 |
 |
| Anim Biosci > Volume 30(9); 2017 > Article |
|
Abstract
Objective
Phellodendron amurense (P. amurense) and Humulus japonicus (H. japonicus) are closely involved in anti-oxidative response and increasing antioxidant enzymes activities. However, the effects of their extracts on development of preimplantation bovine embryos have not been investigated. Therefore, we investigated the effects of P. amurense and H. japonicus extracts on developmental competence and quality of preimplantation bovine embryos.
Methods
After in vitro fertilization, bovine embryos were cultured for 7 days in Charles Rosenkrans amino acid medium supplemented with P. amurense (0.01 μg/mL) and H. japonicus (0.01 μg/mL). The effect of this supplementation during in vitro culture on development competence and antioxidant was investigated.
Results
We observed that the blastocysts rate was significantly increased (p<0.05) in P. amurense (28.9%±2.9%), H. japonicus (30.9%±1.5%), and a mixture of P. amurense and H. japonicus (34.8%± 2.1%) treated groups compared with the control group (25.4%±1.6%). We next confirmed that the intracellular levels of reactive oxygen species (ROS) were significantly decreased (p<0.01) in P. amurense and/or H. japonicus extract treated groups when compared with the control group. Our results also showed that expression of cleaved caspase-3 and apoptotic cells of blastocysts were significantly decreased (p<0.05) in bovine blastocysts derived from both P. amurense and H. japonicus extract treated embryos.
In vitro production of mammalian embryos is essential for breeding, infertility therapy and fertility management in domestic animals [1]. However, in vitro produced (IVP) embryos cultivated under suboptimal culture conditions show decreased developmental competence and quality compared with in vivo produced embryos, [2–4]. Thus, many studies were conducted to improve the quality of IVP embryos via improvement of culture medium with growth factors and anti-oxidants.
In vitro production of mammalian embryos is commonly conducted under 5% CO2 in air. However, IVP embryos grown under high O2 concentrations have poor blastocysts qualities because of the increased accumulation of reactive oxygen species (ROS) in mammalian embryos [5]. ROS, such as superoxide anions (·O2−), hydrogen peroxide (H2O2), hydroxyl radicals (·OH) and induced oxidative stress, are known to damage the lipids, proteins and nucleic acids [6]. Therefore, the addition of antioxidants into culture medium improves developmental competence and qualities of IVP embryos.
Apoptosis is a programmed type of cell death that plays impor tant roles in homeostasis and elimination of damaged cells [7]. In general, apoptosis is related to embryonic development and growth. Additionally, it is an important indicator of inadequate conditions for mammalian embryo development [8]. In addition, apoptosis is greater in IVP embryos than in vivo produced embryos. Therefore, decreasing apoptotic cells in blastocysts indicate improved culture conditions.
Phellodendron amurense (P. amurense) and Humulus japonicus (H. japonicus), which are usually found in forests of China, Korea, and Japan, are widely used in traditional medicine to treat pneumonia, diarrhea, pulmonary, tuberculosis and dysentery, as well as to promote blood circulation [9,10]. Extracts of P. amurense and H. japonicus have recently been reported to contain isoquinoline, alkaloids, phenolic compounds, flavone glycosides, terpenes, lupulones, phenolics and flavonoids [11–13]. Moreover, recent studies have shown that P. amurense and H. japonica extracts possess anti-oxidant functions [11,13]. However, the antioxidant effects of P. amurense and H. japonica extracts during preimplantation development of bovine embryos has not been thoroughly investigated.
Melatonin (N-acetyl-5-methoxytryptamine) is synthesized by the pineal gland in the brain [14], and its secretion is dependent on the sleep-wake cycle, with the highest levels occurring at night [15]. Melatonin acts as a direct scavenger of toxic ROS, and also has the ability to decrease the formation of ROS. This compound also induces the activity of antioxidant enzymes and protects against the damage that may occur in response to oxidative stress. In particular, it plays an important role in decreasing and protecting against mitochondrial oxidative stress, as well as reducing apoptosis [16]. In addition, many studies have reported that melatonin enhances mouse, porcine, bovine and human embryo developmental competence [17–19]. Thus, we used melatonin as a positive control to confirm the effects of P. amurense and/or H. japonicus extracts on developmental competence through their roles as anti-oxidants in bovine embryos.
The present study was conducted to investigate the effects of addition of P. amurense and H. japonicus into culture medium on the developmental competence of preimplantation bovine embryos. We also assessed the effects of P. amurense and H. japonicus on elimination of ROS. Finally, the expression of apoptotic factor (cleaved caspase-3) and the index of apoptotic cells were investigated in bovine blastocysts after treatment with P. amurense and H. japonicus extracts.
Unless otherwise stated, all chemicals used in this study were purchased from Sigma Chemical Co. (St. Louis, MO, USA)
