 |
 |
| Anim Biosci > Volume 29(7); 2016 > Article |
|
Abstract
Milk composition is an imperative aspect which influences the quality of dairy products. The objective of study was to compare the chemical composition, nitrogen fractions and amino acids profile of milk from buffalo, cow, sheep, goat, and camel. Sheep milk was found to be highest in fat (6.82%±0.04%), solid-not-fat (11.24%±0.02%), total solids (18.05%±0.05%), protein (5.15%±0.06%) and casein (3.87%±0.04%) contents followed by buffalo milk. Maximum whey proteins were observed in camel milk (0.80%±0.03%), buffalo (0.68%±0.02%) and sheep (0.66%±0.02%) milk. The non-protein-nitrogen contents varied from 0.33% to 0.62% among different milk species. The highest r-values were recorded for correlations between crude protein and casein in buffalo (r = 0.82), cow (r = 0.88), sheep (r = 0.86) and goat milk (r = 0.98). The caseins and whey proteins were also positively correlated with true proteins in all milk species. A favorable balance of branched-chain amino acids; leucine, isoleucine, and valine were found both in casein and whey proteins. Leucine content was highest in cow (108±2.3 mg/g), camel (96±2.2 mg/g) and buffalo (90±2.4 mg/g) milk caseins. Maximum concentrations of isoleucine, phenylalanine, and histidine were noticed in goat milk caseins. Glutamic acid and proline were dominant among non-essential amino acids. Conclusively, current exploration is important for milk processors to design nutritious and consistent quality end products.
Globally, consumers pay great attention to food and its composition due to a pivotal relationship between diet and human health. Milk is a complex mixture of fat, proteins, carbohydrates, minerals, vitamins and other miscellaneous constituents dispersed in water (Ahmad et al., 2008; Ozrenk and Inci, 2008). The quality of milk products is reliant on milk composition that varies with stage of lactation, milking methods, environment, season, diet, feeding system, breed and species (Kittivachra et al., 2007). However, the composition of milk fluctuates markedly among different species (Pavic et al., 2002; Ahmad et al., 2008). Caseins and whey proteins are the main group of milk proteins found in different ratios in various milk species. Human milk has casein to whey protein ratio of 40:60, quine milk has ratio of 50:50, while cow, sheep, goat and buffalo milk has casein to whey protein of 80:20 (Fox et al., 2000).
Among milk components, proteins are most important constituents of human diet contributing significant nutritional, biological and functional properties. The amino acids profile of caseins and whey proteins occupy a unique position in human nutrition. These proteins are ranked as quality proteins with highest biological value, good digestibility (97% to 98%), rapid absorption and utilization in the body. Specifically, casein is an incredibly efficient nutrient supply owing to its providing a sustained and slow release of amino acids into blood stream (Schaafsma, 2000). The highest concentration of branched chain amino acids (BCAAs) present in milk proteins are important for maintenance of tissue growth, repair and prevention of catabolic actions during exercise. Likewise, the amino acid cysteine enhances glutathione levels, exhibits strong antioxidant properties and assists the body in combating various diseases (Ha and Zemel, 2003). In addition, milk proteins are recognized for their applicability in sports nutrition, baked goods, salad dressings, emulsifiers, infant formulas, and medical nutritional formulas.
Buffalo milk contributes 62.04% of total milk produced in Pakistan while the share of cow, goat, sheep, and camel milk is 34.56%, 1.65%, 0.08%, and 1.81%, respectively (GOP, 2014–15). Milk plays a vital role in building a healthy society and can be used as vehicle for rural development. The importance of milk from non-bovine animals is increasing globally, due to quantity (15% of global milk production) and economical, cultural, and ecological factors. Special nutritional characteristics have been claimed for various types of non-bovine milk and milk products (Al Haj and Al Kanhal, 2010). These underutilized resources are of great significance to milk producers, processors, and consumers for designing the innovative products with versatility, taste and functionality. Hence, valorization of non-bovine milk and milk products requires intensive research, particularly in the area of proteins, peptides and amino acids. Therefore, present research work was carried out on milk composition, protein characterization and amino acid profile of casein and whey proteins with special reference to local milk species found in Pakistan. Purposely, the buffalo (Nili-Ravi), cow (Sahiwal), sheep (Kajli), goat (Beetal), and camel (dromedary) lactating breeds were selected for the study.
