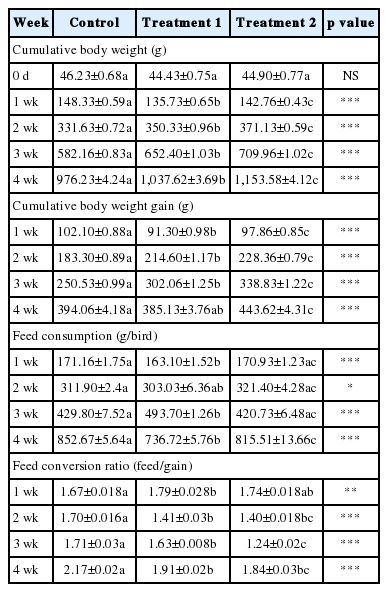
Effect of dietary treatments on cumulative body weight, cumulative body weight gain, feed consumption and feed conversion ratio in broiler chickens
Thyroid hormones play important role in growth regulation in chickens as demonstrated by thyroidectomy, which results in reduced growth rate (King and King, 1973). Thyroid hormone analysis revealed a comparative increase in plasma levels of T3, T4 and thyroid stimulating hormone (TSH) in Treatment 2 as shown in Figure 1 and Table 3. Serum thyroid hormones (T3 and T4) in control and treatment1 did not significantly differ (p>0.05), however, supplementation of KiFAY in Treatment 2 improved thyroid hormones levels than other treatments (p<0.001). The T3 levels were increased in T2 (p<0.001) (Figure 1a). These results correlated with findings of Saki et al. (2011) who reported a decreased level T3 in methionine deficient layers. However, these results contradict with that of Buchansak et al. (2006) and Carew et al. (2003) who explained the increased T3 levels results due to methionine deficient feed diet. This shows that thyroid hormone activity can be potential indicator to the metabolic activity in a commercial poultry system (Melesse et al., 2011). Moravej et al. (2006) who demonstrated increased T4 levels in high protein fed Lohmann strain broilers with improved body weight gain support the data. Thus, increase in serum T3 and T4 levels on KiFAY supplementation can be explained by a preliminary increase in TSH levels, which resulted in improving the metabolic activity in broilers. In the present study, there was a significant increase (p<0.001) in serum TSH levels in broilers supplemented with KiFAY as compared to other treatments (Figure 1c). This alteration could be attributed to the increase in serum insulin level where the thyroid axes connects the somatotropic axes.
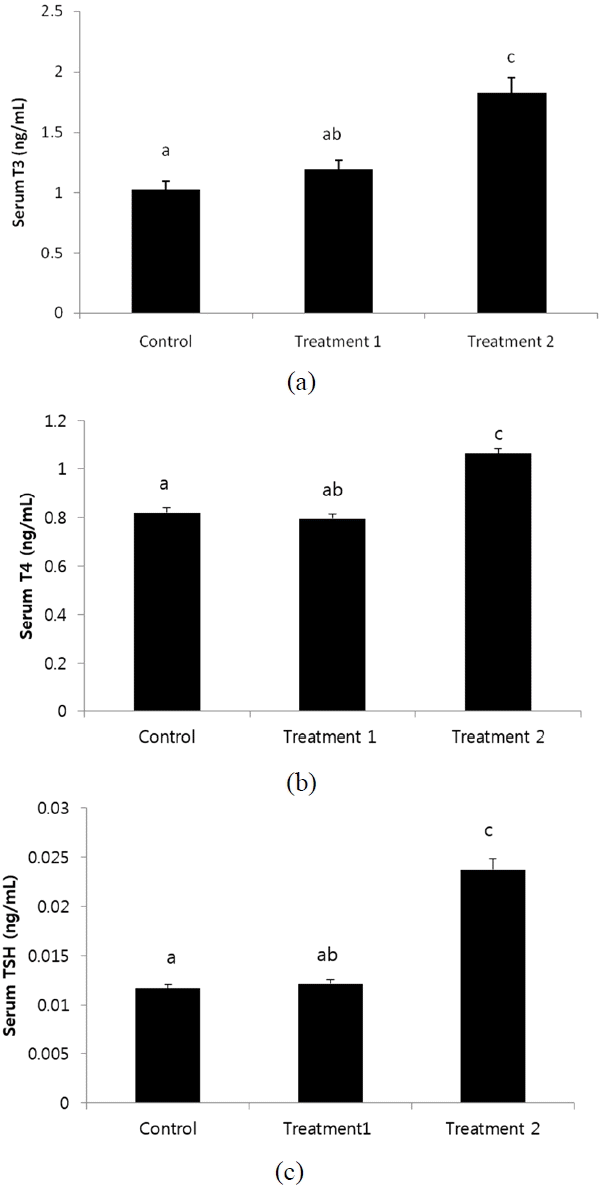
Effect of dietary treatments on (a) T3, (b) T4, and (c) thyroid stimulating hormone (TSH) levels of broilers at 28 days of age. Error bars indicate standard error mean of each treatment. Bars headed by a different letter are significantly different from all others (p<0.05).