P. amurense and H. japonicus extracts were obtained from Kangwon National University. Approximately 300 g of the dried P. amurense and H. japonicas were boiled in 3 L of methanol for 4 h. The extracts were then filtered using absorbent cotton, after which the solvent was removed from the filtered extracts by rotary vacuum evaporation at 40°C. Dried extracts were subsequently stored at room temperature until analysis. The extracts were dissolved in dimethyl sulfoxide before use.
Bovine ovaries were collected from a local slaughterhouse (Gyeongsan, Gyeongbuk, Korea) and then transported to the laboratory in 0.9% saline supplemented with 75 μg/mL potassium penicillin G while maintained at 30°C to 35°C. Cumulus-oocyte complexes (COCs) were aspirated from 3 to 6 mm follicles using a disposable 10 mL syringe with an 18-gauge needle, after which COCs with surrounding cumulus cells and homogeneous cytoplasm were selected. The COCs were then washed three times in the Tyrode’s lactate-N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (TL-HEPES) and twice in in vitro maturation (IVM) medium, after which 50 immature COCs were matured in 500 μL of IVM medium in a four-well multi-dish (Nunc, Roskilde, Denmark) for 20 to 22 h at 38.5°C under 5% CO2 in air. The medium used for oocyte maturation was TCM-199 (Gibco Life Technologies, Inc., Grand Island, NY, USA) supplemented with 10% (v/v) fetal bovine serum (FBS; Gibco Life Technologies, Inc., USA), 10 IU/mL pregnant mare serum gonadotropin, 10 IU/mL human chorionic gonadotropin, 0.6 mM cysteine, 0.2 mM sodium pyruvate, 10 ng/mL epidermal growth factor, 25 μM β-mercaptoethanol, 25 μg/mL gentamycin and 1 μg/mL 17 β-estradiol. After culturing for IVM, 15 oocytes were fertilized with sperm that had been frozen and thawed at 2×106 cells/mL in 50 μL fertilization medium (Fert-TALP). When the sperm were added to the fertilization drops, 2 μg/mL heparin, 20 μM penicillamine, 10 μM hypotaurine, and 1 μM epinephrine were also added. At 22 h after insemination, the cumulus-enclosed oocytes were detached using gentle pipetting and then transferred to Charles Rosenkrans amino acid (CR1aa) medium containing 0.3% bovine serum albumin (BSA), 1× basal medium Eagle amino acids, and 1× minimum essential medium nonessential amino acids for in vitro culture (IVC). After culture for 3 days, we further cultured the cleaved embryos in a medium consisting of 50 μL of CR1aa (with 10% FBS) for 4 days at 38.5°C under 5% CO2 in air. During the culture periods, presumptive embryos were treated with 0.01 μg/mL P. amurense and/or 0.01 μg/mL H. japonicus [20] and 0.1 μM melatonin [21] by the addition to the culture medium. The rates of cleavage and blastocyst stage embryos were determined on day 3 and 7, respectively.
The ROS levels in blastocysts were measured using the dichlorodihydrofluorescein diacetate method (H2DCFDA; Invitrogen Molecular Probes, Eugene, OR, USA) as previously described [5]. Blastocysts were transferred into CR-1aa (with 10% FBS) medium containing 5 μM H2DCFDA for 20 min at 38.5°C under 5% CO2 in air. The permeabilized blastocysts in H2DCFDA were washed three times with 0.1% polyvinyl alcohol (PVA) in phosphate buffer solution (PBS). The fluorescent images were detected by epifluorescence microscopy (IX 51; Olympus, Tokyo, Japan) and then analyzed using the Image J software 1.38 (National Institutes of Health, Bethesda, MD, USA).
Blastocysts were washed with 0.1% PVA in PBS, then fixed in 4% paraformaldehyde (PFA) in PBS for 1 h at room temperature, after which they were made permeable by incubation with 0.2% Triton X-100 at room temperature and then incubated in 0.1% PVA in PBS containing 1% BSA overnight at 4°C. Samples were then incubated in 10% FBS in PBS (v/v) for 45 min at 37°C, then incubated with cleaved caspase-3 antibodies (cat. no. 9664; Cell Signaling Technology, Danvers, MA, USA) diluted 1:2,500 overnight at 4°C and washed with 0.1% PVA in PBS at least three times. The sample was subsequently reacted with the secondary antibodies, rhodamine-conjugated goat anti-rabbit immunoglobulin (Ig) G (cat. no. 31463; Thermo Scientific, Rockford, IL, USA), then diluted 1:50 for 2 h at room temperature. After washing, the samples were incubated in Hoechst 33342 (w/v) for 20 min at room temperature. Finally, the samples were mounted on glass slides and examined with a confocal laser-scanning microscope (Zeiss LSM 700 META; Carl Zeiss, Jena, Germany).
Apoptotic cells among blastocysts were detected using an In Situ Cell Death Detection Kit (Roche Diagnostics GmbH, Mannheim, Germany) according to the manufacturer’s instructions. At Day 7, the IVP blastocysts were washed with 0.1% PVA in PBS, then fixed in 4% PFA (v/v) in PBS for 1 h at room temperature. Blastocysts were then permeabilized using 0.1% (v/v) Triton X-100 for 30 min at 4°C. The fixed embryos were subsequently incubated in terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) reaction medium for 1 h at 38.5°C, then washed and mounted on slides. Whole-mount embryos were examined under an epifluorescence microscope (Olympus, Japan) following a TUNEL assay and DAPI staining to determine the numbers of apoptotic nuclei and total numbers of nuclei.