Thirty fresh samples of buffalo, cow, sheep, goat, and camel milk (six samples of each species) were collected in sterile glass bottles from the Dairy Farm, Department of Livestock Management, University of Agriculture, Faisalabad, Pakistan. These samples were labeled, ice packed and transported to laboratory. All milk samples were then placed in the refrigerator at 4°C for further analysis. Physicochemical analyses were performed (in triplicate) within 24 hours in the Dairy Technology Laboratory, National Institute of Food Science and Technology, University of Agriculture Faisalabad. Freeze-dried samples of caseins and whey proteins were also subjected to estimate their amino acid profile.
The pH of milk samples was measured using portable pH meter (Hanna, HI-99161). The acidity (%) in sample was estimated by titration method (947.05) given in AOAC (2005). Fat of milk was determined by following Gerber method as described by Marshall (1993). Solid-not-fat (SNF) contents were calculated according to David (1977) using Lactometer. Total solids (TS) were determined according to the method described in AOAC- 925.23 (2005). The ash content was estimated by incineration of samples in muffle furnace at 550°C for 6 hours, as given in AOAC, No. 945.46 (2005).
The crude protein (CP), true protein (TP), casein, non-casein-nitrogen (NCN), whey proteins and non-protein-nitrogen (NPN) contents were determined by using Kjeldahl method according to standard protocol of IDF (1993). After calculating the total amount of nitrogen (%), it was multiplied with a factor 6.38 to get CP. The TP in the milk sample were determined by treating with 12% trichloroacetic acid. The nitrogen (%) was converted to NPN and NCN contents by using the conversion factor 3.60 and 6.25 respectively. Protein (nitrogen) fractions were calculated as:
For the separation of caseins and whey proteins, milk samples were defatted by centrifugation at 5,000 g for 15 min at 4°C. The skim milk, heated to 37°C, was separated into whole casein and whey proteins by isoelectric precipitation at pH 4.6 with 1 N HCl. After centrifugation at 5,000 g for 15 min at 30°C, the supernatant (whey proteins), was collected and dialyzed at 4°C against several changes of distilled water while the precipitated caseins were washed with acidified distilled water (pH 4.6). Both fractions were then freeze-dried for further analysis.
The freeze dried samples of casein and whey protein were then subjected to determination of amino acids composition using an amino acid analyzer according to the method described by Schuster (1988). The freeze-dried samples of casein and whey proteins were hydrolyzed with 6 N HCl under vacuum at 110°C for 24 hours. The hydrolysates were dried in a rotary evaporator at 40°C under vacuum to remove the excess acid (6 N HCl). The dry residues were then dissolved in a known quantity of citrate buffer (pH 2.2) and filtered (0.2 μm) to obtain a clean solution of the hydrolysate. An aliquot of hydrolysate was injected into the column (Shim-pack ISC-07/S1504 Na) of the higher performance liquid chromatography based amino acid analyzer (RF-10AXL, Shimadzu Corporation, Tokyo, Japan) equipped with fluorescence detector (FLD-6A). Sodium hypochlorite and o-phthalaldehyde solutions were used as reaction solutions.
Statistical analysis of experimental data was performed by applying completely randomized design at 5% level of significance while significant differences between means were compared using Duncan’s multiple range test (Steel et al., 1997). The relationship between nitrogen fractions was calculated by simple linear correlations.
The chemical composition of milk samples indicated that fat was the most inconsistent component whereas the ash contents showed minimum variations among different milk species (Table 1). It was observed that sheep milk has maximum fat (6.82%±0.04%), SNF (11.24%±0.02%), TS (18.05%±0.05%) and ash (0.85%±0.01%) contents followed by buffalo milk (6.58%±0.02%), (10.09%±0.03%), (16.67±0.03%) and (0.82%±0.02%) respectively. Previous studies depicted that Murrah and Nili-Ravi breeds of buffalo milk had 6.57% and 6.53% fat contents respectively (Han et al., 2007). The results regarding TS contents of buffalo, cow, goat and camel milk are also comparable to previously reported results of Han et al. (2007) and Mal et al. (2007). Several factors such as breed and health of animal, stage of lactation, feeding systems, seasonal changes, milking frequency and milking systems, nutrition