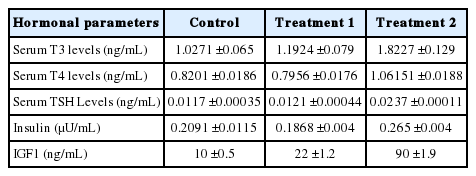
Effect of dietary treatments on thyroid hormones (T3, T4, and TSH) and pancreatic hormones (insulin and IGF 1) in broilers
Serum insulin and IGF1 level in birds supplemented with KiFAY significantly differ than other treatments (p<0.001) (Figure 2). Insulin and IGF-1 is directly related to the protein turnover rate in broilers. McGuinness and Cogburn (1990) report that IGF1 level peak in initial three weeks and T3 level in the fourth week with significant impact on broiler growth performance. This reveals a direct relation between T3 and IGF1 in broilers. This hormonal increase also overlaps with the period for which the maximum growth occurs in broilers and requires maximum protein accretion. The IGF-1 levels have a direct positive correlation with absolute weight gain and carcass cuts in broilers (Zhai et al., 2016). The improved muscle protein fractional synthesis rate was also shown to be maximum between 2 to 4 weeks of age for fast growing birds and decreased as age progressed as described by Tesseraud et al. (2000). Gonzales et al. (1999) attributed same trend with IGF-1 levels in broilers and showed them independent from the effect of thyroid axes. However, goitrogen administration had reduced the growth rate in chicken (Rosebrough and McMurtry, 2003) and anti-thyroid axes administration resulted in decreased circulating IGF-I levels and hepatic IGF-I mRNA restorable by T4 administration (Tsukada et al., 1998). This explains an indirect relationship between both the axes. The increase in the serum levels of insulin in broilers supplemented with KiFAY is in unison with the studies of Bnouham et al. (2006) where the presence of active compound of garlic such as Allicin and S-allylcysteinsulfoxide have shown to increase hepatic metabolism by increased insulin release from pancreatic beta cells of rats. The increased TSH levels could be a direct effect of insulin secretion affecting thyroid axes for metabolic impetus. The increase in IGF-1 could not be explained at this moment and deserves further study.
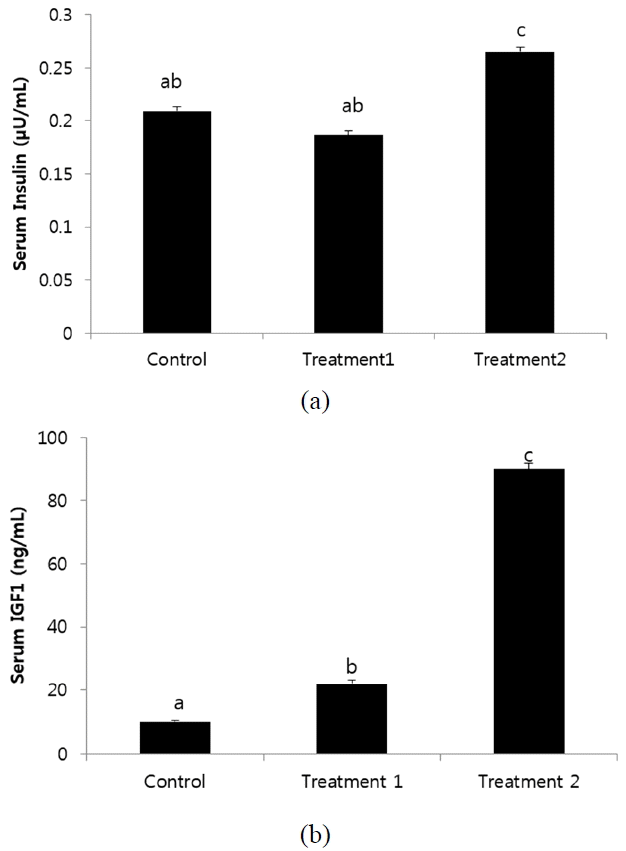
Effect of dietary treatments on (a) Insulin and (b) insulin-like growth factor 1 (IGF1) levels of broilers at 28 days of age. Error bars indicate standard error mean of each treatment. Bars headed by a different letter are significantly different from all others (p<0.05).
In conclusion, supplementation of KiFAY in feed optimized bird performance even when the feed diet was deficient in DL-methionine levels. This can be attributed to the improved protein accretion, which could be confirmed by employing protein turnover studies in the future.
ACKNOWLEDGMENTS
This work was funded by Vinayak Ingredients (India) Private Limited, India.
Notes
CONFLICT OF INTEREST
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