All percentage data obtained in this study are presented as the means±standard deviation. Moreover, of the results of the H2DCFDA experiments are presented as the means±standard error of the mean. All experiments were replicated three times, after which the results were analyzed by one-way analysis of variance followed by Bonferroni’s Multiple Comparison Test using t-tests. All data analysis was performed using the GraphPad Prism 5.0 software package (San Diego, CA, USA). Differences were considered significant at * p<0.05, ** p<0.01, and *** p<0.001.
Here, we evaluated bovine embryos grown in culture medium supplemented with various concentrations (0.01, 0.05, and 0.1 μg/mL) of P. amurense for 6 days. We then confirmed that the developmental competence of bovine embryos increased in response to 0.01 μg/mL P. aamurense extract (Table 1). We also used the optimal H. japonicus extract concentration as defined in a previous study that effects of H. japonicus extract on sperm motility and fertilization status in cattle [20]. Based on this report, we investigated the effects of P. amurense and/or H. japonicus extracts on bovine embryonic development. As shown in Figure 1, the blastocyst development rates were significantly increased (p<0.05) in embryos from P. amurense (28.9%±2.9%) or H. japonicus (30.9% ±1.5%) extract treated groups compared with the control (25.4% ±1.6%), respectively. Moreover, blastocysts development was significantly higher (p<0.05) in both the P. amurense and H. japonicus treated group (34.8%±2.1%) than in the control groups. In addition, this result in both the P. amurense and H. japonicus treated group appeared similar to that of the melatonin treated group (35.7%±5.1%). These results indicate that P. amurense and/or H. japonicus extracts enhanced developmental competence of bovine embryos.
Balanced intracellular ROS levels are very important for improving the efficiency of IVP bovine embryos [22]. Therefore, we measured the intracellular ROS levels in blastocysts from the bovine embryos of IVP treated with P. amurense and/or H. japonicus extract by dichlorodihydrofluorescein diacetate (H2DCFDA) staining to determine if they reduced the intracellular ROS levels of bovine blastocysts generated by oxidative stress. As shown in Figure 2, the intracellular ROS levels were significantly decreased (p<0.05) in P. amurense and H. japonicus extract treated groups when compared with the control group. Moreover, the intracellular ROS levels in bovine blastocysts derived from the P. amurense and H. japonicus extract treated groups were significantly lower (p<0.01) than those of other treatment groups. This result also appeared similar to that of the melatonin treated group. These results suggest that P. amurense and/or H. japonicus extracts improve developmental competence of bovine embryos through elimination of ROS levels during IVC.
Cleaved caspase-3 plays an important role in the apoptotic pathway of animal cells [8]. Therefore, we investigated the expression of cleaved caspase-3 in bovine blastocysts treated with both P. amurense and H. japonicus extracts. As shown in Figure 3, the expression of cleaved caspase-3 decreased in response to treatment with either P. amurense and H. japonicus extracts (1.9±1.0) or melatonin (1.8±0.6) relative to the control group (3.1±1.0). We also confirmed the presence of apoptotic cells in P. amurense and H. japonicus extract treated blastocysts by a TUNEL assay. As shown in Figure 4, no significant differences were detected in total nuclei among the control (112.5±14.8), P. amurense (117.7±14.9) or H. japonicus (114.8±11.7) extract treated groups. However, total nuclei increased significantly (p<0.05) in both the P. amurense and H. japonicus extract treated groups (141.2±15.8). Moreover, apoptotic nuclei significantly decreased (p<0.05) in the P. amurense (2.9%±0.7%) and H. japonicus (3.1%±1.1%) extract treated groups when compared with the control group (4.4%±1.0%). Notably, the apoptosis rate in both the P. amurense and H. japonicus extracts treated group (1.4%±0.6%) or the melatonin treated group (1.3%± 0.7%) was significantly lower (p<0.05) than in the other treatment groups. Therefore, simultaneous treatment with two extracts produced results similar to melatonin treated groups with respect to developmental competence, ROS level and apoptotic index in bovine blastocysts of IVP.
Taken together, these results suggest that treatment with P. amurense and/or H. japonicus extracts improves blastocyst quality by reducing cellular apoptosis.
This study demonstrated that the P. amurense and H. japonicus extracts were an effective means to induce developmental competence and blastocyst quality in bovine embryos. We also confirmed that the intracellular ROS levels were reduced by the anti-oxidant effects of P. amurense and H. japonicus extracts in bovine blastocysts. Moreover, the expression of apoptotic factor (cleaved caspase-3) and apoptotic nuclei decreased in bovine blastocysts derived from P. amurense and H. japonicus extracts treated embryos in vitro. Additionally, we confirmed that these results in both the P. amurense and H. japonicus extract treated group showed a similar response to the melatonin treated group, a known antioxidant.