and genetics can cause variation in relative proportion of milk constituents. Non-significant differences were observed for acidity of milk samples from all species, however, there was some variation in pH values. Milk pH is the most critical factor for manufacturing of various dairy products. It determines the conformation of proteins, the activity of enzymes and dissociation of acids present in milk. The lower pH of fresh milk may be due to bacterial action and higher one indicates the udder infection or mastitis (Uallah et al., 2005). Many previous findings regarding pH and acidity of buffalo milk (Han et al., 2007; Ahmad et al., 2008; Imran et al., 2008), cow milk (Ozrenk and Inci, 2008) and camel milk (Khaskheli et al., 2005; Mal et al., 2007) are in agreement with the findings of current investigation.
Protein is an important constituent of milk which contains about 95% of the total nitrogen present. In the current exploration, protein fractions like CP, TP, caseins and whey proteins, NCN and NPN contents showed significant differences (p<0.05) among different milk species. The CP (5.15%±0.06%), TP (4.53%±0.03%), caseins (3.87%±0.04%) and NPN (0.62%±0.02%) contents were relatively higher in sheep milk followed by buffalo and cow milk (Table 2). The highest whey proteins were in the camel (0.80%±0.03%) milk whereas cow milk represented lowest (0.47%±0.01%). Similarly, the casein contents (2.11%±0.02%) were also found lowest in camel milk as compared to other species. Regarding the NPN fraction, no significant variation was noticed between the buffalo and goat milk. The highest r-values (Table 3) were recorded for the correlations between CP and casein in buffalo (r = 0.82), cow (r = 0.88), sheep (r = 0.86) and goat milk (r = 0.98). The CP also showed positive relationship with whey protein in sheep (r = 0.80) and goat (r = 0.98) milk and NCN contents in camel milk (r = 0.84). Surprisingly, CP was negatively correlated with NPN in buffalo milk (−0.75) while positively correlated with cow milk (r = 0.84) and camel milk (r = 0.85). Different factors such as genetics, species/breed, lactation stages, type of diet, udder health and seasonal variations have pronounced influence on protein contents of milk (Pavic et al., 2002). The caseins and whey proteins were positively correlated with the TP in all milk species but higher results were observed for whey protein in cow (r = 0.83) and camel milk (r = 0.75).
Several findings concerning the protein content of buffalo (Han et al., 2007), cow (Ozrenk et al., 2008), goat (Strzalkowska et al., 2009), and camel milk proteins (Shamsia, 2009) have shown harmony with present research work. Similarly, the TP contents of buffalo, cow, sheep and goat milks are in line with the investigations of Pirsi et al. (2000). The findings of previous studies are comparable with the results of current exploration concerning the casein contents of sheep, goat milk (Borkova and Snaselova, 2005), cow milk (Imran et al., 2008) and camel milk (Khaskheli et al., 2005; Shamsia, 2009). Proteins are an important factor affecting the quality of dairy products as the reduction in proteins and casein (α- and β-casein) contents results in poor cheese making properties (Bernabucci et al., 2002). The findings of Borkova and Snasolva (2005) have shown that cow and goat milk contain 0.47%±0.01% and 0.53%±0.02% whey proteins, respectively.
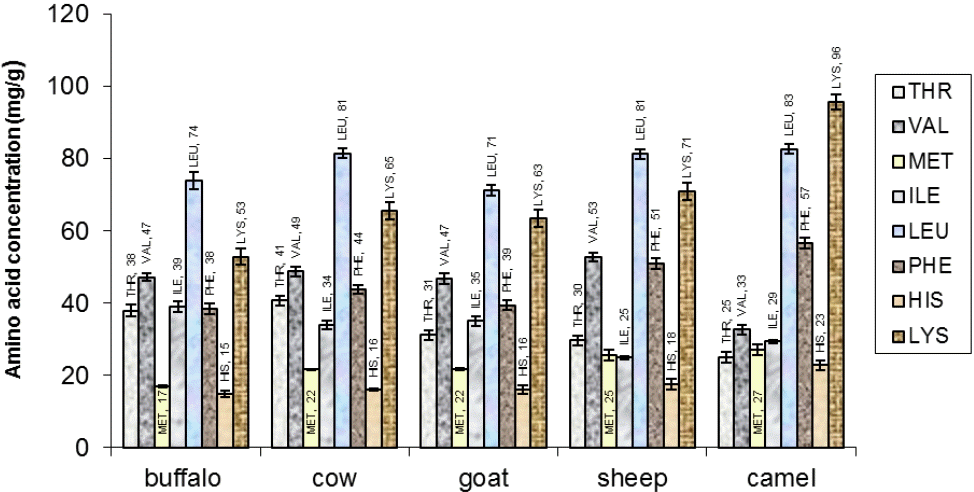
The principal milk proteins such as casein and whey proteins constitute a favorable balance of amino acids, comprised of essential and non-essential amino acids in varying concentrations. The present investigation revealed that leucine was the major amino acid in casein while lysine was second among all essential amino acids as shown in Figure 1. Leucine content was found to be highest in cow milk casein (108±2.30 mg/g) followed by camel (96±2.20 mg/g) and buffalo (90±2.40 mg/g) casein. However, minor differences were noticed in the leucine content of whey proteins from all milk species (Figure 2). Leucine plays a distinct role in protein metabolism and the translation initiation pathway of muscle protein synthesis. It is also involved in reversible phosphorylation of proteins that control mRNA binding to the 40S ribosomal subunit (Anthony et al., 2001).