Synthetic phenolic antioxidants are widely used as antioxidant additives in various studies because of their effects and cost effectiveness. However, they are known to cause an aversion to com-posite food and have toxic effects [23]. Therefore, many studies have investigated antioxidants by using natural products. Natural antioxidants include vitamin C, γ-tocotrienol, flavonoid and phenolic acid. P. amurense and H. japonicus extracts, which have been used as traditional medicine in Korea and China, include flavonoid and polyphenolic substances [11,12]. According to recent studies, P. amurense glycan prevented skin oxidative stress in ultraviolet radiation by reducing lipid peroxidation and increasing antioxidant enzymes activities [13], while H. japonicus improved antioxidant enzyme activities in human fibroblast cells [11]. In a previous study, we confirmed that H. japonicus extract improves early bovine embryo development [20]. In the present study, we also confirmed that P. amurense extract (0.01 μg/mL) enhances preimplantation embryo development in cattle. Therefore, we identified the anti-oxidant effects of treatment with P. amurense and H. japonicus extracts during IVC of bovine embryos. The rate of blastocyst development was significantly higher in the P. amurense or H. japonicus extract treatment groups than the control group (Figure 1). Moreover, both the P. amurense and H. japonicus extract treatment groups were similar to the melatonin treatment group (Figure 1). A previous study reported that melatonin enhanced blastocyst development in vitro [21]. Taken together, these results demonstrated the addition of P. amurense and H. japonicus extracts during IVC enhances the developmental competence of bovine embryos.
Generally, intracellular ROS levels generated in embryos in creased during IVC [22]. ROS play important roles as positive or negative factors influencing embryonic development [24]. However, IVP embryo quality is known to be reduced by increasing ROS levels produced by the relatively higher oxygen levels and lower levels of free radical scavengers when compared with in vivo conditions [25]. Therefore, we identified the expression of intracellular ROS levels in bovine blastocysts derived from P. amurense and/or H. japonicus extract treated embryos. In the present study, the intracellular ROS levels decreased significantly in both the P. amurense and H. japonicus extract treatment groups relative to the control group (Figure 2). Moreover, the ROS levels in both the P. amurense and H. japonicus extract treatment groups were similar to those in the melatonin treatment groups (Figure 2). A previous study demonstrated that melatonin was reduced in intracellular ROS levels of blastocysts in vitro [26]. These results suggest that treatment with P. amurense and H. japonicus extract decreases intracellular ROS levels of bovine blastocysts in vitro.
Apoptosis is an important indicator used in evaluation of blastocyst quality [8]. In a previous study, we demonstrated that decreasing apoptosis enhanced blastocyst quality, while high apoptosis rate reduced blastocyst quality [27]. Apoptosis, which is known to be one of the mechanisms involved in the mitochondrial pathway, is activated by caspase-8 and -9 via the mitochondrial pathways. Cleaved caspase-3 induced the increasing of cleaved poly (ADP-ribose) polymerase (PARP) involved in DNA repair and induced apoptosis [8]. In the present study, the expression of cleaved caspase-3 was decreased in both the P. amurense and H. japonicus extracts treatment groups, similar to the melatonin treatment group (Figure 3). The apoptosis rate was significantly decreased in the P. amurense or H. japonicus extract treatment groups relative to the control group. Moreover, both the P. amurense and H. japonicus extract treatment groups were similar to the melatonin treatment group (Figure 4). A previous report demonstrated that melatonin reduced apoptosis in IVP blastocysts [28]. These findings suggest that treatment with P. amurense and H. japonicus extracts reduces apoptotic nuclei of bovine blastocysts through the regulation of caspase-3 expression in vitro. In summary, the present study provides the first evidence associating P. amurense and H. japonicus extracts treatment with the promotion of bovine embryos developmental competence in vitro. We also found that the P. amurense and H. japonicus extracts increased the qualities of bovine blastocysts by reducing intracellular ROS and apoptosis. Moreover, the effects they induced on developmental competence, elimination of ROS, and apoptosis were similar to those induced by melatonin as a known antioxidant. Overall, these results suggest that P. amurense and H. japonicus extracts improve preimplantation embryos development and quality of bovine blastocysts in vitro. Moreover, our results suggest that natural plant extracts may enable improvement of embryo quality for infertile patients.
ACKNOWLEDGMENTS
This work was supported by grants from the Next-Generation BioGreen 21 Program (PJ01117604) and the Bio-industry Technology Development Program (316037-04-2-HD020) through the Rural Development Administration, the Ministry of Agriculture, Food and Rural Affairs, and the KRIBB Research Initiative Program (KGM4251723), Republic of Korea.
Figure 1
Effect Phellodendron amurense and/or Humulus japonicus extracts on preimplantation development of bovine embryos in vitro. (A) Diagram of cleavage stage and blastocyst formation pattern after in vitro fertilization (IVF). (B) Summary of bovine embryonic development after IVF. Data are the means±standard deviation. a–c Values from three times replicates with different superscripts denote a significant difference relative to other groups (p<0.05).