In the current study, high concentration of essential amino acid lysine was found in camel milk casein (67±2.39 mg/g) and whey proteins (96±2.20 mg/g). A substantial quantity of valine, isoleucine, threonine and phenylalanine was also observed in both casein and whey proteins. Highest concentration of valine was found in cow casein (54±1.42 mg/g) and sheep whey proteins (53±1.30 mg/g). The isoleucine, phenylalanine and histidine amino acids were at a maximum in casein of goat milk. Phenylalanine concentration was higher in camel (57±1.50 mg/g), sheep (51±1.39 mg/g) and cow (44±1.25 mg/g) milk whey proteins. Similarly, Stancheva et al. (2011) reported the highest percentages for leucine (10.09%) followed by lysine (8.40%) and valine (6.73%) among the essential amino acids and the lowest content was determined for methionine in sheep milk. It was reported by Shamsia (2009) that camel milk proteins possess a high content of essential amino acids except threonine and valine. The lysine and threonine are limiting amino acids in various protein resources. These are strictly indispensable, sensitive to catabolism and important for protein synthesis. Importantly, the amino acid profile differences in dietary proteins also influence their utilization in body. Milk proteins specifically elicited a greater increase in BCAAs (26%) concentrations in peripheral tissues compared to soy protein (Bos et al., 2000; Fouillet et al., 2002). Furthermore, the BCAAs play significant role in weight control via glucose homeostasis and lipid metabolism.
Among the non-essential amino acids, the glutamic acid content was highest in both casein and whey proteins (Figure 3). However, the buffalo (367±4.50 mg/g) and goat (359±5.29 mg/g) whey proteins have maximum glutamic acid concentration as compared to casein. The goat casein (144±3.29 mg/g) and camel whey proteins (129±4.79 mg/g) represented good content of proline. Cysteine was also observed in casein and whey proteins with minute differences among all milk species. Asparagine and serine was found high in buffalo milk whey proteins as compared to other species under observation (Figure 4). Camel whey proteins indicated higher content of alanine and tyrosine. Previous investigations on amino acid profile of goat milk (Salem et al., 2009) and camel milk (Shamsia, 2009) also depicted that leucine and glutamic acid were the major amino acid in whole casein, while methionine and glycine were present in traces. Similarly, Stancheva et al. (2011) reported highest percentage of glutamic acid (19.08%) followed by proline (10.63%) and aspartic acid (7.27%). Whey protein and amino acid supplements are potential means to enhance lean body mass. Moreover, sulfur containing amino acids (methionine, cysteine) boost up the immune functions through intracellular conversion to glutathione, thereby serves as antioxidants (Hall et al., 2003). These proteins are the subject of great attention for specific dietary manipulations that aim to enhance host defenses.
It is concluded that milk composition and nitrogen characterization differ greatly among all milk species under investigation. Moreover, favorable balance of all the essential amino acids, especially, branched-chain amino acids (luecine, isoleucin, and valine) were found in both casein and whey proteins. The present investigation would be useful for the dairy processing industries to formulate nutritionally enhanced milk based functional products for vulnerable segment of population, even from the milk of non-bovine species.
Table 1
Chemical composition of different milk species
| Species | pH | Acidity (%) | Ash (%) | Fat (%) | SNF (%) | TS (%) |
|---|---|---|---|---|---|---|
| Buffalo | 6.66±0.01a | 0.12±0.01ab | 0.82±0.02ab | 6.58±0.02b | 10.09±0.03b | 16.67±0.03b |
| Cow | 6.63±0.02ab | 0.13±0.01b | 0.72±0.01d | 4.17±0.03d | 9.13±0.02c | 13.32±0.04d |
| Sheep | 6.64±0.02b | 0.14±0.01a | 0.85±0.01a | 6.82±0.04a | 11.24±0.02a | 18.05±0.05a |
| Goat | 6.49±0.05d | 0.12±0.02ab | 0.82±0.02ab | 4.61±0.02c | 8.95±0.04d | 13.56±0.03c |
| Camel | 6.54±0.04c | 0.12±0.02ab | 0.79±0.02c | 3.11±0.03e | 8.15±0.05e | 11.26±0.04e |
Table 2
Milk protein fractions of different milk species
| Species | CP (%) | TP (%) | Casein (%) | WP (%) | NCN (%) | NPN (%) |
|---|---|---|---|---|---|---|
| Buffalo | 4.25±0.07b | 3.87±0.02b | 3.20±0.03b | 0.68±0.02b | 1.05±0.02c | 0.38±0.02bc |
| Cow | 3.57±0.03c | 3.25±0.03c | 2.79±0.02c | 0.47±0.01e | 0.77±0.02e | 0.33±0.03d |
| Sheep | 5.15±0.06a | 4.53±0.03a | 3.87±0.04a | 0.66±0.02c | 1.28±0.03a | 0.62±0.02a |
| Goat | 3.35±0.02d | 2.95±0.02d | 2.44±0.03d | 0.53±0.02d | 0.94±0.01d | 0.39±0.01b |
| Camel | 3.24±0.04e | 2.89±0.02e | 2.11±0.02e | 0.80±0.03a | 1.13±0.02b | 0.36±0.02bc |
Table 3
Correlation between nitrogen fractions of milk from different species
REFERENCES
Ahmad S, Gaucher I, Rousseau F, Beaucher E, Piot M, Grongnet JF, Gaucheron F. 2008. Effects of acidification on physico-chemical characteristics of buffalo milk: A comparison with cow’s milk. Food Chem 106:11–17.