Figure 2
Detection of ROS level in bovine blastocysts derived from Phellodendron amurense and/or Humulus japonicus extract treated embryos by H2DCFDA staining. (A) Fluorescence microscopy imaging of ROS production in bovine blastocysts by H2DCFDA staining. (B) Quantification of ROS levels in bovine blastocysts. Scale bars = 200 μm. Quantification of fluorescence intensity in H2DCFDA stained bovine blastocysts was obtained using the Image J software. This experiment was replicated at three times. Data in the bar graph represent the means±standard error of the mean of three independent experiments. Statistically significant differences are indicated by asterisks (* p<0.05, ** p<0.01, and *** p<0.001, compared to control group). ROS, reactive oxygen species; H2DCFDA, dichlorodihydrofluorescein diacetate.
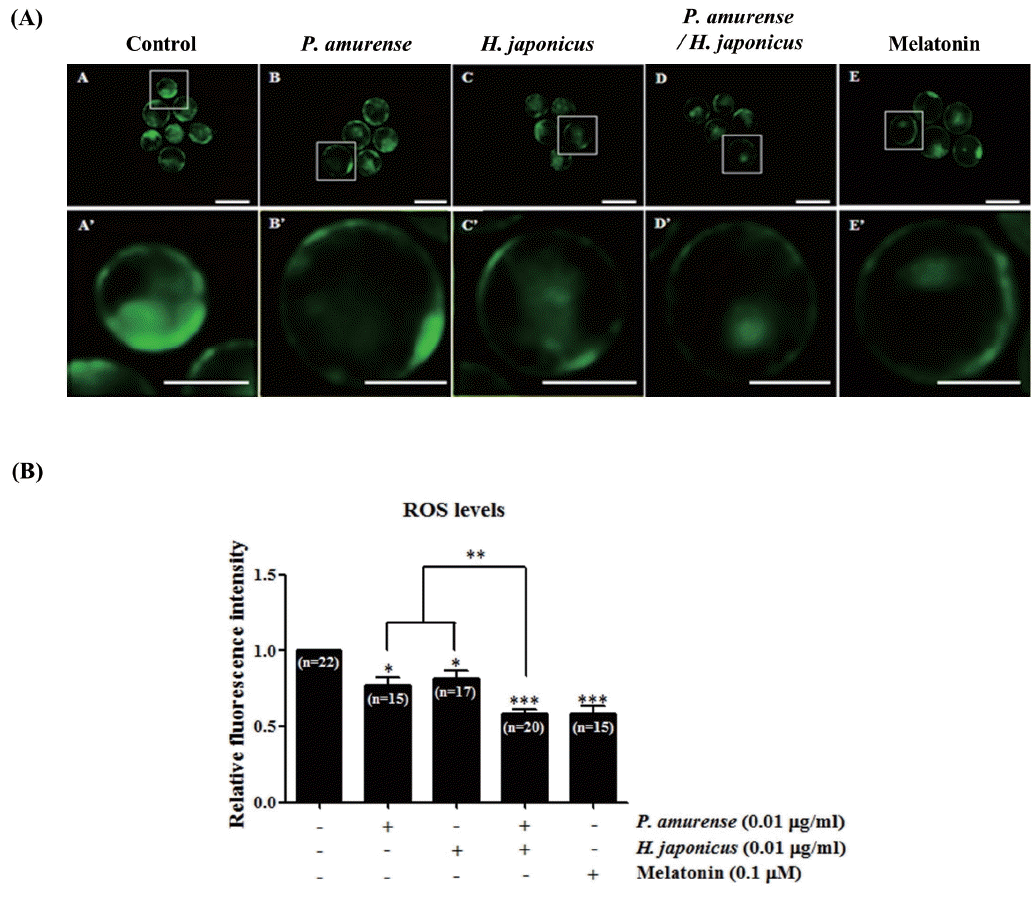
Figure 3
Detection of cleaved caspase-3 in bovine blastocysts derived from Phellodendron amurense and Humulus japonicus extract treated embryos by immunofluorescence staining. (A) A–C: the chromatin content was determined by Hoechst staining (total DNA), A’–C’: cleaved caspase-3 (#9664; Cell Signaling) was labeled with Alexa Fluor 555 in fluorescence in bovine blastocysts. Scale bars = 20 μm. (B) Summary of total cells and cleaved caspase-3 cells pattern in bovine blastocysts. Data in the bar graph represent the means± standard deviation of three independent experiments. a,b Values from three replicates with different superscripts denote a significant difference relative to other groups (p<0.05).
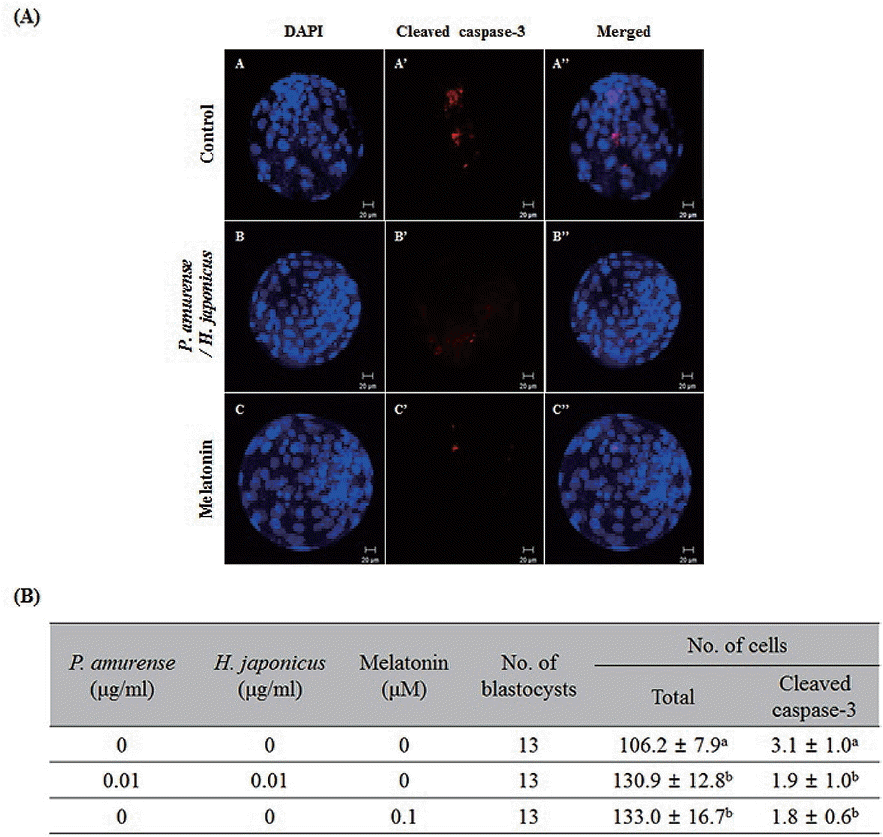
Figure 4
Evaluation of cellular apoptosis in bovine blastocysts derived from Phellodendron amurense and/or Humulus japonicus extract treated embryos by TUNEL assay. (A) Detection of apoptosis fluorescent images in bovine blastocysts by TUNEL assay. A–E: the chromatin content was determined by