Al Haj OA, Al Kanhal HA. 2010. Compositional, technological and nutritional aspects of dromedary camel milk. Int Dairy J 20:811–821.

Anthony JC, Anthony TG, Kimball SR, Jefferson LS. 2001. Signaling pathways involved in translational control of protein synthesis in skeletal muscle by leucine. J Nutr 131:856–860.

AOAC. 2005. Official Methods of Analysis of AOAC International. 18th ednAssociation of Official Analytical Chemists; Gaithersburg, MD, USA:
Bernabucci UN, Lacetera N, Ronchi B, Nardone A. 2002. Effects of the hot season on milk protein fractions in Holstein cows. Anim Res 51:25–33.

Borkova M, Snaselova J. 2005. Possibilities of different milk detection in milk and dairy products—A review. Czech J Food Sci 23:41–50.

Bos C, Gaudichon C, Tome D. 2000. Nutritional and physiological criteria in the assessment of milk protein quality for humans. J Am Coll Nutr 19:191–205.

David P. 1976. The chemical analysis of foods. 7th ednChurchill Livingston; London, UK:
Fouillet H, Mariotti F, Gaudichon C, Bos C, Tome D. 2002. Peripheral and splanchnic metabolism of dietary nitrogen are differently affected by the protein source in humans as assessed by compartmental modeling. J Nutr 132:125–133.