DAPI staining (total DNA); A’–E’: fragmented DNA detected by TUNEL (apoptotic nuclei, white arrows). Scale bars = 100 μm. (B) Percent of TUNEL positive cell per blastocyst and (C) total cell number per blastocyst. (D) Summary of apoptosis pattern in bovine blastocysts. Data in the bar graph represents the means±standard deviation of three independent experiments. a–c Values from three replicates with different superscripts denote a significant difference relative to other groups (p<0.05). TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling; DAPI, 4′,6-diamidino-2-phenylindole.
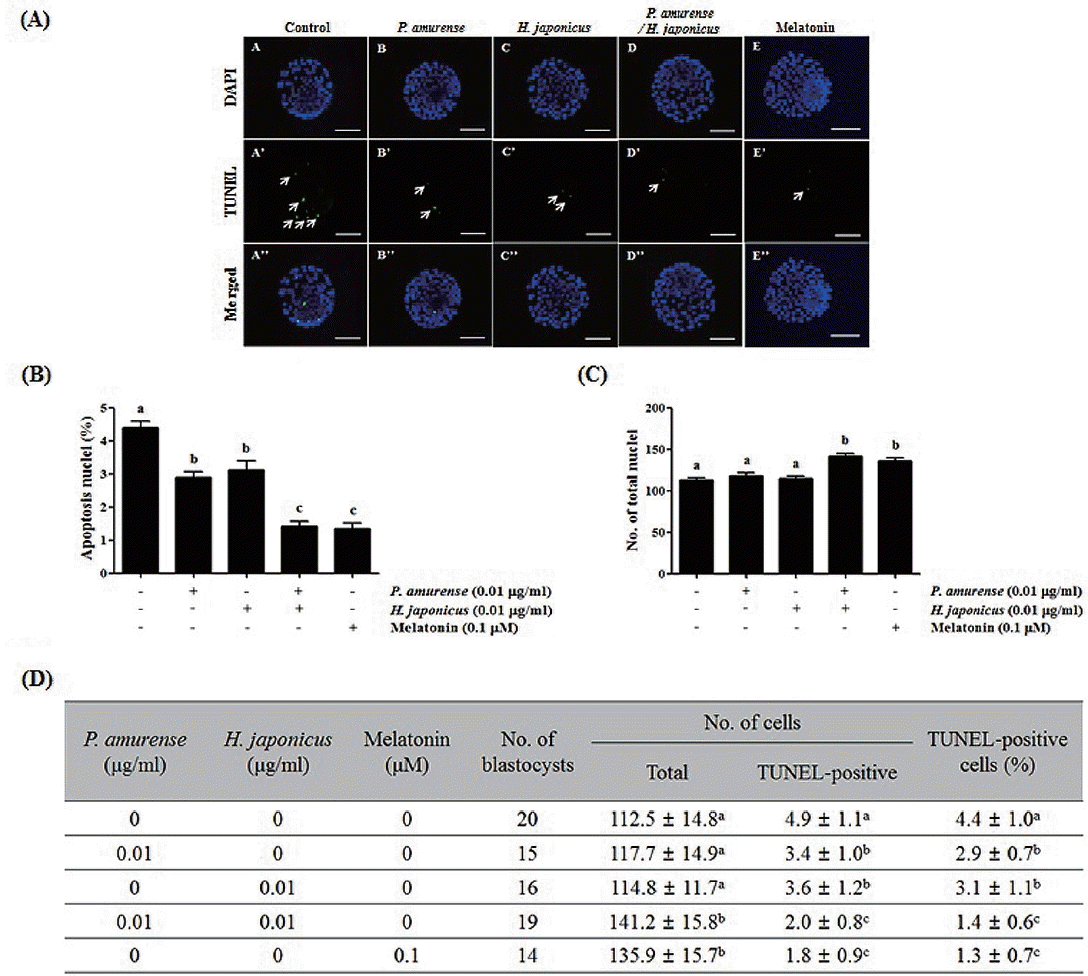
Table 1
Effect of various concentrations of Phellodendron amurense extract on preimplantation development of bovine embryos in vitro
REFERENCES
1. Zullo G, Albero G, Neglia G, et al. L-ergothioneine supplementation during culture improves quality of bovine in vitro-produced embryos. Theriogenology 2016; 85:688–97.


2. Boni R, Tosti E, Roviello S, Dale B. Intercellular communication in in vivo-and in vitro-produced bovine embryos. Biol Reprod 1999; 61:1050–5.



3. Leibfried-Rutledge ML, Critser ES, Eyestone WH, Northey DL, First NL. Development potential of bovine oocytes matured in vitro or in vivo. Biol Reprod 1987; 36:376–83.


4. Rizos D, Ward F, Duffy P, Boland MP, Lonergan P. Consequences of bovine oocyte maturation, fertilization or early embryo development in vitro versus in vivo: implications for blastocyst yield and blastocyst quality. Mol Reprod Dev 2002; 61:234–48.


5. Lee KS, Kim EY, Jeon K, et al. 3,4-Dihydroxyflavone acts as an antioxidant and antiapoptotic agent to support bovine embryo development in vitro. J Reprod Dev 2011; 57:127–34.


6. Thannickal VJ, Fanburg BL. Reactive oxygen species in cell signaling. Am J Physiol Lung Cell Mol Physiol 2000; 279:L1005–28.


8. Betts DH, King WA. Genetic regulation of embryo death and senescence. Theriogenology 2001; 55:171–91.


9. Kumar AP, Bhaskaran S, Ganapathy M, et al. Akt/cAMP-responsive element binding protein/cyclin D1 network: a novel target for prostate cancer inhibition in transgenic adenocarcinoma of mouse prostate model mediated by Nexrutine, a Phellodendron amurense bark extract. Clin Cancer Res 2007; 13:2784–94.



10. Naya Y, Kotake M. The Constituents of Hops. V. The Volatile Composition of Humulus japonicus Sieb. et Zucc. Bull Chem Soc Japan 1970; 43:3594–6.

11. Sung B, Chung JW, Bae HR, et al. Humulus japonicus extract exhibits antioxidative and anti-aging effects via modulation of the AMPK-SIRT1 pathway. Exp Ther Med 2015; 9:1819–26.



12. Wang W, Li Q, Liu Y, Chen B. Ionic liquid-aqueous solution ultrasonic-assisted extraction of three kinds of alkaloids from Phellodendron amurense Rupr and optimize conditions use response surface. Ultrason Sonochem 2015; 24:13–8.


13. Yan H, Sun X, Sun S, et al. Anti-ultraviolet radiation effects of Coptis chinensis and Phellodendron amurense glycans by immunomodulating and inhibiting oxidative injury. Int J Biol Macromol 2011; 48:720–5.


14. Dehghani-Mohammadabadi M, Salehi M, Farifteh F, et al. Melatonin modulates the expression of BCL-xl and improve the development of vitrified embryos obtained by IVF in mice. J Assist Reprod Genet 2014; 31:453–61.



15. Niknafs B, Mehdipour A, Mohammadi Roushandeh A. Melatonin improves development of early mouse embryos impaired by actinomycin-D and TNF-alpha. Iran J Reprod Med 2014; 12:799–804.


16. Mehaisen GM, Saeed AM, Gad A, et al. Antioxidant capacity of melatonin on preimplantation development of fresh and vitrified rabbit embryos: morphological and molecular aspects. PLoS One 2015; 10:e0139814



17. Rodriguez-Osorio N, Kim IJ, Wang H, Kaya A, Memili E. Melatonin increases cleavage rate of porcine preimplantation embryos in vitro. J Pineal Res 2007; 43:283–8.


18. Papis K, Poleszczuk O, Wenta-Muchalska E, Modlinski JA. Melatonin effect on bovine embryo development in vitro in relation to oxygen concentration. J Pineal Res 2007; 43:321–6.


19. Ishizuka B, Kuribayashi Y, Murai K, Amemiya A, Itoh MT. The effect of melatonin on in vitro fertilization and embryo development in mice. J Pineal Res 2000; 28:48–51.


20. Min SH, Kim JW, Do GY, et al. Effect of Humulus japonicus extract on sperm motility, fertilization status and subsequent preimplantation embryo development in cattle. Reprod Dev Biol 2014; 38:115–21.

21. Wang F, Tian X, Zhou Y, et al. Melatonin improves the quality of in vitro produced (IVP) bovine embryos: implications for blastocyst development, cryotolerance, and modifications of relevant gene expression. PLoS One 2014; 9:e93641



22. Takahashi M. Oxidative stress and redox regulation on in vitro development of mammalian embryos. J Reprod Dev 2012; 58:1–9.


23. Branen AL. Toxicology and biochemistry of butylated hydroxyanisole and butylated hydroxytoluene. J Am Oil Chem Soc 1975; 52:59–63.


24. Dennery PA. Effects of oxidative stress on embryonic development. Birth Defects Res C Embryo Today 2007; 81:155–62.


25. Menezo Y, Dale B, Cohen M. DNA damage and repair in human oocytes and embryos: a review. Zygote 2010; 18:357–65.


26. Wang F, Tian X, Zhang L, et al. Beneficial effects of melatonin on in vitro bovine embryonic development are mediated by melatonin receptor 1. J Pineal Res 2014; 56:333–42.


- TOOLS





 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print