Fox PF, Guinee TP, Cogan TM, McSweeney PLH. 2000. Fundamentals of Cheese Science. Aspen Publishers, Inc; Gaithersburg, MD, USA:
GOP (Government of Pakistan). 2014–15. Pakistan Economic Survey. Economic Advisor’s Wing, Finance Division; Islamabad, Pakistan:
Ha E, Zemel MB. 2003. Functional properties of whey, whey components, and essential amino acids: mechanisms underlying health benefits for active people. J Nutr Biochem 14:251–258.


Hall WL, Millward DJ, Long SJ, Morgan LM. 2003. Casein and whey exert different effects on plasma amino acid profiles, gastrointestinal hormone secretion and appetite. Br J Nutr 89:239–248.


Han BZ, Meng Y, Li M, Yang YX, Ren FZ, Zeng QK, Robert Nout MJ. 2007. A survey on the microbiological and chemical composition of buffalo milk in China. Food Control 18:742–746.

IDF (International Dairy Federation). 1993. Determination of milk nitrogen content. 4. Determination of non-protein nitrogen. International Standard FIL-IDF 20B.
Imran M, Khan H, Hassan SS, Khan R. 2008. Physicochemical characteristics of various milk samples available in Pakistan. J Zhejiang Univ Sci 9:546–551.



Khaskheli M, Arain MA, Chaudhry S, Soomro AH, Qureshi TA. 2005. Physico-chemical quality of camel milk. J Agric Soc Sci 1:164–166.
Kittivachra RR, Sanguandeekul R, Sakulbumrungsil R, Phongphanphanee P. 2007. Factors affecting lactose quantity in raw milk. Songklanakarin J Sci Technol 29:937–943.
Mal G, Suchitra SD, Sahani MS. 2007. Changes in chemical and macro-minerals content of dromedary milk during lactation. J Camel Pract Res 14:195–197.
Marshall RT. 1993. Standard methods for the examination of dairy products. 16th ednAmerican Publication Health Association (APHA); Washington, DC, USA:
Ozrenk E, Inci SS. 2008. The effect of seasonal variation on the composition of cow milk in Van Province. Pakistan J Nutr 7:161–164.

Pavic V, Antunac N, Mioc B, Ivankovic A, Havranek JL. 2002. Influence of stage of lactation on the chemical composition and physical properties of sheep milk. Czech J Anim Sci 47:80–84.
Pirsi A, Piredda G, Corona M, Pes M, Pintus S, Ledda A. 2000. Influence of somatic cell count in ewe’s milk composition, cheese yield and cheese quality. In : Proceeding of the 6th great lakes, Dairy Sheep Symposium; Guelph, Canada. p. 47–59.
Salem SA, El-Agamy EI, Salama FA, Abu-Soliman NH. 2009. Isolation, molecular and biochemical characterization of goat milk casein and its fractions. Trop Subtrop Agroecosyst 11:29–35.
Schuster R. 1988. Determination of amino acids in biological, pharmaceutical, plant and food samples by automated precolumn derivatization and high-performance liquid chromatography. J Chromatogr 431:271–284.


Shamsia SM. 2009. Nutritional and therapeutic properties of camel and human milks. Int J Genet Mol Biol 1:52–58.
Stancheva , Naydenova NN, Staikova G. 2011. Physicochemical composition, properties, and technological characteristics of sheep milk from the Bulgarian dairy synthetic population. Macedonian J Anim Sci 1:73–76.

Steel RGD, Torrie JH, Dickey DA. 1997. Principles and Procedures of Statistics. A Bio-Metrical Approach. 3rd EdMcGraw Hill Book Co. Inc.; New York, NY, USA:
Strzalkowska N, Jozwik A, Bagnicka E, Krzyzewski J, Horbanczuk K, Pyzel B, Horbanczuk JO. 2009. Chemical composition, physical traits and fatty acid profile of goat milk as related to the stage of lactation. Anim Sci Pap Rep 27:311–320.
Uallah S, Ahmad T, Bilal MQ, Zia-ur-Rahman , Muhammad G. 2005. The effect of severity of mastitis on protein and fat contents of buffalo milk. Pak Vet J 25:1–4.
- TOOLS








 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print





